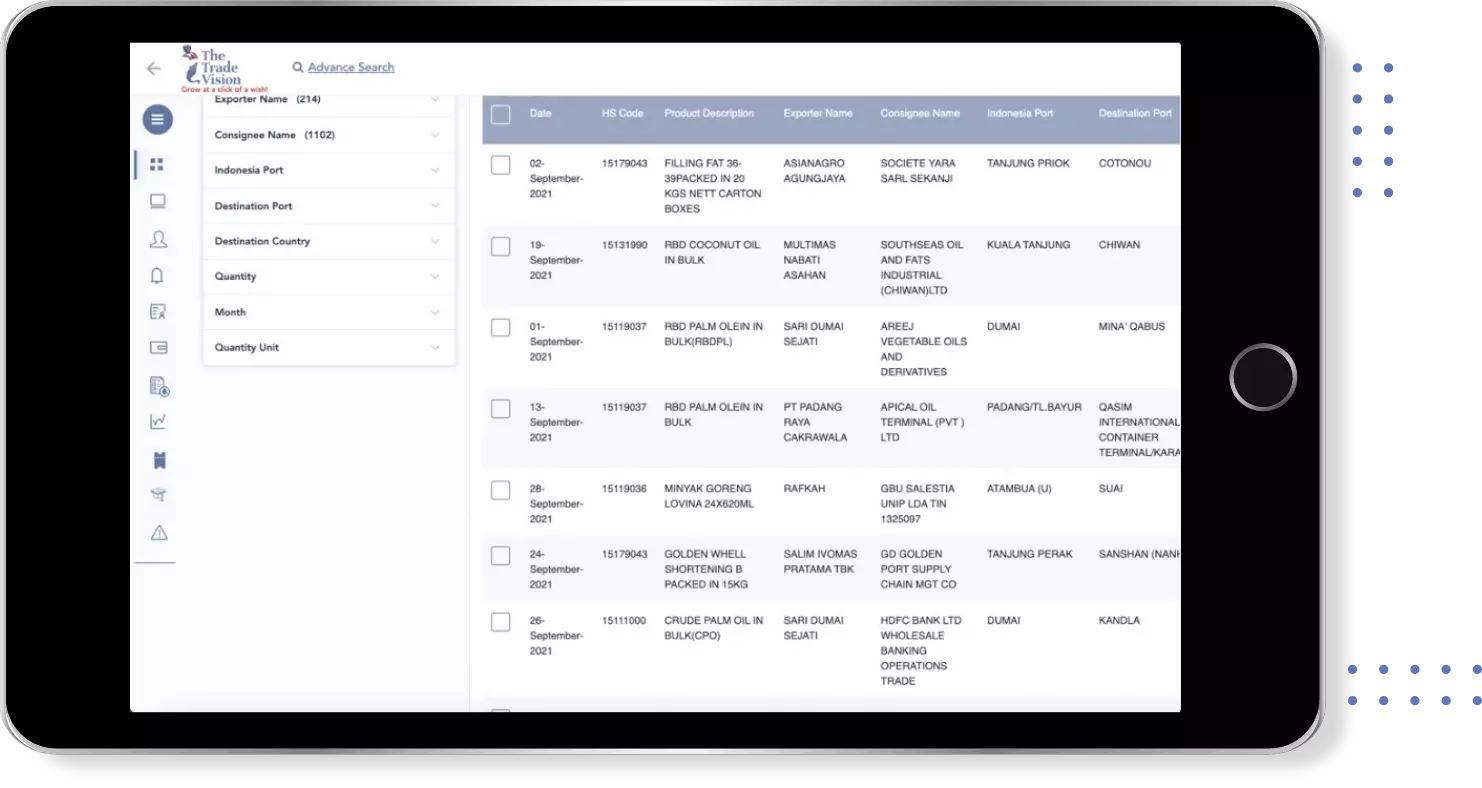
Find verified buyers and sellers of vaccine vial monitor in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- vaccine vial monitor import export data
Overview
- The top 3 supplier countries for vaccine vial monitor are “UNITED STATES OF AMERICA” , “DENMARK” , “SINGAPORE” ,
- The top 3 Buyer countries for vaccine vial monitor are “UZBEKISTAN” , “PAKISTAN” , “PHILIPPINES” , .
- Top 3 Product Categories for vaccine vial monitor are 482190, 300241, 482110,
The above summary is based on TTV’s Global Export Import data of vaccine vial monitor, compiled from 180+ countries export import shipments updated till .
Global Buyers of Vaccine vial monitor
SERVICE OF SANITARY AND EPIDIMIOLOGICAL WELFARE AND PUBLIC HEALTH OF THE RUZ

UZBEKISTAN
HS-Code
Products
3002200009
1 . HUMAN VACCINES: S359524 ROTAVIRUS VACCINE ORAL 2DS 5ML WITH VACCINE VIAL MONITOR (VVM) SER NO1 461 O01 5 YEAR. TO 03. 24G (32954FL) NO. 1 461 O01 6 YEAR. TO 03. 24G (8721 0FL) NO. 1 461 O01 7 YEAR. BEFORE 03
3002200009
1 . VACCINES FOR HUMANS: S359524 ROTAVIRUS VACCINE ORAL 2DS 5ML WITH VACCINE VIAL MONITOR (VVM) SER NO1 461 O008 YEAR. TO 02. 24G (201 82FL) NO. 1 461 O009 YEAR. TO 03. 24G (86992FL) NO. 1 461 O01 0 YEAR. TO 0
3002200009
1 . S3591 23 DTP VACCINE ADSORBED DIPHTHERIA TETANUS AND PERTUSSIS VACCINE PEDIATRIC DOSE VIAL OF 1 0 DOSE WITH VACCINE VIAL MONITOR (VVM) 5ML - 10 DOSES (LIQUID
SERVICE SANITARNO EPIDEMIOLOGICHESKOGO BLAGOPOLUCHIYA I OBSHCHESTVENNOGO ZDOROVYA RUZ

UZBEKISTAN
HS-Code
Products
3002410000
1. S359323 HEP B VACCINE PEDIATRIC VIAL OF 10 DOSE HEPATITIS B VACCINE DNA RECOMBINANT 10 DOSE VIAL PEDIATRIC DOSE WITH VACCINE VIAL MONITOR (VVM) INJECTION SUSPENSION PEDIATRIC DOSE 0.5 ML 10 DOSES IN A BOTTLE
3002410000
1. S359323 HEP B VACCINE PEDIATRIC VIAL OF 10 DOSE HEPATITIS B VACCINE DNA RECOMBINANT 10 DOSE VIAL PEDIATRIC DOSE WITH VACCINE VIAL MONITOR (VVM) INJECTION SUSPENSION PEDIATRIC DOSE 0.5 ML 10 DOSES IN A BOTTLE
3002410000
1. S359848 ROTAVIRUS VACCINE LIQ. ORAL 2 DS VIAL 2 DOSE VIAL W/VVM/ ROTAVIRUS VACCINE ORAL 2 DOSE WITH MONITOR. 346000FL. SER NO. 3953O010 (16000FL) NO. 3953O011 (91480 FL) NO. 3953O012 (90770 FL) NO. 3953O013 (91310 FL) NO. 3953O014 (56440 FL.) YEAR BEFORE JAN 2025; IN TO
GU TSENTR BEZOPASNOSTI FARMATSEVTICHESKOY PRODUKTSII

UZBEKISTAN
HS-Code
Products
3002410000
1. SAMPLES SUPPLIED FOR STATE REGISTRATION IN THE REPUBLIC OF UZBEKISTAN: VACCINE FOR POLIO-POLIO VACCINE POLIOMYELITIS VACCINE (ORAL) BIVALENT TYPE 1 AND 3 - 20 DOSE WITH DROPPERS WITH VACCINE VIAL MONITOR (VVM) WITH REFERENCE STANDARDS. - COUNT
3002410000
1. SAMPLES SUPPLIED FOR STATE REGISTRATION IN THE REPUBLIC OF UZBEKISTAN: VACCINE FOR POLIO-POLIO VACCINE POLIOMYELITIS VACCINE (ORAL) BIVALENT TYPE 1 AND 3 - 10 DOSE WITH DROPPERS WITH VACCINE VIAL MONITOR (VVM) WITH REFERENCE STANDARDS. - COUNT
INTERNATIONAL ORGANIZATION FOR MIGRATION

KENYA
HS-Code
Products
3002200000
S359143 TD VACCINE ADSORBED TETANUS & DIPHTHERIA VACCINE WITH VAC VIAL MONITOR;
Global Suppliers of Vaccine vial monitor
TEMPTIME CORPORATION

UNITED STATES OF AMERICA
HS-Code
Products
482190900000
HEATMARKER VACCINE VIAL MONITOR SII HEATMARKER VVM2 HAPA FULL LABEL 10K/ROLL SII PART 20011249 TTC PART 10470(PO NO. 51
482190900000
HEATMARKER VACCINE VIAL MONITOR HEATMARKER VVM30 DOT (504) A SII PART 20000688 TTC PART 10389(PO NO. 5100022590)
482190900000
HEATMARKER VACCINE VIAL MONITOR SII VVM7 HAPA FULL LABLE 20017668 - 10904 REG NO. ORIGINAL-DPIIT-PPR-2023-035866 DT. 12
TEMPTIME CORP

UNITED STATES OF AMERICA
HS-Code
Products
482190900000
HEATMARKER VACCINE VIAL MONITOR. HEATMARKER VVM DOT. SII PART 20007913. TTC PART 10832.(PO NO. 5100022939)
482190900000
HEATMARKER VACCINE VIAL MONITOR. VVM 7 HAPA FULL LABEL. SII PART 20014663. TTC PART 10903.(PO NO. 5100022179)
482190900000
HEATMARKER VACCINE VIAL MONITOR. SII HEATMARKER VVM7 HAPA FULL LABEL. SII PART 20014663 TTC PART 10903. (PO NO.510002434
SERUM INSTITUTE OF INDIA PVT LTD

DENMARK
HS-Code
Products
300241000000
1. S359848 ROTAVIRUS VACCINE LIQ. ORAL 2 DS VIAL 2 DOSE VIAL W/VVM/ ROTAVIRUS VACCINE ORAL 2 DOSE WITH MONITOR. 193000FL. SER NO. 3953O014 (34840 FL.) YEAR TO JAN 2025; NO. 3953?015(91300FL) NO.3953?016(66860FL) YEAR BEFORE FEB 2025; INCLUDED WITH A SPECIAL ORAL SYRINGE
300241000000
1. S359848 ROTAVIRUS VACCINE LIQ. ORAL 2 DS VIAL 2 DOSE VIAL W/VVM/ ROTAVIRUS VACCINE ORAL 2 DOSE WITH MONITOR. 346000FL. SER NO. 3953O010 (16000FL) NO. 3953O011 (91480 FL) NO. 3953O012 (90770 FL) NO. 3953O013 (91310 FL) NO. 3953O014 (56440 FL.) YEAR BEFORE JAN 2025; IN TO
300241000000
1. S359163 MEASLES VACCINE LIVE ATTENUATED FREEZE-DRIED VIAL OF 10 DOSES WITH DILUENT. WITH VACCINE VIAL MONITOR (VVM)/ MEASLES VACCINE LIVE ATTENUATED FREEZE-DRIED WITH DILUENT - 294900 BOTTLES SERIAL NO. 0043N051 (80460 PCS); NO. 0043N052 (899
XXMPTXXE CXXP

UNITED STATES OF AMERICA
HS-Code
Products
482190900000
HEATMARKER VACCINE VIAL MONITOR. VVM7 HAPA LABEL. SII PART 20014663 TTC PART 10903.(PO NO. 5100024805)
482190900000
HEATMARKER VACCINE VIAL MONITOR. VVM14 HAPA LABEL. SII PART 20010855 TTC PART 10460.(PO NO. 5100024810)
482190900000
HEATMARKER VACCINE VIAL MONITOR. VVM14 HAPA LABEL. SII PART 20010855 TTC PART 10460.(PO NO. 5100024810)
Global Export Import Trade Data for vaccine vial monitor
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UZBEKISTAN UZBEKISTAN | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UNITED STATES OF AMERICA UNITED STATES OF AMERICA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UNITED STATES OF AMERICA UNITED STATES OF AMERICA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 BANGLADESH BANGLADESH | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
Get most profitable Buyers & Suppliers
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries