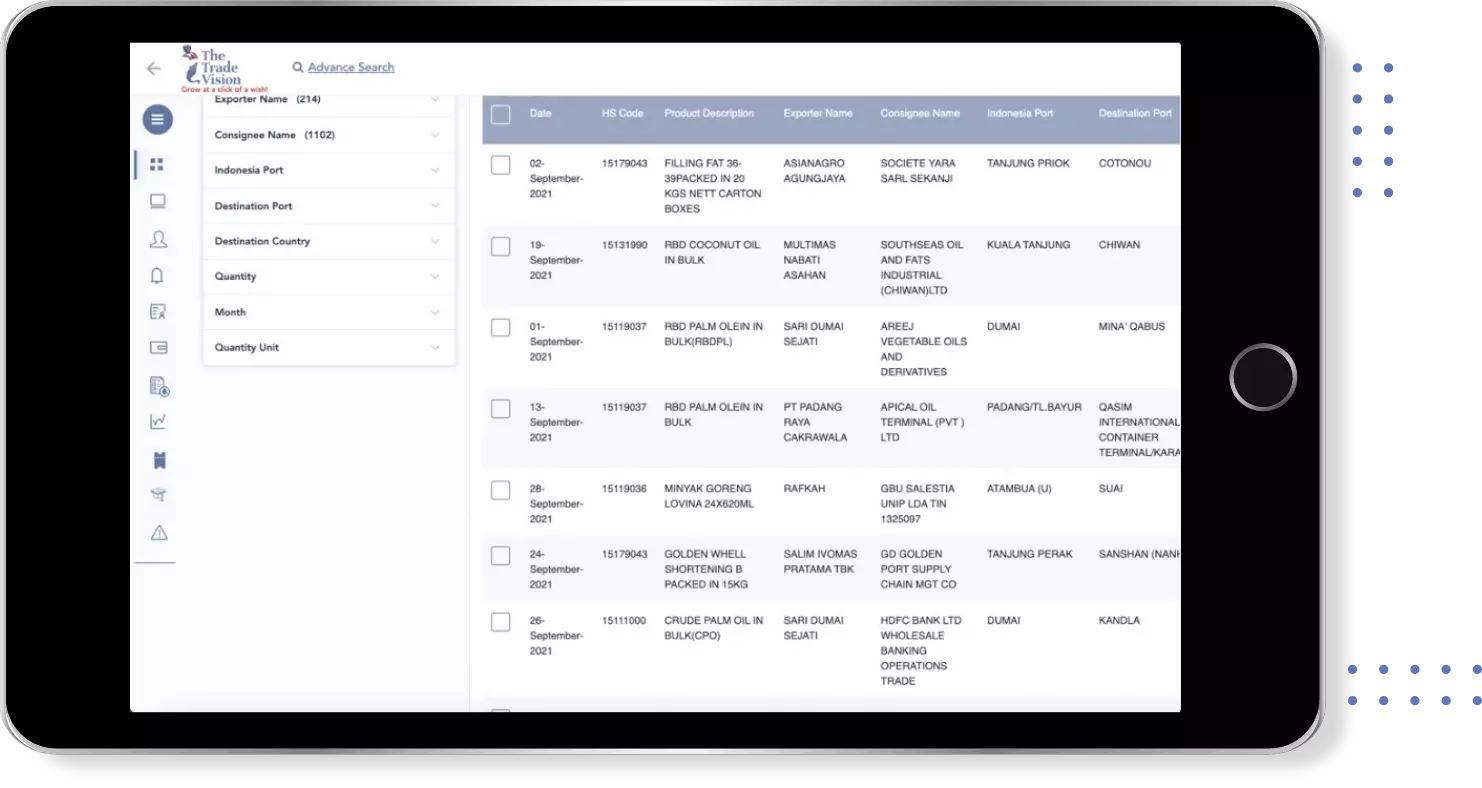
Find verified buyers and sellers of each capsule in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- each capsule hs code 300490 import export data
Overview
- The top 3 supplier countries for HS Code 300490 are “BRAZIL” , “NIGERIA” , “AFGHANISTAN” ,
- The top 3 Buyer countries for HS Code 300490 are “GERMANY” , “FRANCE” , “UNITED STATES OF AMERICA” , .
- The top 3 Importing countries for HS Code 300490 are “BELGIUM” , “UNITED STATES OF AMERICA” , “UNITED KINGDOM” , .
- The top 3 Exporting countries for HS Code 300490 are “GERMANY” , “UNITED STATES OF AMERICA” , “UKRAINE” , .
- Top 1 Product Categories for HS Code 300490 are 300490,
The above summary is based on TTV’s Global Export Import data of HS Code 300490, compiled from 180+ countries export import shipments updated till .
- HS Code: 300490
Global Buyers of each capsule under HS-Code 300490
PPD PHARMACEUTICAL DEVELOPMENT PHIL

UNITED STATES OF AMERICA
HS-Code
Products
300490990000
TREPROSTINIL PALMITIL INHALATION POWDER 160MG OR PLACEBO/35 CAPSULES EACH BOTTLE
300490990000
TREPROSTINIL PALMITIL INHALATION POWDER 320MG OR PLACEBO/35 CAPSULES EACH BOTTLE
300490990000
TREPROSTINIL PALMITIL INHALATION POWDER 80MG OR PLACEBO/35 CAPSULES EACH BOTTLE
ROCHE FARMA (PERU) SA

GERMANY
HS-Code
Products
3004902400
ALECENSA MEDICINE S/M ALECTINIB HYDROCHLORIDE 161.33 MG RD 12128-2017/DIGEMID OF 12/14/2017 MEDICATION FOR HUMAN USE TO TREAT LUNG CANCER BOX CONTAINING 4 BOXES AND EACH INDIVIDUAL BOX CONTAINING 56 HARD CAPSULES 10231266 ALECENSA CAPSULES (HARD) 150 MG 224 LOT B1445M6 VCTO:05/2024
3004902400
MEDICATION S / MS / M 21 PALBOCICLIB HARD CAPSULES 100 MG MEDICINE FOR HUMAN USE FOR USE IN CLINICAL TRIALS. MEDICINE AGAINST BREAST CANCER. PROTOCOL BO41843 3 BLISTERS WITH 7 CAPSULES EACH RO499-1855 PALBOCICLIB 100 MG LOT: EC0815 1169016 F.VENC: 03.31.2024
3004902400
MEDICATION S / MS / M 21 PALBOCICLIB HARD CAPSULES 75 MG MEDICINE FOR HUMAN USE FOR USE IN CLINICAL TRIALS. MEDICINE AGAINST BREAST CANCER. PROTOCOL BO41843 3 BLISTERS OF 7 CAPSULES EACH RO499-1855 PALBOCICLIB 75 MG LOT: DX2895 1167606 F.VENC: 04.30.2024
ALLMED LABORATORIES

CHINA
HS-Code
Products
30049099
CYCLOSPORINE CAPSULES (BIORAL 100MG) BATCH NO: C210402 MFG DT:APR 2021 EXP DT:APR 2023 REG NO:039853 = 1000 BOXES. (EACH BOX CONTAINS 50 CAPSULES) EACH BOX CONTAINS:) 1500 BOXES REF GD NO:KPAF-HC-57650 DT:28052021).
30049099
CYCLOSPORINE CAPSULES BIORAL 100 MG BATCH NO C180303 MFG DATE MAR 2018 EXP DATE 2020 1000 BOXES EACH BOX CONTAINS 50 CAPSULES P.O NO.AL002417
30049099
CYCLOSPORINE CAPSULES BIORAL 25 MG BATCH C171001 MFG. DATE OCT.2017 EXPDATE 0CT 2019 3000 BOXES EACH BOX CONTAINS 50 CAPSULES.P NO AL 002517
HOSPITAL K

SWEDEN
HS-Code
Products
3004909900
ACALABRUTINIB (ACP-196) 100MG OR PLACEBO CAPSULE EACH VIAL CONTAINS 30 TABLETS. LOT NUMBER E158870-0098L. EXPIRY DATE IS OCTOBER 31 2023. MANUFACTURER ASTRAZENECA AB. NEW 100%
3004909900
ACALABRUTINIB (ACP-196) 100MG OR PLACEBO CAPSULE EACH VIAL CONTAINS 30 TABLETS. LOT NUMBER E158870-0098L. EXPIRY DATE IS OCTOBER 31 2023. MANUFACTURER ASTRAZENECA AB. NEW 100%
30049099
ACALABRUTINIB (ACP-196) 100MG OR PLACEBO CAPSULE EACH VIAL CONTAINS 30 TABLETS. LOT NUMBER E158870-0103L. EXPIRY DATE IS 31/07/2024. MANUFACTURER ASTRAZENECA AB. NEW 100%
Global Suppliers of each capsule under HS-Code 300490
LABORATORIOS SAVAL SA

PARAGUAY
HS-Code
Products
30049092
ANALGESIC ANTIEPILEPTICONEURUMCAPSULES X 6 EACH CAPSULE CONTAINS 50 MG OF PREGABALIN DRUG FOR HUMAN USE. R-1 7
30049052
ANTIDEPRESSIVE ALTACAPSULES X 7 EACH CAPSULE CONTAINS 30 MG OF DULOXETINE HYDROCHLORIC DRUG FOR HUMAN USE. R-1 7
30049052
ANTIDEPRESSIVE ALTACAPSULES X 7 EACH CAPSULE CONTAINS 30 MG OF DULOXETINE HYDROCHLORIC DRUG FOR HUMAN USE. R-1 7
SAVAL LABORATORIES S A

PARAGUAY
HS-Code
Products
30049058
ANTIDEPRESSIVE ACTAN CAPSULES X 60 EACH CAPSULE CONTAINS 20 MG OF FLUOXETINE MEDICATION FOR HUMAN USE. R-1 7
30049042
ANTI-INFLAMMATORY NON-STEROIDALGESIXCAPSULES X 1 0 EACH CAPSULE CONTAINS 200 MG CELECOXIB MEDICINE FOR HUMAN USE. R-1 7
30049042
NON-STEROID ANTI-INFLAMMATORY GESIX CAPSULES X 30 EACH CAPSULE CONTAINS 200 MG CELECOXIB MEDICATION FOR HUMAN USE. R-1 7
SAVAL LABORATORIES SA

ECUADOR
HS-Code
Products
30049052
ANTIDEPRESSIVE ALTACAPSULES X 30 EACH CAPSULE CONTAINS 60 MG OF DULOXETINE HYDROCHLORIC DRUG FOR HUMAN USE. R-1 7
30049051
ANTIDEPRESSIVE ACTAN CAPSULES X 30 EACH CAPSULE CONTAINS 20 MG OF FLUOXETINE MEDICATION FOR HUMAN USE. R-1 7
30049092
ANALGESICO ANTIEPILEPTICONEURUMCAPSULAS X 30 EACH CAPSULE CONTAINS 50 MG OF PREGABALINE DRUG FOR HUMAN USE. R-1 7
PHARM START LTD

KAZAKHSTAN
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE. - BIFREN CAPSULES NO. 20 TABLETS 250MG EACH. BY 1 0 TABLETS IN THE BLISTER BY 2 BLISTERS IN THE PACK OF PAPER -1 2363UP. SERIES 1 1 021 9. MANUFACTURING DATE 06. 02. 201 9. EXPIRY DATE 03. 2021 R. COMPOSITION PER ACTIVE SUBST
3004900000
1. MEDICINES FOR PEOPLE. - BIFREN CAPSULES NO. 20 TABLETS 250MG EACH. BY 1 0 TABLETS IN THE BLISTER BY 2 BLISTERS IN THE PACK OF PAPER -24566UP. SERIES 91 091 9. MANUFACTURING DATE 22. 09. 201 9. EXPIRY DATE 1 0. 2021 R. COMPOSITION PER ACTIVE SUBSTANCE TABLE: FENIBUT - 250MG. - RISOPTAN TABLES 1 0MG. ?3 3 TABLETS IN THE BLISTER 1 BLISTER IN THE PACK OF PAPERBOARD-51 1 8UP. SERIES 02091 9. PRODUCTION DATE 02. 09. 201 9. SHELF LIFE 1 0. 2022P. ACTIVE SUBSTANCES: 1 TABLE CONTAINING RISATRIPTANE BENZOATE 1 4 53MG. IN TRANSFER TO RIZATRIPTAN 1 0MG. - RISOPTAN TABLES 1 0MG. ? 6 3 TABLETS IN THE BLISTER 2 TABLETS IN THE PACK OF PAPER -1 877UP. SERIES 23091 9. PRODUCTION DATE 02. 09. 201 9. EXPIRY DATE 1 0. 2022P. ACTIVE SUBSTANCES: 1 TABLE CONTAINING RISATRIPTANE BENZOATE 1 4 53MG. IN TRANSFER TO RIZATRIPTAN 1 0MG. NOT IN THE AEROSOL PACK. WITHOUT CONTENT OF DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS. MANUFACTURER: PHARM START LTD. UA. TRADE MARK: PHARM START. PRODUCTION COUNTRY UA.
Global Export Import Trade Data for each capsule under HS-Code 300490
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 CHILE CHILE | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 BELGIUM BELGIUM | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 BELGIUM BELGIUM | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 BRAZIL BRAZIL | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
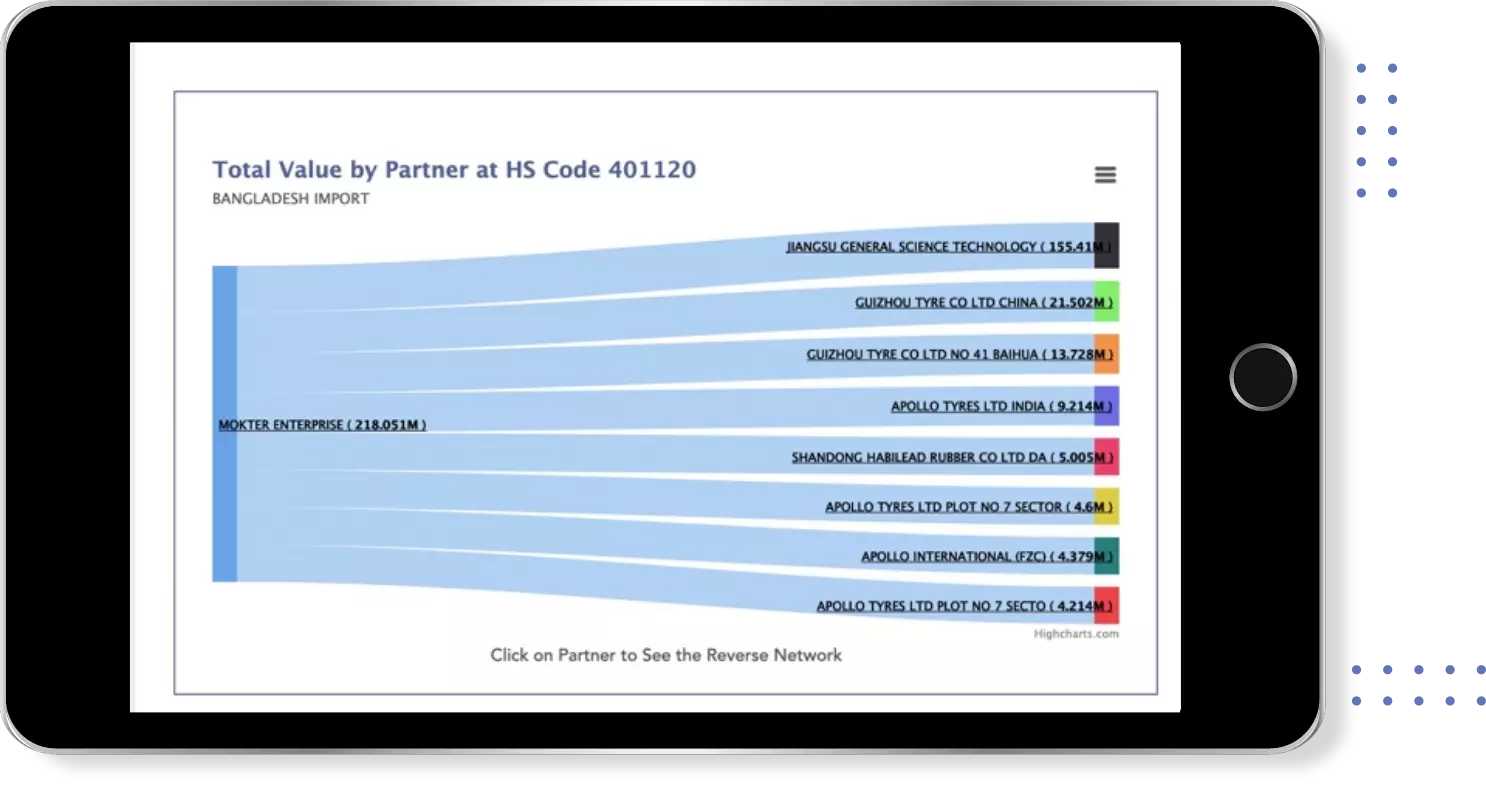
Get most profitable Buyers & Suppliers
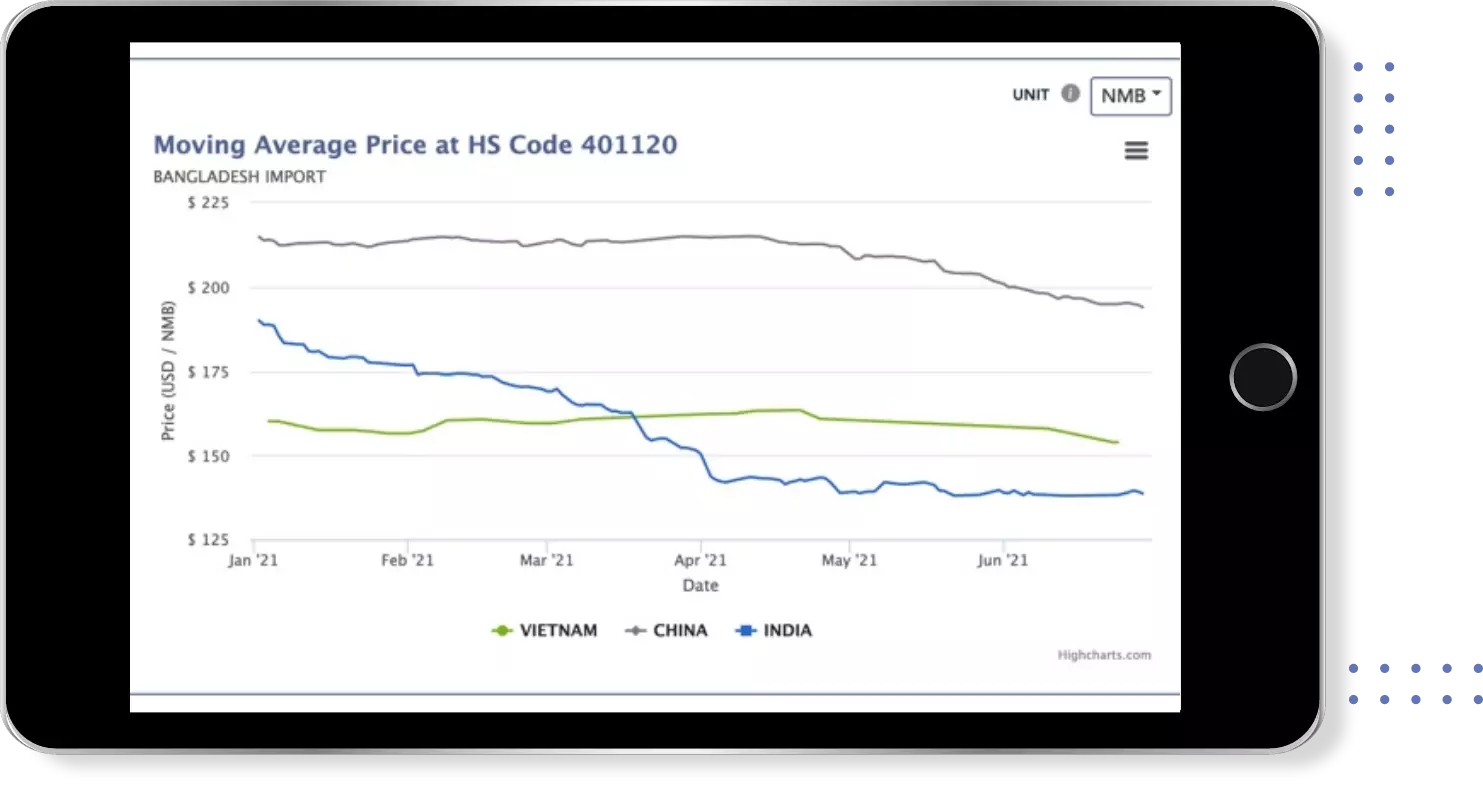
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries