Find verified buyers and sellers of each capsule in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- each capsule hs code 300490 buyers in ukraine
- HS Code: 300490
- COD Ukraine
Ukraine Buyers of each capsule under HS-Code 300490
SM AGENCY LLC UKRAINE

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES NOT REGISTERED (NOT IN AEROSOL PACKAGING) FOR PEOPLE INTENDED FOR CLINICAL TRIALS ACCORDING TO THE PROTOCOL LPS15021 NOT FOR RETAIL TRADE NOT OF ANIMAL ORIGIN.- SET CONTAINING 6 BLISTERS OF ROSUVASTATIN 20 MG (CRESTOR) / COMBINED DRUG IN A FIXED DOSE ROSUVASTATIN 10 MG AND EZETIMIBE 10 MG (ZENON) / PLACEBO FOR ORAL ADMINISTRATION EACH BLISTER CONTAINS 8 CAPSULES OF ROSUVASTATIN 20 MG (CRESTOR) OR THE CORRESPONDING PLACEBO 8 TABLETS OF THE COMBINED DRUG IN A FIXED DOSE OF ROSUVASTATIN 10 MG / EZETIMIBE 10 MG (ZENON) OR APPROPRIATE PLACEBO) - 16 SETS .; PACKING SERIES: IP0009658. SERIES: C1090616. EXPIRY DATE: 08.2021. MANUFACTURER: SANOFI-AVENTIS R&D. TRADEMARK: SANOFI (CRESTOR / ZENON). COUNTRY OF MANUFACTURE: FR.
3004900000
1. MEDICINES NOT REGISTERED (NOT IN AEROSOL PACKAGING) FOR PEOPLE INTENDED FOR CLINICAL TRIALS UNDER THE PROTOCOL LPS15021 NOT FOR RETAIL TRADE NOT OF ANIMAL ORIGIN.- PACKAGING CONTAINING (CRESTOR) ROSUVASTATIN 10 MG COATED TABLETS FOR ORAL USE 28 TABLETS IN A PACKAGE - 76 UNITS; PACKING SERIES: IP0009888. SERIES: C1092758. SHELF LIFE: 07.2021.- SET CONTAINING 6 BLISTERS OF ROSUVASTATIN 20 MG (CRESTOR) / COMBINATION DRUG IN A FIXED DOSE OF ROSUVASTATIN 10 MG AND EZETIMIBE 10 MG (ZENON) / PLACEBO FOR ORAL USE (EACH BLISTER CONTAINS 8 CAPSULES OF ROSUVASTATIN 20 MG ( CRESTOR) OR THE APPROPRIATE PLACEBO 8 TABLETS OF THE COMBINED DRUG IN A FIXED DOSE OF ROSUVASTATIN 10 MG / EZETIMIBE 10 MG (ZENON) OR THE APPROPRIATE PLACEBO) - 24 SETS .; PACKING SERIES: IP0009658. SERIES: C1090616. SHELF LIFE: 08.2021.- SET CONTAINING 6 BLISTERS OF ROSUVASTATIN 20 MG (CRESTOR) / COMBINATION DRUG IN A FIXED
3004900000
1. MEDICINES NOT REGISTERED (NOT IN AEROSOL PACKAGING) FOR PEOPLE INTENDED FOR CLINICAL TRIALS UNDER THE PROTOCOL LPS15021 NOT FOR RETAIL TRADE NOT OF ANIMAL ORIGIN.- PACKAGING CONTAINING (CRESTOR) ROSUVASTATIN 10 MG COATED TABLETS FOR ORAL USE 28 TABLETS IN A PACKAGE - 158 UNITS; PACKING SERIES: IP0009888. SERIES: C1092758. SHELF LIFE: 07.2021.- SET CONTAINING 6 BLISTERS OF ROSUVASTATIN 20 MG (CRESTOR) / COMBINATION DRUG IN A FIXED DOSE OF ROSUVASTATIN 10 MG AND EZETIMIBE 10 MG (ZENON) / PLACEBO FOR ORAL USE (EACH BLISTER CONTAINS 8 CAPSULES OF ROSUVASTATIN 20 MG ( CRESTOR) OR THE APPROPRIATE PLACEBO 8 TABLETS OF THE COMBINED DRUG IN A FIXED DOSE OF ROSUVASTATIN 10 MG / EZETIMIBE 10 MG (ZENON) OR THE APPROPRIATE PLACEBO) - 28 SETS .; PACKING SERIES: IP0009658. SERIES: C1090616. EXPIRY DATE: 08.2021. MANUFACTURER: SANOFI-AVENTIS R&D. TRADEMARK: SANOFI (CRESTOR / ZENON). COUNTRY OF MANUFACTURE: FR.
BADM LTD LIABILITY COMPANY

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE WITHOUT CONTENT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS PACKAGED FOR RETAIL SALE NOT IN AEROSOL PACKAGING: ABIZOL TABLETS OF 5 MG 14 TABLETS IN A BLISTER 2 BLISTERS IN A CARDBOARD PACKAGE / 1 TABLET CONTAINS ARIPIPRAZOLE 5 MG / MANUFACTURER NOBEL ILAC SANAI VE TICARET AS TURKEY ABIZOL 10 MG TABLETS 14 TABLETS IN A BLISTER 2 BLISTERS IN A CARTON / 1 TABLET CONTAINING ARIPIPRAZOLE 5 MG / MANUFACTURER NOBELI NABELIALA A.SH. TURKEY DUXET INTESTINAL SOLUBLE CAPSULES 60 MG 14 CAPSULES IN A BLISTER; 2 BLISTERS IN A CARDBOARD PACKAGE / 1 CAPSULE CONTAINS DULOXETINE HYDROCHLORIDE EQUIVALENT TO DULOXETINE 60 MG / MANUFACTURER NOBEL ILACH SANAI VE TIJARET A.SH. (FULL PRODUCTION CYCLE RESPONSIBLE FOR BATCH PRODUCTION) TURKEY / ULKAR KIMIYA SANAI VE TIJARET A.SH. (ALTERNATIVE PELLET MANUFACTURER) TURKEY DUXET ENTERIC CAPSULES 30 MG 14 CAPSULES IN A BLISTER; 2 BLISTERS IN A CARDBOARD PACKAGE / 1 CAPSULE CONTAINS DULOXETINE HYDROCHLORIDE EQUIVALENT TO DULOXETINE 30 MG / MANUFACTURER NOBEL ILACH SANAI VE TICARET A.SH. (FULL PRODUCTION CYCLE RESPONSIBLE FOR BATCH PRODUCTION) TURKEY / ULKAR KIMIYA SANAI VE TIJARET A.SH. (ALTERNATIVE PELLET MANUFACTURER) TURKEY ESSOBEL ® FILM-COATED TABLETS 10 MG 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARTON / 1 TABLET CONTAINING ESCITALOPRAM OXALATE EQUIVALENT TO ESCITALOPRAM 10 MG / MANUFACTURER NOBEL ILAC SANAI VE TIJARET AS TURKEY ESSOBEL ® FILM-COATED TABLETS 20 MG TABLETS 14 MG EACH; 2 BLISTERS IN A CARTON / 1 TABLET CONTAINING ESCITALOPRAM OXALATE EQUIVALENT TO ESCITALOPRAM 20 MG / MANUFACTURER NOBEL ILAC SANAYI VE TICARET AS TURKEY CANTAB TABLETS 16 MG 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARTON / 1 TABLET CONTAINING CANDESARTAN CILEXETIL 16 MG / MANUFACTURER NOBEL ILAC SANAYI VE TICARET AS TURKEY LANSOPROL ® 15 MG CAPSULES 14 CAPSULES IN A BLISTER; 2 BLISTERS IN A CARDBOARD PACKAGE / 1 CAPSULE CONTAINS LANSOPRAZOLE 15 MG / MANUFACTURER NOBEL ILACH SANAYI VE TIJARET A.SH. (FULL PRODUCTION CYCLE RESPONSIBLE FOR BATCH PRODUCTION) TURKEY ULKAR KIMIYA SANAI VE TIJARET A.SH. (MICROPELLET PRODUCTION) TURKEY LANSOPROL ® 30 MG CAPSULES 14 CAPSULES IN A BLISTER; 2 BLISTERS IN A CARDBOARD PACKAGE; / 1 CAPSULE CONTAINS LANSOPRAZOLE 30 MG / MANUFACTURER NOBEL ILACH SANAI VE TICARET A.SH. (FULL PRODUCTION CYCLE RESPONSIBLE FOR BATCH PRODUCTION) TURKEY / ULKAR KIMIYA SANAI VE TIJARET A.SH. (MICROPELLET PRODUCTION) TURKEY MELBEC ® 15 MG TABLETS 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARDBOARD PACKAGE / 1 TABLET CONTAINS MELOXICAM 15 MG / MANUFACTURER NOBEL ILAC SANAYA VE TICARET AS TURKEY MELBEC ® SOLUTION FOR INJECTION 15 MG / 1.5 ML 1.5 ML IN AMPOULES VOLUME OF 2 ML; 3 AMPOULES IN A CARDBOARD PACKAGE / 1 AMPOULE (1.5 ML) OF THE DRUG CONTAINS MELOXICAM 15 MG / MANUFACTURER IDOL ILACH DOLUM SANAI VE TICARET AS TURKEY MEXIA 10 FILM-COATED TABLETS 10 MG
3004900000
1. MEDICINES FOR PEOPLE WITHOUT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS FOR RETAIL NOT IN AEROSOL PACKAGING: ZOKARDIS ® 30 MG FILM-COATED TABLETS 30 MG 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX / 1 FILM-COATED TABLET CONTAINING 30 MG ZOFENOPRIL CALCIUM EQUIVALENT TO 28.7 MG ZOFENOPRIL / MANUFACTURER A. MENARINI MANUFACTURING LOGISTICS AND SERVICES S.R.L. (PRODUCTION IN BULK PRIMARY AND SECONDARY PACKAGING BATCH RELEASE) ITALY / DOMPE PHARMACEUTICS S.P.A. (BATCH CONTROL) ITALY / MENARINI-VON HAYDEN GMBH (PRODUCTION IN BULK PRIMARY AND SECONDARY PACKAGING BATCH CONTROL AND RELEASE) GERMANY ZOCARDIS ® PLUS 30 / 12.5 FILM-COATED TABLETS 14 TABLETS EACH IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX / 1 FILM-COATED TABLET CONTAINING ZOFENOPRIL CALCIUM 30 MG EQUIVALENT TO ZOFENOPRIL 28.7 MG AND HYDROCHLOROTHIAZIDE 12.5 MG / MANUFACTURER A. MENARINI MANUFACTURING LOGISTICS AND SERVICES S.R.L. (BULK PRODUCTION FINAL PACKAGING BATCH RELEASE; BATCH CONTROL) ITALY / DOMPE PHARMACEUTICALS S.P.A. (BATCH CONTROL) ITALY / MENARINI-VON HAYDEN GMBH (PRODUCTION IN BULK FINAL PACKAGING BATCH CONTROL AND RELEASE) GERMANY LERKAMEN® ACE 10/20 FILM-COATED TABLETS 14 TABLETS IN A BLISTER; 2 BLISTERS PER CARTON / 1 FILM-COATED TABLET CONTAINING LERCANIDIPINE HYDROCHLORIDE 10 MG EQUIVALENT TO LERCANIDIPINE 9.44 MG AND ENALAPRIL MALEATE 20 MG EQUIVALENT TO ENALAPRIL 15.29 MG / MANUFACTURED BY RECORDATI INDUSTRY S. PA ITALY LERCAMEN® ACE 10/10 FILM-COATED TABLETS 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARTON / 1 FILM-COATED TABLET CONTAINING LERCANIDIPINE HYDROCHLORIDE 10 MG EQUIVALENT TO LERCANIDIPINE 9.44 MG AND ENALAPRIL MALEATE 10 MG EQUIVALENT TO ENALAPRIL 7.64 MG / MANUFACTURED BY RECORDATI INDUSTREA C. PA ITALY MEZIM® CAPSULES 10000 HARD CAPSULES WITH ENTERIC-COATED MINI-TABLETS 20 CAPSULES IN A POLYPROPYLENE JAR 1 JAR IN A CARDBOARD BOX / 1 HARD CAPSULE CONTAINS POWDER FROM PANCREATIC GLANDS (PIGS) 153.5 ( 98.3 - 178.6) MG WITH A MINIMUM LIPOLYTIC ACTIVITY OF 10 000 IU EF (EUROPEAN PHARMACOPOEIA UNIT) A MINIMUM AMYLOLYTIC ACTIVITY OF 9 000 IU IU A MINIMUM PROTEOLYTIC ACTIVITY OF 500 IU EF / MANUFACTURER ADER PHARMACEUTICALS SRL (IN BULK PRODUCTION CONTROL SERIES) ITALY / ADVANCE PHARMA GMBH (FINAL PACKAGING) GERMANY / ARTESAN PHARMA GMBH & CO. KG (FINAL PACKAGING) GERMANY / BERLIN-CHEMI AG (CONTROL AND PRODUCTION SERIES) GERMANY NIXAR ® TABLETS OF 20 MG 10 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX / 1 TABLET CONTAINS BILASTINE 20 MG / MANUFACTURER A. MENARINI MANUFACTURING LOGISTICS AND SERVICES S.R.L. (PRODUCTION IN BULK PACKAGING BATCH PRODUCTION BATCH CONTROL) ITALY ALTAR ® 2 MG 2 MG TABLETS 30 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX / 1 TABLET
3004900000
1. MEDICINES FOR PEOPLE WITHOUT CONTENT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: ACTIPROL TABLETS OF 100 MG 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS AMISULPRIDE 100 MG / MANUFACTURER MEDOKEMI LTD (CENTRAL PLANT) (FULL CYCLE PRODUCTION) CYPRUS / MEDOKEMI LTD (PLANT AZ) (PRIMARY AND SECONDARY PACKAGING) CYPRUS ACTIPROL 200 MG TABLETS 10 TABLETS IN A BLISTER 3 BLISTERS IN CARTON / 1 TABLET CONTAINING AMISULPRIDE 200 MG / MANUFACTURER MEDOKEMI LTD (CENTRAL PLANT) (FULL CYCLE PRODUCTION) CYPRUS / MEDOKEMI LTD (PLANT AZ) (PRIMARY AND SECONDARY PACKAGING) CYPRUS ALMIRAL SOLUTION FOR INJECTION 75MG / 3ML 3ML IN AN AMPOULE 5 AMPOULES IN A BLISTER 1 BLISTER IN A CARDBOARD BOX / 3 ML SOLUTION (1 AMPOULE) CONTAINS: DICLOFENAC SODIUM 75 MG / MANUFACTURER MEDOKEMI LTD (AMPOULE INJECTION PLANT) CYPRUS BETA TABLETS FILM-COATED 10MG 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARDBOARD BOX THESE / 1 TABLET CONTAINS BETAXOLOL HYDROCHLORIDE 10 MG / PRODUCER MEDOKEMI LTD (CENTRAL PLANT) CYPRUS BETAK TABLETS COVERED WITH FILM MEMBRANE FOR 20 MG 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARTON BOX / 1 TABLET CONTAINS BETAXOLOL HYDROCHLORIDE 20 MG / MANUFACTURER MEDOKEMI LTD (CENTRAL PLANT) CYPRUS ZIKALOR TABLETS 10 MG 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARTON BOX / 1 TABLET CONTAINS ARIPIPRAZOLE 10 MG / PRODUCER MEDOKEMI LTD (FACTORY AZ) (PRIMARY AND SECONDARY PACKAGING CONTROL AND BATCH RELEASE) CYPRUS KONVERIUM TABLETS 150 MG 10 TABLETS IN A BLISTER 3 BLISTERS IN PACK / 1 TABLET CONTAINS: IRBESARTAN 150 MG / PRODUCER MEDOKEMI LTD (CENTRAL PLANT) CYPRUS LIHATO CAPSULE HARD 75 MG 7 CAPSULES IN BLISTER 3 BLISTERS IN A CARDBOARD BOX / 1 CAPSULE CONTAINS 75 MG OF PREGABALIN / MANUFACTURER MEDOKEMI LTD (PLANT AZ) (PRIMARY AND SECONDARY PACKAGING) CYPRUS / MEDOKEMI LTD (CENTRAL PLANT) (MANUFACTURE OF FINISHED PRODUCT QUALITY CONTROL BATCH RELEASE) CYPRUS MAGUROL TABLETS OF 4 MG OF 10 TABLETS IN A BLISTER; 2 BLISTERS IN A CARTON BOX / 1 TABLET CONTAINS DOXAZOSIN MESYLATE EQUIVALENT TO 4.86 MG OF DOXAZOSIN 4 MG / PRODUCER MEDOKEMI LTD (CENTRAL PLANT) CYPRUS MEDOHISTYN TABLETS 24MH 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARTON BOX / 1 EACH TABLET CONTAINS 24 MG BETAHISTINE DIHYDROCHLORIDE / PRODUCER MEDOKEMI LTD (CENTRAL PLANT) CYPRUS / FARMATSEVTISH ANALITISH LABORATORIUM BV DUIVEN THE NETHERLANDS; MEDOHISTYN TABLETS 16 MG; 10 TABLETS IN ALUMINUM BLISTER 3 BLISTERS IN A BOX / 1 TABLET CONTAINS BETAHISTINE DIHYDROCHLORIDE 16 MG / PRODUCER MEDOKEMI LTD (CENTRAL PLANT) CYPRUS / FARMATSEVTISH ANALITISH LABORATORIUM BV DUIVEN THE NETHERLANDS; MEDOHREL TABLETS FILM COATED SHELL ON 75 MG 10 TABLETS IN A BLISTER 3 BLISTERS IN A BOX / 1 CLOPIDOGREL BISULFATE TABLET CONTAINS 97.86 MG EQUIVALENT TO CLOPIDOGREL 75 MG / PRODUCER MEDOKEMI LTD (CENTRAL PLANT) (PRODUCTION NEROZFASOVANOHO ETC.
JV OPTIMA PHARM LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE. SERIES 120022: ARLEVERT® TABLETS OF 20 TABLETS IN A BLISTER; ON 1 BLISTER IN A CARDBOARD BOX - 3060 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - DIMENHYDRINATE CINNARIZINE) SERIES 114062: ARLEVERT® TABLETS OF 25 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 540 UNITS. (ACTIVE SUBSTANCE - DIMENHYDRINATE CINNARIZINE) SERIES 120022: ARLEVERT® TABLETS OF 25 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 1800 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - DIMENHYDRINATE CINNARIZINE) MANUFACTURER: HENNIG ARNZEIMITTEL GMBH & CO. KG DE SERIES 11060D: ATTENTO® 20/5 FILM-COATED TABLETS 20 MG / 5 MG; 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 384 UNITS. (ACTIVE SUBSTANCE - AMLODIPINE BESYLATE OLMESARTAN MEDOXOMIL) SERIES 11053B: ATTENTO® 40/10 FILM-COATED TABLETS 40 MG / 10 MG; 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 192 UNITS. (ACTIVE SUBSTANCE - AMLODIPINE BESYLATE OLMESARTAN MEDOXOMIL) MANUFACTURER: PRODUCTION IN BULK SERIES CONTROL: DAICHI SANKIO EUROPE GMBH DE PACKAGING AND PRODUCTION SERIES: BERLIN-CHEMI AG DE. SERIES 10 TABLETS 12522A: MG 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARDBOARD BOX - 9600 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - ENALAPRIL MALEATE) SERIES 11003A: BERLIPRIL® 20 TABLETS OF 20 MG 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARDBOARD BOX - 800 UNITS. (ACTIVE SUBSTANCE - ENALAPRIL MALEATE) MANUFACTURER: PRODUCTION IN BULK PACKAGING CONTROL AND RELEASE OF SERIES: BERLIN-CHEMI AG DE. SERIES 12006A: BERLIPRIL® PLUS 10/25 TABLETS OF 10 MG / 25 MG OF 10 TABLETS IN BLISTERS; 3 BLISTERS IN A CARDBOARD BOX - 16 900 UNITS. (ACTIVE SUBSTANCE - HYDROCHLOROTHIAZIDE ENALAPRIL MALEATE) MANUFACTURER: PRODUCTION IN BULK BATCH CONTROL FINAL PACKAGING CONTROL AND RELEASE OF THE SERIES: BERLIN-CHEMI AG DE. SERIES 11007: BERLITION® 600 IU CONCENTRATE FOR SOLUTION FOR INFUSION OD (600 MG) / 24 ML 24 ML IN AN AMPOULE; ON 5 AMPOULES IN A CARDBOARD BOX - 900 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - THIOCTIC ACID) MANUFACTURER: SERIES RELEASE: BERLIN-CHEMI AG DE. SERIES 11520A: BROMHEXIN 8 BERLIN-CHEMI COATED TABLETS 8 MG 25 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX - 17400 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - BROMHEXINE HYDROCHLORIDE) MANUFACTURER: PRODUCTION OF BULK PRODUCT BATCH CONTROL; PACKAGING CONTROL AND RELEASE OF THE SERIES: BERLIN-CHEMI AG DE SERIES 08092: GLIBOMET® FILM-COATED TABLETS OF 20 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 1152 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - GLIBENCLAMIDE METFORMIN HYDROCHLORIDE) MANUFACTURER: PRODUCTION IN BULK PACKAGING CONTROL AND RELEASE OF SERIES: MENARINI-VON HAYDEN GMBH DE SERIES 18001: DICLOBERL® RETARD HARD CAPSULES PROLONGED-RELEASE 10 MG PER 100 MG ; 2 BLISTERS IN A CARDBOARD BOX - 5832 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - DICLOFENAC SODIUM) MANUFACTURER: PRODUCTION OF HARD CAPSULES IN BULK SERIES CONTROL: HENNIG ARNZEIMITTEL GMBH & CO. KG DE FINAL PACKAGING SERIES RELEASE: BERLIN-CHEMI AG DE. SERIES 11012A: DICLOBER 10 EACH
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE. SERIES H91092A: PREDIZIN® PROLONGED-RELEASE TABLETS FILM-COATED 35 MG 10 TABLETS IN A BLISTER; 6 BLISTERS IN A CARDBOARD BOX - 400 UNITS (ACTIVE SUBSTANCE - TRIMETAZIDINE DIHYDROCHLORIDE) MANUFACTURER: QUALITY CONTROL PRODUCTION PERMIT PRODUCTION OF BULK PRODUCT PRIMARY PACKAGING SECONDARY PACKAGING: LLC GIDEON RICHTER POLAND PL SERIES RELEASE: OJSC GIDEON RICHTER HU. SERIES H84079B: STOPI HARD CAPSULES OF 200 MG 12 CAPSULES IN A BLISTER 1 BLISTER IN A CARTON -384 PACK (ACTIVE SUBSTANCE - NIFUROXAZIDE) SERIES H8C016A: NIFUROXAZIDE RICHTER COATED TABLETS 100 MG 24 TABLETS IN EACH; 1 BLISTER IN A CARDBOARD BOX -3360 PACK (ACTIVE INGREDIENT - NIFUROXAZIDE) MANUFACTURER: QUALITY CONTROL BATCH APPROVAL PRODUCTION OF BULK PRODUCT PRIMARY PACKAGING SECONDARY PACKAGING: LLC GIDEON RICHTER POLAND PL.
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE. SERIES 1045731: ALLESTA® FILM-COATED TABLETS 20 MG 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARDBOARD BOX - 19000 UNITS (ACTIVE SUBSTANCE - SIMVASTATIN) SERIES : LOTAR® FILM-COATED TABLETS OF 100 MG 15 TABLETS IN A BLISTER 2 BLISTERS IN A CARDBOARD BOX - 219 PACK (ACTIVE SUBSTANCE - LOSARTAN POTASSIUM) SERIES 1032904: LOTAR® FILM-COATED TABLETS 50 MG EACH 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARDBOARD BOX - 161 UNITS (ACTIVE INGREDIENT - LOSARTAN POTASSIUM) SERIES 1045418: NIFUROXASIDE ALKALOID HARD CAPSULES OF 100 MG 10 CAPSULES IN A BLISTER 3 BLISTERS IN A CARDBOARD PACK OF 30 PACKS. (ACTIVE SUBSTANCE - NIFUROXAZIDE) SERIES 1044037: NIFUROXAZIDE ALKALOID ORAL SUSPENSION 200 MG / 5 ML 90 ML IN A VIAL 1 VIAL COMPLETE WITH A MEASURING CUP IN A CARDBOARD BOX - 618 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE 10 - 25) NIFUROXAZIDE ALKALOID HARD CAPSULES OF 200 MG
KUZAR IRINA OLEKSANDRIVNA PASSPORT MM 0381 1 9 ISSUED BY KOMINTERNIVSKY RV KHMU UMVD IN KHARKIV REGION Â OCTOBER 23 1998

UKRAINE
HS-Code
Products
3004900000
1. UNREGISTERED MEDICINAL PRODUCTS FOR HUMAN USE THERAPEUTIC AND PROPHYLACTIC FOR INDIVIDUAL USE NOT CONTAINING NARCOTIC DRUGS AND PRECURSORS NOT IN AEROSOL PACKAGING PACKAGED FOR RETAIL SALE: -DRB436 (DABRAFENIB) 75 MG 28 X HNGC CAPSULE LABBLEDBOTTLE - 13 PACKS CAPSULES IN EACH. TOTAL: 364 CAPSULES. SERIAL NUMBER OF THE MANUFACTURER: 1010023597. PACKING SERIES: 2036322. SHELF LIFE UNTIL JULY 31 2022. ACTIVE SUBSTANCE DABRAFENIB. COMES PACKAGED IN A THERMAL BOX WITH REFRIGERANTS AND A DISPOSABLE SENSOR FOR MONITORING THE TEMPERATURE OF THE INTERIOR OF THE PACKAGING. THE GOODS ARE IMPORTED FREE OF CHARGE WITHOUT THE RIGHT TO SELL SELL AND ONLY FOR CLINICAL TRIALS.
3004900000
1.UNREGISTERED MEDICINAL PRODUCTS FOR HUMAN USE THERAPEUTIC AND PROPHYLACTIC FOR INDIVIDUAL USE NOT CONTAINING NARCOTIC DRUGS AND PRECURSORS NOT IN AEROSOL PACKAGING PACKAGED FOR RETAIL SALE: -TAFINLAR (DABRAFENIB) CAPSULE 75 MG 1 X 120 NVS U57 (NNC) - 1 PACK OF 120 CAPSULES EACH. TOTAL: 120 CAPSULES. SERIAL NUMBER OF THE MANUFACTURER: 9V8V. SHELF LIFE UNTIL JULY 31 2022. ACTIVE SUBSTANCE DABRAFENIB. IT IS DELIVERED IN PACKING FROM A THERMAL BOX WITH REFRIGERANTS AND THE INCLUDED DISPOSABLE SENSOR OF CONTROL OF TEMPERATURE OF INTERNAL SPACE OF A PACKING SET. THE GOODS ARE IMPORTED FREE OF CHARGE WITHOUT THE RIGHT TO SELL SELL AND ONLY FOR CLINICAL TRIALS.
3004900000
1.UNREGISTERED MEDICINAL PRODUCTS FOR HUMAN USE THERAPEUTIC AND PROPHYLACTIC FOR INDIVIDUAL USE NOT CONTAINING NARCOTIC DRUGS AND PRECURSORS NOT IN AEROSOL PACKAGING PACKAGED FOR RETAIL SALE: -TAFINLAR (DABRAFENIB) CAPSULE 75 MG 1 X 120 NVS U57 (NNC) - 2 PACKS OF 120 CAPSULES EACH. TOTAL: 240 CAPSULES. SERIAL NUMBER OF THE MANUFACTURER: 9V8V. SHELF LIFE UNTIL JULY 31 2022. ACTIVE SUBSTANCE DABRAFENIB. IT IS DELIVERED IN PACKING FROM A THERMAL BOX WITH REFRIGERANTS AND THE INCLUDED DISPOSABLE SENSOR OF CONTROL OF TEMPERATURE OF INTERNAL SPACE OF A PACKING SET. THE GOODS ARE IMPORTED FREE OF CHARGE WITHOUT THE RIGHT TO SELL SELL AND ONLY FOR CLINICAL TRIALS.
WORLDWIDE CLINICAL TRAILS UKR LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINAL PRODUCTS NOT REGISTERED IN UKRAINE FOR PEOPLE IMPORTED EXCLUSIVELY FOR USE IN CLINICAL TRIALS PROTOCOL MIN-117C03: - A SET CONTAINING 6 BLISTERS EACH BLISTER CONTAINING 16 CAPSULES MIN-117 2.5 MGA OR PLACEBO. QUANTITY - 12 SETS. SERIAL NUMBER E1530. SHELF LIFE -11/01/20 THE PREPARATIONS DO NOT CONTAIN ALCOHOL VITAMINS INFECTED AND / OR BIOLOGICALLY DANGEROUS COMPONENTS NARCOTIC AND / OR PSYCHOTROPIC SUBSTANCES PRECURSORS OF OTHER SUBSTANCES THE CIRCULATION OF WHICH IS UNDER THE SPECIAL ROLE OF THE MINISTRY OF HEALTH. NOT FOR SALE RESALE SALES. MANUFACTURER - AMATSI AQUITAINE -ARTIGUES PR ES BORDEAUXBRAND - AMATSI AQUITAINE -AR TIGUES PRES BORDEAUXCOUNTRY OF PRODUCTION - FR
3004900000
1. ABX464-103 PROTOCOL UNREGISTERED IN UKRAINE FOR HUMAN USE IMPORTED EXCLUSIVELY FOR USE IN CLINICAL TRIALS: - SET CONTAINING 5 BLISTERS EACH BLISTER CONTAINING 14 CAPSULES EACH CONTAINING 50 MG OF ABX464 OR 25 MG OF ABX464 OR PLACEBO. NUMBER -400 SETS. CLI9800 BATCH. SHELF LIFE - 06/2020 THE DRUGS DO NOT CONTAIN ALCOHOL VITAMINS INFECTED AND / OR BIOLOGICALLY DANGEROUS COMPONENTS NARCOTIC AND / OR PSYCHOTROPIC DRUGS PRECURSORS OR OTHER SUBSTANCES WHICH ARE UNDER THE SPECIAL ROLE OF THE MINISTRY OF HEALTH. NOT FOR SALE RESALE SALE. MANUFACTURER - CREAPHARM CLINICAL SUPPLIES TRADEMARK - CREAPHARM COUNTRY OF MANUFACTURE - FR
3004900000
1. MEDICINAL PRODUCTS NOT REGISTERED IN UKRAINE FOR PEOPLE IMPORTED EXCLUSIVELY FOR USE IN CLINICAL TRIALS PROTOCOL MIN-117C03: - A SET CONTAINING 6 BLISTERS EACH BLISTER CONTAINING 16 CAPSULES MIN-117 2.5 MGA OR PLACEBO. NUMBER - 11 SETS. SERIAL NUMBER E1530. SHELF LIFE -11/01/20 THE PREPARATIONS DO NOT CONTAIN ALCOHOL VITAMINS INFECTED AND / OR BIOLOGICALLY DANGEROUS COMPONENTS NARCOTIC AND / OR PSYCHOTROPIC SUBSTANCES PRECURSORS OF OTHER SUBSTANCES THE CIRCULATION OF WHICH IS UNDER THE SPECIAL ROLE OF THE MINISTRY OF HEALTH. NOT FOR SALE RESALE SALES. MANUFACTURER - AMATSI AQUITAINE -ARTIGUES PR ES BORDEAUXBRAND - AMATSI AQUITAINE -AR TIGUES PRES BORDEAUXCOUNTRY OF PRODUCTION - FR
GEVAK GALINA IVANIVNA PASSPORT SO 361 1 01 VIDANIYM MINSKYM RU GU MVS UKRAINI IN MISTI KYIVI 25 05 2000 ROCU

UKRAINE
HS-Code
Products
3004900000
1.UNREGISTERED MEDICINAL PRODUCTS FOR HUMANS THERAPEUTIC AND PROPHYLACTIC FOR INDIVIDUAL USE NOT CONTAINING NARCOTIC DRUGS AND PRECURSORS NOT IN AEROSOL PACKAGING PACKAGED FOR RETAIL SALE: -TAFINLAR (DABRAFENIB) CAPSULE 75 MG 1 X 120 NVS U57-1 PACKING BY 120 CAPSULES IN EACH. TOTAL: 120 CAPSULES. SERIAL NUMBER OF THE MANUFACTURER: 9V8V. SHELF LIFE UNTIL JULY 31 2022. ACTIVE SUBSTANCE DABRAFENIB. IT IS DELIVERED IN PACKING FROM A THERMAL BOX WITH REFRIGERANTS AND THE INCLUDED DISPOSABLE SENSOR OF CONTROL OF TEMPERATURE OF INTERNAL SPACE OF A PACKING SET. THE GOODS ARE IMPORTED FREE OF CHARGE WITHOUT THE RIGHT TO SELL SELL AND ONLY FOR CLINICAL TRIALS.
3004900000
1. UNREGISTERED MEDICINAL PRODUCTS FOR HUMAN USE THERAPEUTIC AND PROPHYLACTIC FOR INDIVIDUAL USE NOT CONTAINING NARCOTIC DRUGS AND PRECURSORS NOT IN AEROSOL PACKAGING PACKAGED FOR RETAIL SALE: -TAFINLAR (DABRAFENIB) CAPSULE 75 MG 1 X 120 NVS U57-2 PACKING ON 120 CAPSULES IN EACH. TOTAL: 240 CAPSULES. SERIAL NUMBER OF THE MANUFACTURER: 9V8V. SHELF LIFE UNTIL JULY 31 2022. ACTIVE SUBSTANCE DABRAFENIB. IT IS DELIVERED IN PACKING FROM A THERMAL BOX WITH REFRIGERANTS AND THE INCLUDED DISPOSABLE SENSOR OF CONTROL OF TEMPERATURE OF INTERNAL SPACE OF A PACKING SET. THE GOODS ARE IMPORTED FREE OF CHARGE WITHOUT THE RIGHT TO SELL SELL AND ONLY FOR CLINICAL TRIALS.
3004900000
1.UNREGISTERED MEDICINAL PRODUCTS FOR HUMANS THERAPEUTIC AND PROPHYLACTIC FOR INDIVIDUAL USE NOT CONTAINING NARCOTIC DRUGS AND PRECURSORS NOT IN AEROSOL PACKAGING PACKAGED FOR RETAIL SALE: -DRB436 (DABRAFENIB) 75 MG 28 X HNGC CAPSULE LABBLEDBOTTLE -13 PACKS CAPSULES IN EACH. TOTAL: 364 CAPSULES. SERIAL NUMBER OF THE MANUFACTURER: 1010023597. PACKING SERIES: 2036322. SHELF LIFE UNTIL JULY 31 2022. ACTIVE SUBSTANCE DABRAFENIB. COMES PACKAGED IN A THERMAL BOX WITH REFRIGERANTS AND A DISPOSABLE SENSOR FOR MONITORING THE TEMPERATURE OF THE INTERIOR OF THE PACKAGING. THE GOODS ARE IMPORTED FREE OF CHARGE WITHOUT THE RIGHT TO SELL SELL AND ONLY FOR CLINICAL TRIALS.
LLC RECORDS UKRAINE

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE PACKED FOR RETAIL TRADE: - LOMEXIN (R) SOFT VAGINAL CAPSULES ON 1000 MG ON 2 CAPSULES IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 CAPSULE CONTAINS: FENTICONAZOLE NITRATE 1000 MG) SERIES: 40129 -5880UP MANUFACTURER: CATALENT ITALY SPA ITALY COUNTRY OF MANUFACTURE: IT TRADEMARK: RECORDATI LOMEXIDE FILM-COATED 10 MG 14 TABLETS IN A BLISTER; 7 BLISTERS IN A CARDBOARD BOX (ACTIVE INGREDIENTS: 1 TABLET CONTAINS: 10 MG OF LERCANIDIPINE HYDROCHLORIDE (EQUIVALENT TO 9.4 MG OF LERCANIDIPINE) SERIES: ZB9M75 -2660UP-ZANIDIP (R) FILM-COATED TABLETS 7 MG PACKS OF 14 MG EACH; IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 TABLET CONTAINS: 20 MG OF LERCANIDIPINE HYDROCHLORIDE (EQUIVALENT TO 18.8 MG OF LERCANIDIPINE) SERIES: ZE9M14 -3000UP MANUFACTURER: RECORDS INDUSTRIA CHEMIKA E PHARMACEUTICS SPORA: ITACI: ITAC ITALY CORIPREN 20MG / 10MG TABLETS
3004900000
1. MEDICINES FOR PEOPLE PACKED FOR RETAIL TRADE: - LOMEXIN (R) SOFT VAGINAL CAPSULES ON 1000 MG ON 1 CAPSULE IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 CAPSULE CONTAINS: FENTICONAZOLE NITRATE 1000 MG) SERIES: 36079 -7810UP-LOMEXIN (R) SOFT VAGINAL CAPSULES 600 MG 1 CAPSULE IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 CAPSULE CONTAINS: FENTICONAZOLE NITRATE 600 MG) SERIES: 94109-4800UP MANUFACTURER: CATALENT ITALY SPA ITALY COUNTRY OF MANUFACTURE: IT TRADEMARK: RECORDATI LOMEXIDINE-TABLETS FILM-COATED 10 MG 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 TABLET CONTAINS: 10 MG OF LERCANIDIPINE HYDROCHLORIDE (EQUIVALENT TO 9.4 MG OF LERCANIDIPINE) SERIES: ZB9F64 -11500UP-ZANIDIP (R) TABLETS FILM-COATED TABLETS 14 TABLETS OF 7 MG EACH; IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 TABLET CONTAINS: 10 MG LERCANIDIPINE HYDROCHLORIDE (EQUIVALENT TO 9.4 MG LER
3004900000
1. MEDICINES FOR PEOPLE PACKED FOR RETAIL TRADE: - LOMEXIN (R) SOFT VAGINAL CAPSULES ON 1000 MG ON 1 CAPSULE IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 CAPSULE CONTAINS: FENTICONAZOLE NITRATE 1000 MG) SERIES: 40129 -7860UP MANUFACTURER: CATALENT ITALY SPA ITALY COUNTRY OF MANUFACTURE: IT TRADEMARK: RECORDATI LOMEXIDE FILM-COATED 20 MG 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 TABLET CONTAINS: 20 MG OF LERCANIDIPINE HYDROCHLORIDE (EQUIVALENT TO 18.8 MG OF LERCANIDIPINE) SERIES: ZE0E25 -12002UP-ZANIDIP (R) FILM-COATED TABLETS 14 BLISTERS OF 14 20 MG EACH; IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 TABLET CONTAINS: 20 MG OF LERCANIDIPINE HYDROCHLORIDE (EQUIVALENT TO 18.8 MG OF LERCANIDIPINE) SERIES: ZE0E25 -7070UP MANUFACTURER: RECORDS INDUSTRIA CHEMIKA E PHARASEVTIKA SPARIATI ITALY ITALY) CORIPREN 10MG / 10MG TABLETS
LLC SMO GROUP UKRAINE

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES ARE NOT REGISTERED (NOT IN AEROSOL PACKAGING) FOR PEOPLE INTENDED FOR CLINICAL TRIALS UNDER THE BUX-4 / UCA PROTOCOL NOT FOR RETAIL TRADE NOT OF ANIMAL ORIGIN. - A SET OF THE PATIENT CONTAINING: - 4 BOXES FOR VISITS IN EACH CONTAINING: 2 BLISTERS-WALLETS WITH 10 HARD CAPSULES OF BUDESONIDE 9 MG OR 6 MG OR PLACEBO AND 10 FILM-COATED TABLETS OF BUDESONIDE-MMX 9 MG OR PLACEBO AND - 1 BOX FOR VISITS WHICH CONTAINS: 2 BLISTERS-WALLETS WITH 5 HARD CAPSULES OF BUDESONIDE 9 MG OR 6 MG OR PLACEBO AND 5 FILM-COATED TABLETS OF BUDESONIDE-MMX 9 MG OR PLACEBO - 228 SETS PACKAGING SERIES: BUX004-VK01 SHELF LIFE: 07.2020.MANUFACTURER: DR. FALK PHARMA GMBH. TRADEMARK: DR. FALK PHARMA. COUNTRY OF MANUFACTURE DE.
3004900000
1. MEDICINES NOT REGISTERED (NOT IN AEROSOL PACKAGING) FOR HUMANS INTENDED FOR CLINICAL TRIALS UNDER THE PROTOCOL RGH-188-203 NOT FOR RETAIL NON-ANIMAL ORIGIN.- SET WEEK 0 CONTAINING CARIPRAZINE HARD CAPSULES OF MIXED DOSAGE 0 5 AND 1.5 MG IN A BLISTER ?10 - 30 SETS. PACKING SERIES: 3681959-01. SHELF LIFE: 04.2022.- SET WEEK 1-3 CONTAINING CARIPRAZINE 1.5 MG HARD CAPSULES IN A BLISTER ?10 - 38 SETS. PACKING SERIES: 3681959-02. SHELF LIFE: 04.2024.- SET WEEK 1-3 CONTAINING CARIPRAZINE HARD CAPSULES 3 MG IN A BLISTER ?10 - 38 SETS. PACKING SERIES: 3681959-03. SHELF LIFE: 04.2024.- SET WEEK 1 CONTAINING CARIPRAZINE HARD CAPSULES OF MIXED DOSAGE 3 AND 4.5 MG IN A BLISTER ?10 - 26 SETS. PACKING SERIES: 3681959-04. SHELF LIFE: 04.2024.- SET WEEK 2-3 CONTAINING CARIPRAZINE HARD CAPSULES 4.5 MG IN A BLISTER ?10 - 26 SETS. PACKING SERIES: 3681959-05. SHELF LIFE: 04.2024.- SET WEEK 2-3 CONTAINING CARIPRAZINE HARD CAPSULES 6 MG IN A BLISTER ?10 - 26 SETS. PACKING SERIES: 3681959-06. SHELF LIFE: 04.2024.- MONTHLY SET CONTAINING CARIPRAZINE 1.5 MG IN A BLISTER ?4 8 CAPSULES IN EACH - 98 SETS. PACKING SERIES: 3681959-07. SHELF LIFE: 04.2024.- MONTHLY SET CONTAINING CARIPRAZINE 3 MG IN A BLISTER ?4 8 CAPSULES IN EACH - 98 SETS. PACKING SERIES: 3681959-08. SHELF LIFE: 04.2024.- MONTHLY SET CONTAINING CARIPRAZINE 4.5 MG IN A BLISTER ?4 8 CAPSULES IN EACH - 98 SETS. PACKING SERIES: 3681959-09. SHELF LIFE: 04.2024.- MONTHLY SET CONTAINING CARIPRAZINE 6 MG IN A BLISTER ?4 8 CAPSULES IN EACH - 98 SETS. PACKING SERIES: 3681959-10. SHELF LIFE: 04.2024.MANUFACTURER: GEDEON RICHTER PLC. BRAND: GEDEON RICHTER. COUNTRY OF MANUFACTURE: HU.
3004900000
1. MEDICINES ARE NOT REGISTERED (NOT IN AEROSOL PACKAGING) FOR PEOPLE INTENDED FOR CLINICAL TRIALS UNDER THE BUX-4 / UCA PROTOCOL NOT FOR RETAIL TRADE NOT OF ANIMAL ORIGIN. - A SET OF THE PATIENT CONTAINING: - 4 BOXES FOR VISITS IN EACH CONTAINING: 2 BLISTERS-WALLETS WITH 10 HARD CAPSULES OF BUDESONIDE 9 MG OR 6 MG OR PLACEBO AND 10 FILM-COATED TABLETS OF BUDESONIDE-MMX 9 MG OR PLACEBO AND - 1 BOX FOR VISITS WHICH CONTAINS: 2 BLISTERS-WALLETS WITH 5 HARD CAPSULES OF BUDESONIDE 9 MG OR 6 MG OR PLACEBO AND 5 FILM-COATED TABLETS OF BUDESONIDE-MMX 9 MG OR PLACEBO - 228 SETS PACKAGING SERIES: 011013-3431204R (BUX004-VK03) / 011014-3431204R (BUX004) / 011015-3431204R (BUX004-VK03) / 011016-3431204R (BUX004-VK03) / 011017-3431204R (BUX004-VK03) / 011018-3431204R (BUX004-VK03) EXPIRATION DATE: 31.10.2022.MANUFACTURER GMBH. TRADEMARK: DR. FALK PHARMA. COUNTRY OF MANUFACTURE DE.
ROSHKA VIKTOR AURELOVYCH PASSPORT FOR 576427 ISSUED BY THE KHMELNYTSK GUUMVD OF UKRAINE IN THE KHMELNYTSK REGION 03 1 0 1 998

UKRAINE
HS-Code
Products
3004900000
1. UNREGISTERED MEDICINAL PRODUCTS FOR HUMAN USE THERAPEUTIC AND PROPHYLACTIC FOR INDIVIDUAL USE NOT CONTAINING NARCOTIC DRUGS AND PRECURSORS NOT IN AEROSOL PACKAGING PACKAGED FOR RETAIL SALE: -TAFINLAR (DABRAFENIB) CAPSULE 75 MG 1 X 120 NVS U57 -1 120 CAPSULES IN EACH. TOTAL: 120 CAPSULES. SERIAL NUMBER OF THE MANUFACTURER: VR3M. SHELF LIFE UNTIL SEPTEMBER 30 2021. ACTIVE ACTIVE SUBSTANCE DABRAFENIB. IT IS DELIVERED IN PACKING FROM A THERMAL BOX WITH REFRIGERANTS AND THE INCLUDED DISPOSABLE SENSOR OF CONTROL OF TEMPERATURE OF INTERNAL SPACE OF A PACKING SET. THE GOODS ARE IMPORTED FREE OF CHARGE WITHOUT THE RIGHT TO SELL SELL AND ONLY FOR CLINICAL TRIALS.
3004900000
1. UNREGISTERED MEDICINAL PRODUCTS FOR HUMAN USE THERAPEUTIC AND PROPHYLACTIC FOR INDIVIDUAL USE NOT CONTAINING NARCOTIC DRUGS AND PRECURSORS NOT IN AEROSOL PACKAGING PACKAGED FOR RETAIL SALE: -DRB436 (DABRAFENIB) 75 MG 28 X HNGC CAPSULE LABBLEDBOTTLE -13 PACKS CAPSULES IN EACH. TOTAL: 364 CAPSULES. SERIAL NUMBER OF THE MANUFACTURER: 1010023597. PACKING SERIES: 2036322. SHELF LIFE UNTIL JULY 31 2022. ACTIVE SUBSTANCE DABRAFENIB. COMES PACKAGED IN A THERMAL BOX WITH REFRIGERANTS AND A DISPOSABLE SENSOR TO CONTROL THE TEMPERATURE OF THE INTERIOR OF THE PACKAGING. THE GOODS ARE IMPORTED FREE OF CHARGE WITHOUT THE RIGHT TO SELL SELL AND ONLY FOR CLINICAL TRIALS.
3004900000
1.UNREGISTERED MEDICINAL PRODUCTS FOR HUMANS THERAPEUTIC AND PROPHYLACTIC FOR INDIVIDUAL USE DO NOT CONTAIN NARCOTIC DRUGS AND PRECURSORS NOT IN AEROSOL PACKAGING PACKAGED FOR RETAIL TRADE: -DRB436 (DABRAFENIB) 75MG 28X HNGC LABELED BOTTLE-5 PACKS OF 28 CAPSULES EACH. TOTAL: 140 CAPSULES. SERIAL NUMBER OF THE MANUFACTURER: 1010023596. PACKING SERIES: 2036321. SHELF LIFE UNTIL JULY 31 2022. ACTIVE SUBSTANCE DABRAFENIB. COMES IN A PACKAGE FROM A THERMAL BOX WITH REFRIGERANTS AND THE SENSOR OF CONTROL OF TEMPERATURE OF INTERNAL SPACE OF A PACKING SET. THE GOODS ARE IMPORTED FREE OF CHARGE WITHOUT THE RIGHT TO SELL SELL AND ONLY FOR CLINICAL TRIALS.
TOV VENTA Â LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. LIPRIMAR® TABLETS FILM-COATED 20 MG 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARTON / 1 TABLET CONTAINS ATORVASTATIN CALCIUM EQUIVALENT TO 20 MGATORVASTATIN / PFIZER PHARMACEUTICALS LLS (PRODUCTION IN BULK QUALITY CONTROL) USAINSPRA® FILM-COATED TABLETS 3 MG TABLETS 50 MG EACH; BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS 50 MG OF EPLERENONE / PFIZER PHARMACEUTICALS LLS (PRODUCTION OF THE DRUG IN BULK AND QUALITY CONTROL) USA CELEBREX® CAPSULES OF 200 MG 10 CAPSULES IN A BLISTER 2 BLISTERS IN A CARDBOARD BOX / 1 CAPSULE CONTAINS 200 MG / PFIZER PHARMACEUTICALS LLS (BULK PRODUCTION AND QUALITY CONTROL) USA
3004900000
1. MEDICINES FOR PEOPLE FREE OF: ANTIBIOTICS ALKALOIDS HORMONES VITAMINS PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: ALERZINE DROPS ORAL SOLUTION 5 MG / ML 20 ML IN A BOTTLE WITH A DROPPER 1 BOTTLE 1 DROPPER IN A DROPPER MG OF LEVOCETIRIZINE DIHYDROCHLORIDE (CORRESPONDING TO 4.21 MG OF LEVOCETIRIZINE) / CJSC EGIS PHARMACEUTICAL PLANT HUNGARYKETILEPT® FILM-COATED TABLETS 25 MG OF 10 TABLETS IN A BLISTER 3 BLISTERS IN A CARTON PACK EACH CONTAINING 28 MG / 1 TABLET CORRESPONDS TO 25 MG OF QUETIAPINE / CJSC PHARMACEUTICAL PLANT EGIS HUNGARYHARTIL® - AM CAPSULES OF 10 MG / 10 MG OF 10 CAPSULES IN A BLISTER; 3 BLISTERS IN A CARTON / 1 CAPSULE CONTAINS 10 MG OF RAMIPRIL AND 10 MG OF AMLODIPINE (CORRESPONDING TO 13.9 MG OF AMLODIPINE BESYLATE) / CJSC PHARMACEUTICAL PLANT EGIS HUNGARYHARTIL® TABLETS OF 5 MG 7 TABLETS IN A BLISTER; 4 BLISTERS IN A CARDBOARD BOX 1 TABLET CONTAINS RAMIPRIL 5 MG / CJSC PHARMACEUTICAL PLANT EGIS
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. PHYSIOTENS® FILM-COATED TABLETS 0.4 MG EACH 14 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD BOX / 1 TABLET CONTAINS MOXONIDINE 0.4 MG / MYLAN LABORATORIES CAC (FULL CYCLE PRODUCTION) FRANCE. DUSPATALIN® PROLONGED-RELEASE CAPSULES SOLID 200 MG; 15 CAPSULES IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX / 1 CAPSULE CONTAINS MEBEVERINE HYDROCHLORIDE 200 MG / MYLAN LABORATORIES CAC FRANCE.
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
Get most profitable Buyers & Suppliers
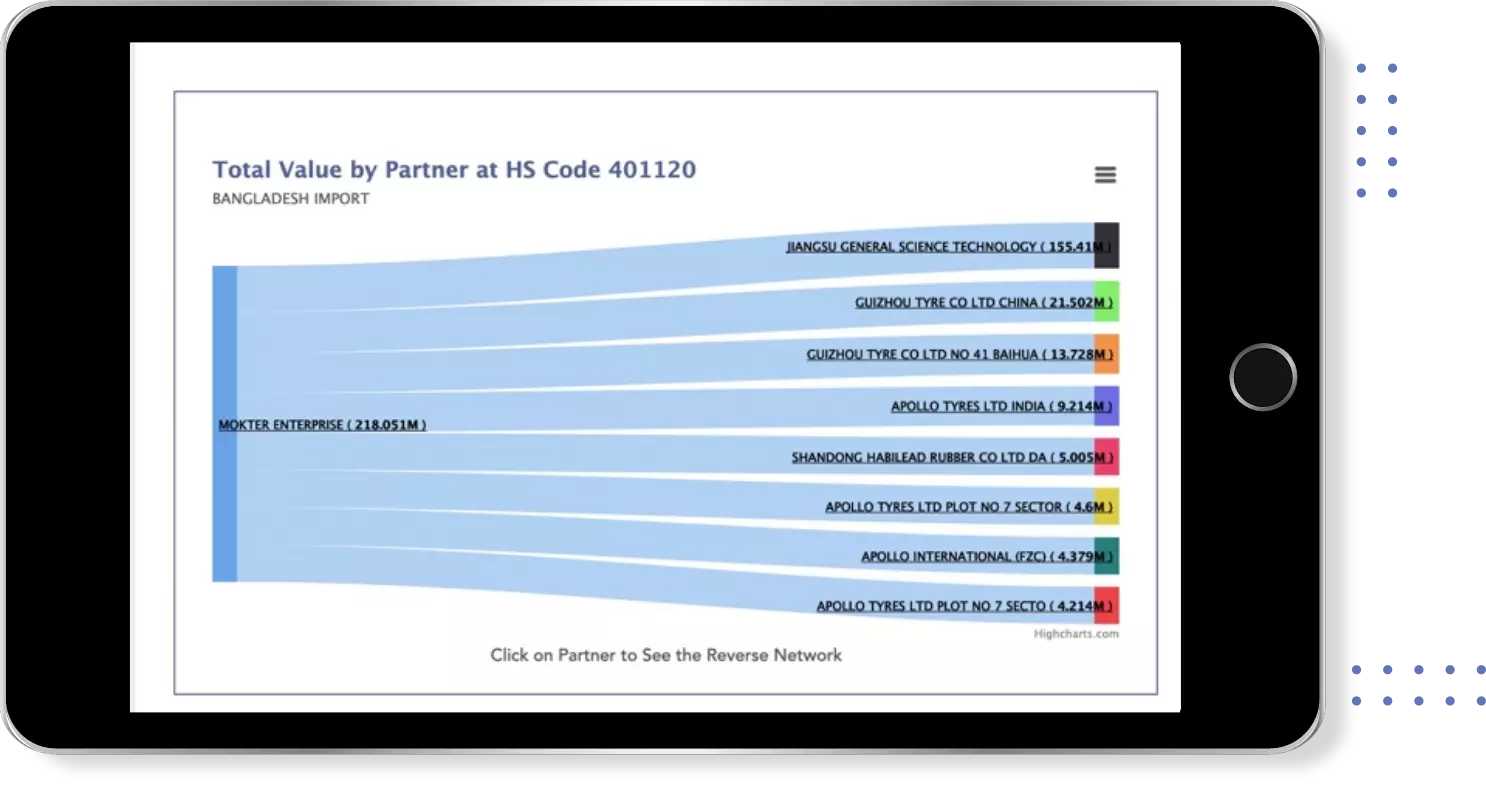
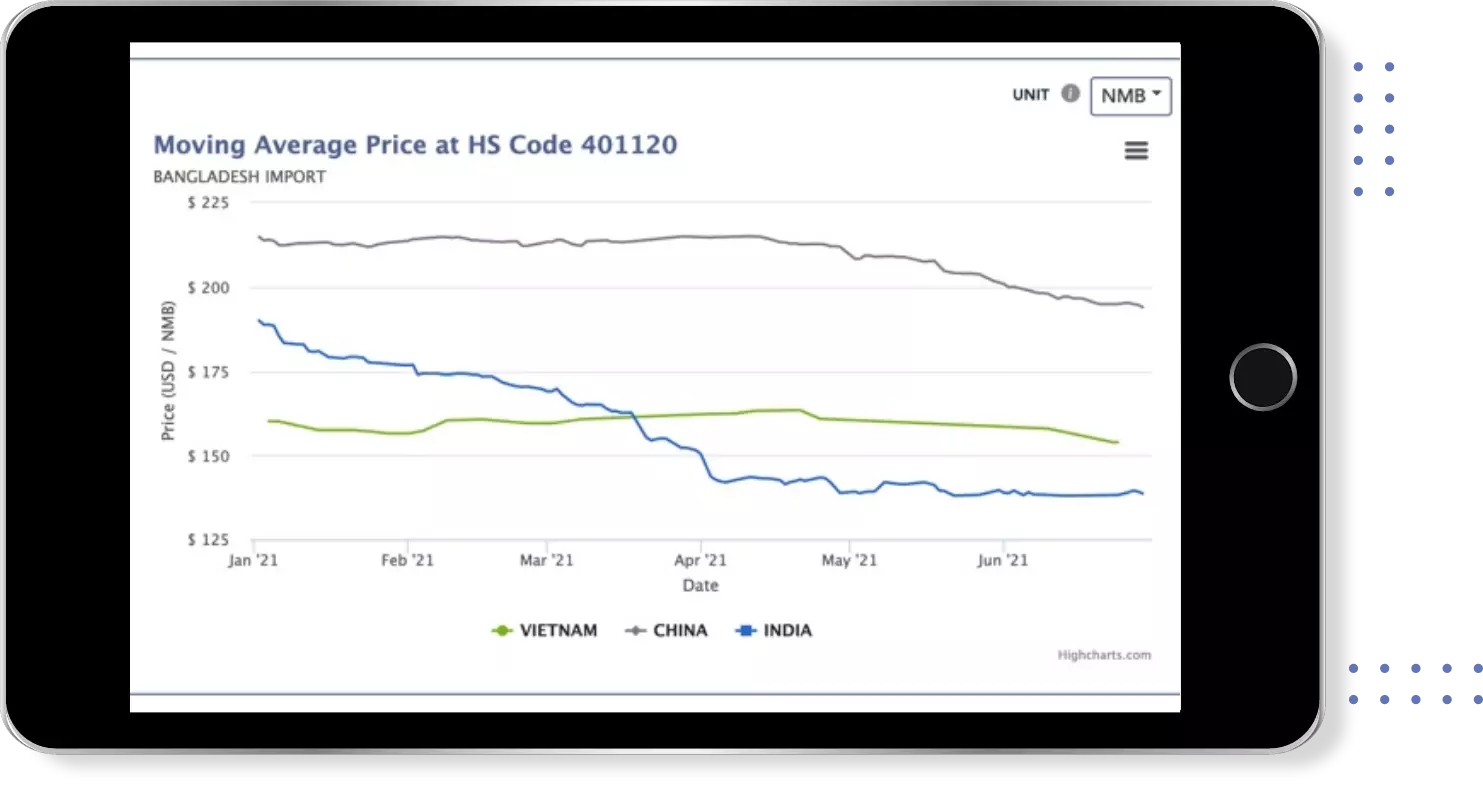
Instant insights on Price , Quantity & Value trends
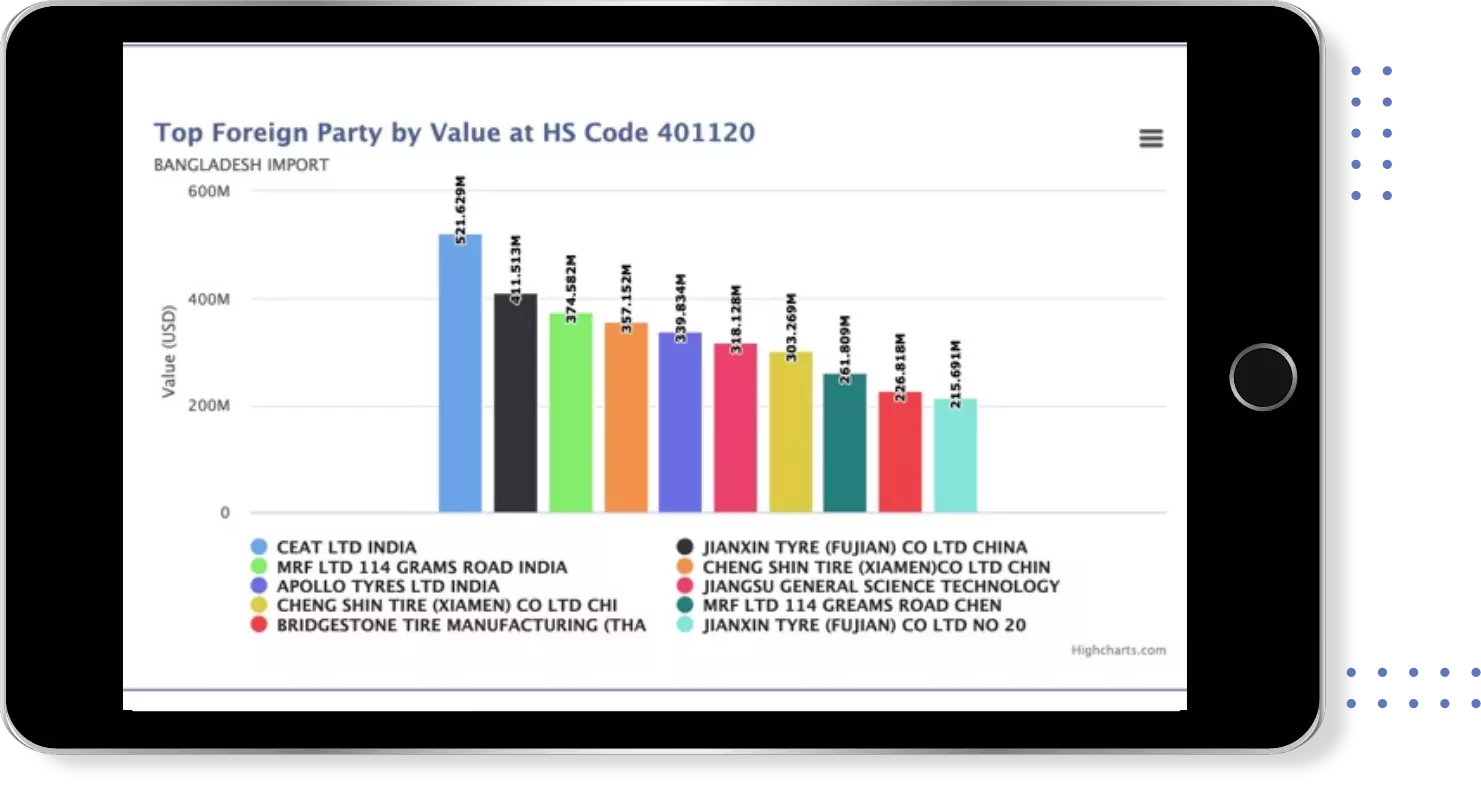
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
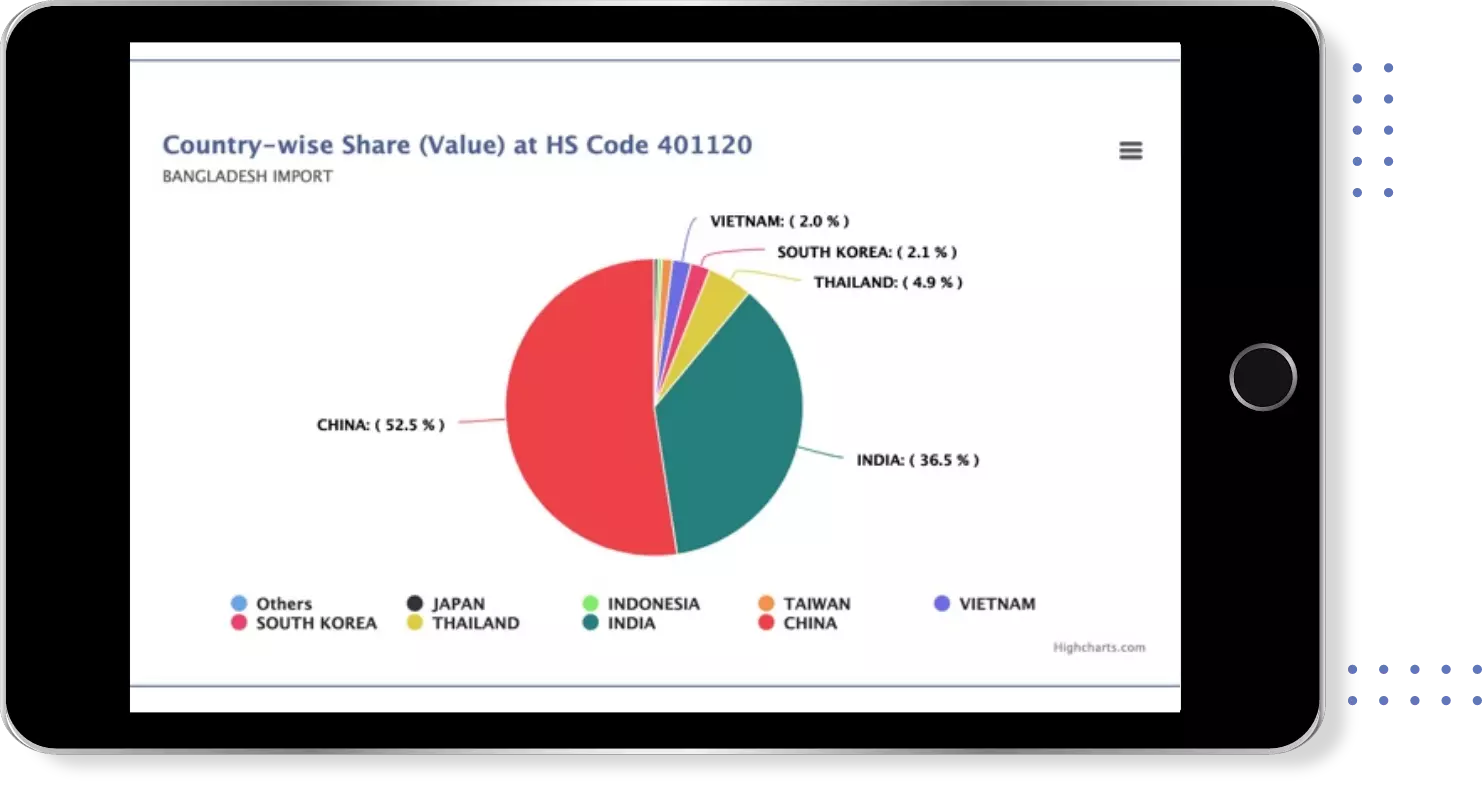
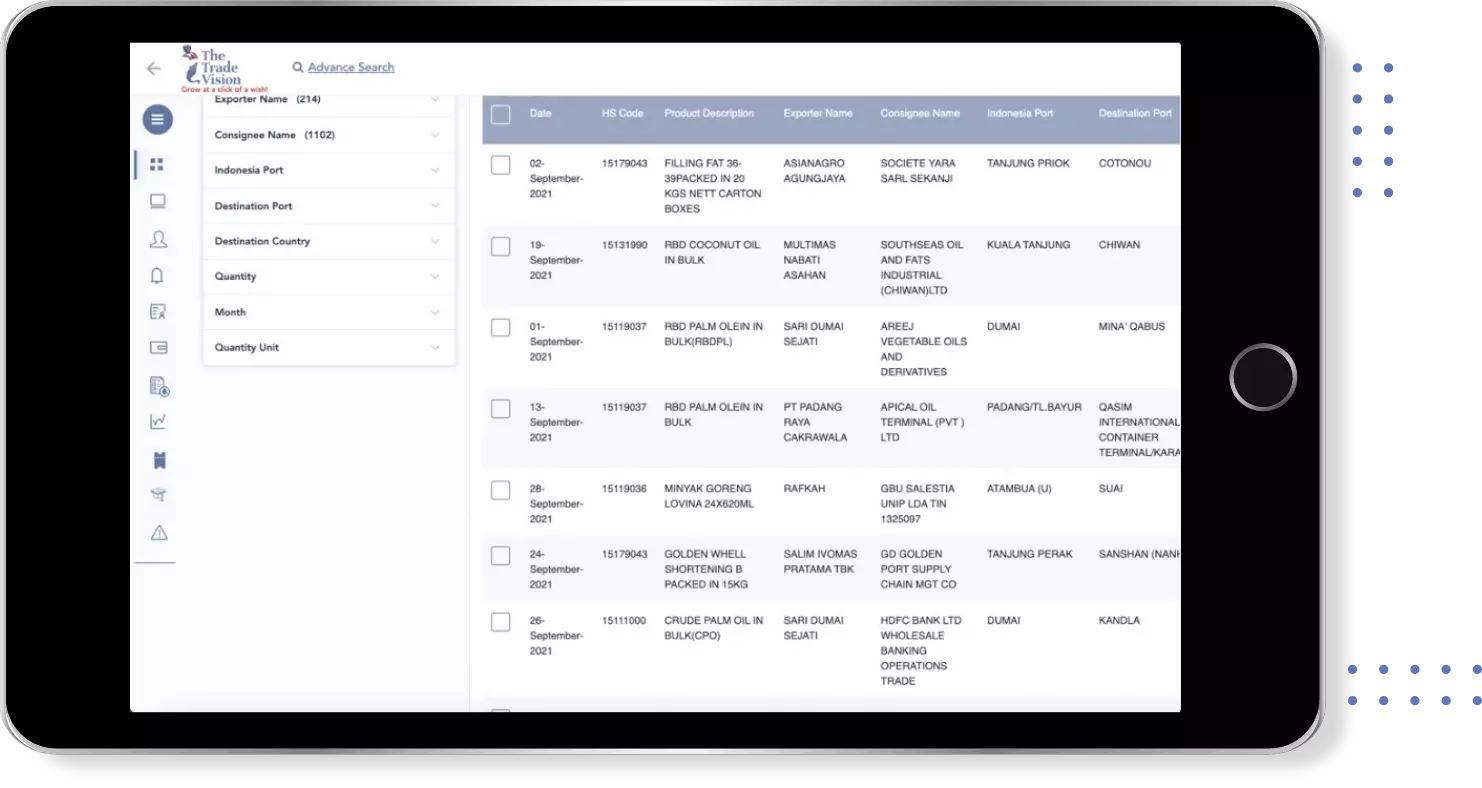
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.