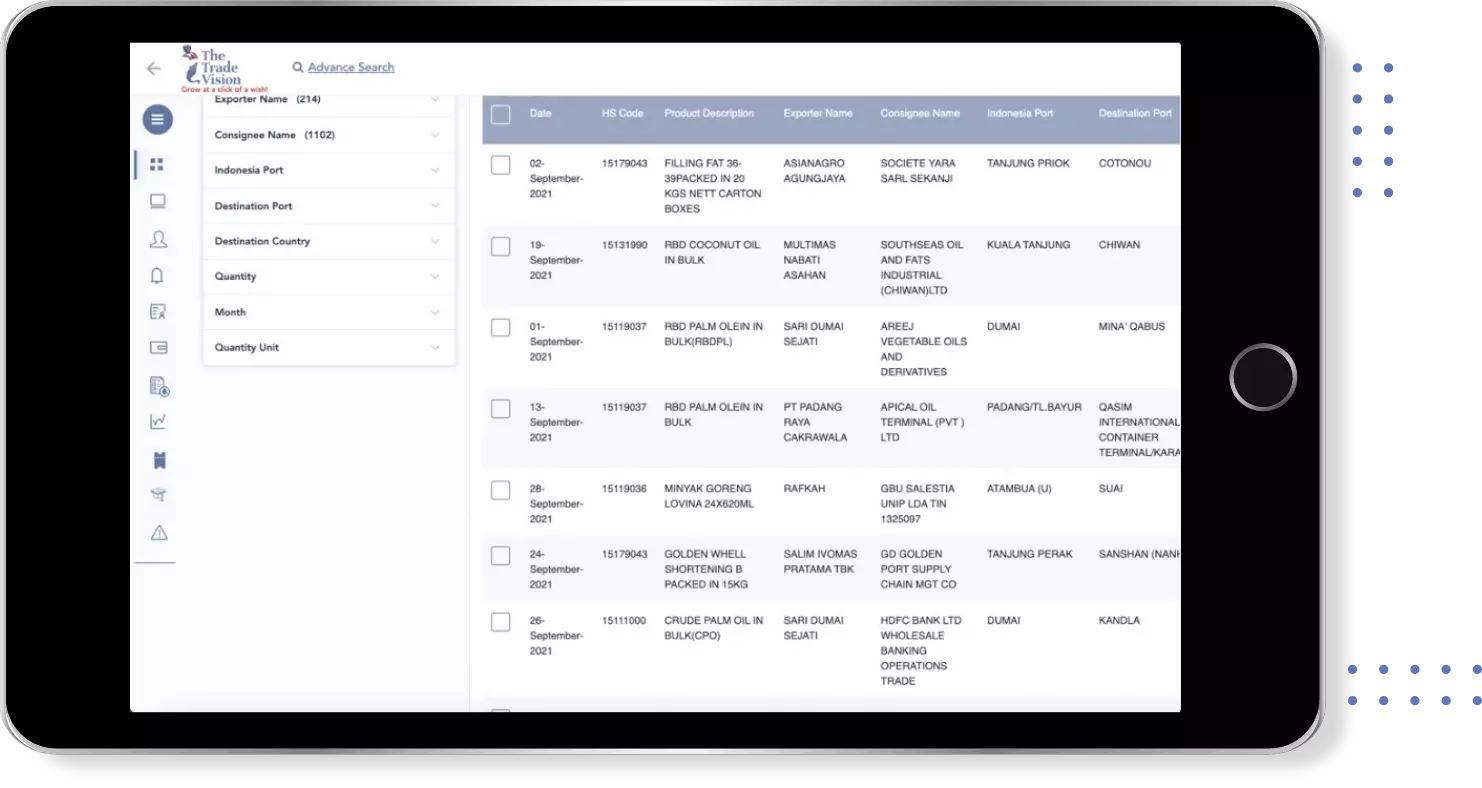
Find verified buyers and sellers of vaccine for polio in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- vaccine for polio import export data
Overview
- The top 3 supplier countries for vaccine for polio are “INDONESIA” , “ARGENTINA” , “FRANCE” ,
- The top 3 Buyer countries for vaccine for polio are “UKRAINE” , “UZBEKISTAN” , “ARGENTINA” , .
- Top 3 Product Categories for vaccine for polio are 300220, 300241, 300290,
The above summary is based on TTV’s Global Export Import data of vaccine for polio, compiled from 180+ countries export import shipments updated till .
- UKRAINE
- UZBEKISTAN
- ARGENTINA
- PHILIPPINES
- PAKISTAN
- KAZAKHSTAN
- INDONESIA
- PANAMA
- CAMEROON
- GHANA
- PARAGUAY
- PERU
- SRI LANKA
- TURKEY
- VIETNAM
- UNITED ARAB EMIRATES
- CONGO-KINSHASA
- KENYA
- AFGHANISTAN
- NETHERLANDS
- FRANCE
- NIGERIA
- MOZAMBIQUE
- MYANMAR
- UNITED KINGDOM
- RWANDA
- VENEZUELA
- SOUTH AFRICA
- PAPUA NEW GUINEA
- SAUDI ARABIA
- BELGIUM
- SPAIN
- QATAR
- BRAZIL
Global Buyers of Vaccine for polio
LTD PARTNERSHIP KNYAZ PHARMA LTD

KAZAKHSTAN
HS-Code
Products
3002410000
1 . GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZAE TYPE B CONJUGATED INFECTION. SUSPENSION FOR INJECTION. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPHTHERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM ® 1 SYRINGE 1 DOSE + 2 NEEDLES BATCH/SERIES: U3E021 V-341 500 PACKS. DATE OF PRODUCTION - 23. 02. 2020. EXPIRY DATE UP TO - 31. 01 . 2024. REGISTER. CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. NOT FOR VETERINARY. DO NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. 1 . GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZAE TYPE B CONJUGATED INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM ® 1 SYRINGE 1 DOSE + 2 NEEDLES BATCH/SERIES: U3E021 V-85000 PACK. DATE OF PRODUCTION - 23. 02. 2020. EXPIRY DATE UP TO - 31. 01 . 2024. REGISTER. CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. MANUFACTURER - SANOFI PASTER S.A. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:GEXAXIM ® PRODUCT NUMBER:4000331 1 0 QUANTITY:85000 PCS ; 2. GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLLESS) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. SOFTWARE 0. 5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM ® 1 SYRINGE 1 DOSE + 2 NEEDLES BATCH/SERIES: U3E021 V-85000 PACKS. DATE OF PRODUCTION - 23. 02. 2020. EXPIRY DATE UP TO - 31. 01 . 2024. REGISTER. CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. MANUFACTURER - SANOFI PASTER S.A. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:GEXAXIM ® PRODUCT NUMBER:4000331 1 1 QUANTITY:85000 PCS ; 3. GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLLESS) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM ® 1 SYRINGE 1 DOSE + 2 NEEDLES BATCH/SERIES: U3E021 V-85000 PACKS. DATE OF PRODUCTION - 23. 02. 2020. EXPIRY DATE UP TO - 31. 01 . 2024. REGISTER. CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. MANUFACTURER - SANOFI PASTER S.A. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:GEXAXIM ® PRODUCT NUMBER:4000331 1 2 QUANTITY:85000 PCS ; 4. GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARD
3002410000
1 . HEXAXIM VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZA TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZA TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND AGAINST BACTERIAL VACCINES. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. HEXAXIM ® 1 SYRINGE + 2 NEEDLES BATCH: U3E021 V. TOTAL - 30 PACKS. REGISTER CERTIFICATE: 1 3. 02. 201 9 RK-BP-5 NO. 020263. PRODUCTION DATE - 23. 02. 2020 / EXPIRY DATE UP TO - 31 . 01 . 2024. VKA DELIVERY ACCORDING TO THE APPENDIX TO THE DELIVERY CONTRACT: 30 DOSES FREE OF CHARGE WITH EACH BATCH SUPPLIED. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS.
3002410000
1 . PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPTHTERIA AND TETANIUS ACELLULAR PERTUSSIS INACTIVE POLIO AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED. LYOPHILIZATE COMPLETE WITH SUSPENSION. 0.5 ML/1 DOSE. LYOPHILIZATE FOR 1 DOSE IN A VIAL COMPLETE WITH A SUSPENSION IN A SYRINGE FOR 1 DOSE (0.5 ML) 1 VIAL AND 1 SYRINGE WITH ONE NEEDLE OR WITHOUT NEEDLE OR WITH TWO SEPARATE NEEDLE IN A COLORLESS POLYMERIC LOOP PACK. ON 1 BELL PACK IN A CARDBOARD BOX. ATC CODE: J07CA06 VACCINE AGAINST DIPHTHERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANUS. PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE BATCH/ SERIES: V0D351 M V0D361 M V0D371 M - 329370 PACKS. PRODUCTION DATES: 09. 09. 2020. EXPIRY DATES: 31 . 08. 2023. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. 1 . PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPTHTERIA AND TETANIUS; WHOOPING COUGH ACELLULAR; INACTIVATED POLIO AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED. LYOPHILIZATE COMPLETE WITH SUSPENSION. 0.5 ML/1 DOSE. LYOPHILIZATE FOR 1 DOSE IN A BOTTLE COMPLETE WITH SUSPENSION IN A SYRINGE FOR 1 DOSE (0.5 ML); 1 VIAL AND 1 SYRINGE WITH ONE NEEDLE OR WITHOUT NEEDLE OR WITH TWO SEPARATE NEEDLE IN COLORLESS POLYMERIC BLACK PACK. ON 1 BELL PACK IN A CARDBOARD BOX. ATC CODE: J07CA06 VACCINE AGAINST DIPHTHERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANUS. PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE. PRODUCTION DATE: 09/09/2020 EXPIRY DATE: 31 . 08. 2023. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:PENTAXIM GOODS NUMBER:1 1 091 681 0 QUANTITY:84832 PCS ; 2. PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPHTHERIA AND TETANIUS; WHOOPING COUGH ACELLULAR; INACTIVATED POLIO AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED. LYOPHILIZATE COMPLETE WITH SUSPENSION. 0.5 ML/1 DOSE. LYOPHILIZATE FOR 1 DOSE IN A BOTTLE COMPLETE WITH SUSPENSION IN A SYRINGE FOR 1 DOSE (0.5 ML); 1 VIAL AND 1 SYRINGE WITH ONE NEEDLE OR WITHOUT NEEDLE OR WITH TWO SEPARATE NEEDLE IN COLORLESS POLYMERIC BLACK PACK. ON 1 BELL PACK IN A CARDBOARD BOX. ATC CODE: J07CA06 VACCINE AGAINST DIPHTHERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANUS. PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE. PRODUCTION DATE: 09/09/2020 31 . 08. 2023. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:PENTAXIM GOODS NUMBER:1 1 091 6830 QUANTITY:85000 PCS ; 3. PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPTHTERIA AND TETANIUS; WHOOPING COUGH ACELLULAR; INACTIVATED POLIO AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED. LYOPHILIZATE COMPLETE WITH SUSPENSION. 0.5 ML/1 DOSE. LYOPHILIZATE FOR 1 DOSE IN A BOTTLE COMPLETE WITH SUSPENSION IN A SYRINGE FOR 1 DOSE (0.5 ML); 1 VIAL AND 1 SYRINGE WITH ONE NEEDLE OR WITHOUT NEEDLE OR WITH TWO INDIVIDUAL NEEDLES IN A COLORLESS POLYMERIC BLACK PACK. ON 1 BELL PACK IN A CARDBOARD BOX. ATC CODE: J07CA06 VACCINE AGAINST DIPHTHERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANUS. PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE. PRODUCTION DATE: 09/09/2020 EXPIRY DATE: 31 . 08. 2023. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:PENTAXIM GOODS NUMBER:1 1 091 6850 QUANTITY:85000 PCS ; 4. PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPTHTERIA AND TETANIUS; WHOOPING COUGH ACELLULAR; INACTIVATED POLIO AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED. LYOPHILIZATE COMPLETE WITH SUSPENSION. 0.5 ML/1 DOSE. LYOPHILIZATE FOR 1 DOSE IN A BOTTLE CO
LTD LIABILITY PARTNERSHIP KNYAZ PHARMA LTD

KAZAKHSTAN
HS-Code
Products
3002410000
1. PENTAXIM 1 BOTTLE 1 DOSE +1 SYRINGE. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPHTHERIA AND TETANUS ACELLULAR PERTUSH INACTIVATED POLIOMYELITIS AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATE. LYOPHILIZATE COMPLETE
3002410000
1. PENTAXIM 1 BOTTLE 1 DOSE +1 SYRINGE. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPHTHERIA AND TETANUS ACELLULAR PERTUSH INACTIVATED POLIOMYELITIS AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATE. LYOPHILIZATE COMPLETE
3002410000
1. PENTAXIM 1 BOTTLE 1 DOSE +1 SYRINGE. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPHTHERIA AND TETANUS ACELLULAR PERTUSH INACTIVATED POLIOMYELITIS AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATE. LYOPHILIZATE COMPLETE
SERVICE OF SANITARY AND EPIDIMIOLOGICAL WELFARE AND PUBLIC HEALTH OF THE RUZ

UZBEKISTAN
HS-Code
Products
3002200009
1 . VACCINES FOR HUMANS: SABIN 1 0-0-6VIAL 20DX1 00 C5-20 VVM DX WH ORAL POLIO VACCIPE (LIVE)/POLIO SABIN-ONE AND THREE (ORAL) 1 DOSE (2DROPS)BIVALENT ORAL POLIOMYELITIS VACCINE TYP 1 AND 3 1 00 VIALS X 20 DOSES (2ML)/ VACCINE POLIO SABIN TM ONE AND THREE (ORAL)
3002410000
1. HUMANITARIAN CARGO: VACCINES FOR HUMANS: 600055 IPV PACK 280X5D ENG-RUS UNICEF-A7/ POLIO VACCINE - 149240 BOTTLES 5 DOSES IN ONE BOTTLE. SERIES NO. 2308006A YEAR UNTIL 31-JAN-2026 (63816FL); NO. 2308007A YEAR UNTIL 31-JAN-2026 (64151FL); 2317005A YEAR.BEFORE
3002410000
1. VACCINES FOR HUMANS: SABIN 10-0-6VIAL 20DX100 C5-20 VVM DX WH ORAL POLIO VACCINE (LIVE) / ORAL POLIO VACCINE 1420 PACKAGES 100 FL. IN ONE PACKAGE TOTAL 142 000 FL. 20 DOSES IN ONE BOTTLE SER NO. AOR4A741AA PRODUCTION DATE: 01-SEP-2022 EXPIRATION DATE
OOO NOVOTEK

UZBEKISTAN
HS-Code
Products
3002200009
VACCINE FOR HUMANS 1) TETRAXIM SUSPENSION FOR INTRAMUSCULAR INTRODUCTION 1 DOSE - 0.5 ML (SYRINGES) (INN: DIPHTHERIA TETANUS PERTUSSIS /ACELLULAR COMPONENT/ AND INVACTIVATED POLIOMYELITIS ADSORBED VACCINE ) QTY: 400 PACK. SERIES: U3L493V EXPIRED UNTIL: 01 . 05.20
3002200009
VACCINE FOR HUMANS:1) HEXAXIM SUSPENSION FOR INTRAMUSCULAR ADMINISTRATION 1 DOSE-0.5ML (SYRINGES) INN: DIPHTHERIA TETANUS PERTUSSIS (ACELULLAR COMPONENT) HEPATITIS B (RDNA) POLIOMYELITIS (INACTIVATED) HAEMOPHILUS INFLUNZAE TYBE B CONJUZAE TYBE B VACCINE (ADS
3002200009
VACCINE FOR HUMANS:1) TETRAXIM SUSPENSION FOR INTRAMUSCULAR INTRODUCTION 1 DOSE - 0.5 ML (SYRINGES) INN: DIPHTHERIA TETANUS PERTUSSIS /ACELLULAR COMPONENT/ AND INVACTIVATED POLIOMYELITIS ADSORBED VACCINE QTY: 300 SERIES: V3C5 EXPIRED UNTIL: 01 . 08.2023
Global Suppliers of Vaccine for polio
BIO FARMA (PERSERO)

INDONESIA
HS-Code
Products
30022090
MONOVALENT BULK ORAL POLIOMYELITISVACCINE TYPE-2 CANDIDATE (NOPV2) FORHUMAN VACCINE
30022090
MONOVALENT BULK ORAL POLIOMYELITISVACCINE TYPE-2 CANDIDATE (NOPV2) FORHUMAN VACCINE
30022020
NOVEL ORAL POLIOMYELITIS VACCINE TYPE 2(NOPV2) BIVALENT ORAL POLIOMYELITISVACCINE (BPOV) DROPPER FOR NOPV2 DROPPER FORBOPV
BIO FARMA

INDONESIA
HS-Code
Products
30022090
NOVEL ORAL POLIOMYELITIS TYPE 2 VACCINECANDIDATE (NOPV2) FOR HUMAN VACCINE
30022090
NOVEL ORAL POLIOMYELITIS TYPE 2 VACCINECANDIDATE (NOPV2) FOR HUMAN VACCINE
SANOFI PASTEUR SA

FRANCE
HS-Code
Products
300241000000
1 . GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION.
300241000000
1 . GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZAE TYPE B CONJUGATED INFECTION. SUSPENSION FOR INJECTION. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM® 1 SYRINGE 1 DOSE + 2 NEEDLES. DELIVERY ACCORDING TO THE APPENDIX TO THE DELIVERY CONTRACT: 30 DOSES FREE OF CHARGE WITH EACH SUPPLIED BATCH. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. 1 . GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZAE TYPE B CONJUGATED INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM® 1 SYRINGE 1 DOSE + 2 NEEDLES. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. BATCH/SERIES: U3E021 V-30 PACKS. PRODUCTION DATE: 23.02.2020. EXPIRY DATE: 31. 01 . 2024. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:GEXAXIM ® PRODUCT NUMBER:4000331 20 QUANTITY:30 PCS ; 2. GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLLESS) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 DIPHTHERIA VACCINE HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM® 1 SYRINGE 1 DOSE + 2 NEEDLES. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. BATCH/SERIES: U3N229V-30 PACKS. DATE OF PRODUCTION: 23.07.2020. EXPIRY DATE: 30.06.2024. :MANUFACTURER:SANOFI PASTER S.A. 3. GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. SOFTWARE 0. 5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM® 1 SYRINGE 1 DOSE + 2 NEEDLES. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. BATCH/SERIES: U3N246V-30 PACKS. PRODUCTION DATE: 04. 08. 2020. EXPIRY DATE: 31 . 07. 2024. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:GEXAXIM ® PRODUCT NUMBER:4000331 60 QUANTITY:30 PCS ; 4. GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELL-FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZAE TYPE B CONJUGATED INFECTION. SUSPENSION FOR INJECTION J. 0.5 ML/1 DOSE. ON 0.5 ML (1 DOSE) IN A SYRINGE WITH ONE OR TWO NEEDLE. 1 SYRINGE IN A CARDBOARD BOX. ATC: J07CA09 VACCINE AGAINST DIPTHTERIA HAEMOPHILUS INFLUENZAE TYPE B PERTUSSIS POLIO TETANEUS HEPATITIS B. COMBINATION OF ANTI-VIRAL AND ANTI-BACTERIAL VACCINES. HEXAXIM® 1 SYRINGE 1 DOSE + 2 NEEDLES. NOT FOR VETERINARY. DOES NOT CONTAIN ALCOHOL. THE VACCINE IS IN A SPECIAL THERMAL CONTAINER WITH REFRIGERANTS. BATCH/SERIES: V3B695V-30 PACKS. PRODUCTION DATE: 02. 08. 2020. EXPIRY DATE: 31 . 07. 2024. :MANUFACTURER:SANOFI PASTER S.A. TRADEMARK:GEXAXIM ® ARTICLE
300241000000
1 . GEXAXIM ® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETANIUS PERTUSSIS (CELLULAR FREE) HEPATITIS B (RECOMBINANT RDNA) POLIO (INACTIVATED) AND HEMOPHILUS INFLUENZAE TYPE B CONJUGATED HEMOPHILUS INFECTION. SUSPENSION FOR INJECTION.
SANOFI PASTEUR S A

FRANCE
HS-Code
Products
300241000000
1. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPHTHERIA AND TETANUS ACELLULAR PERTUSSIS INACTIVATED POLIOMYELITIS AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED. 0.5ML/1 DOSE. LYOPHILIZATE 1 DOSE IN A BOTTLE SET
300241000000
1 . PENTAXIM 1 VIAL 1 DOSE +1 SYRINGE. PENTAXIM ADSORBED VACCINE FOR THE PREVENTION OF DIPTHTERIA AND TETANIUS ACELLULAR PERTUSSIS INACTIVE POLIO AND INFECTION CAUSED BY HAEMOPHILUS INFLUENZAE TYPE B CONJUGATED. LYOPHILIZATE IN COMPLETE
300241000000
1. HEXAXIM® VACCINE (ADSORBED) AGAINST DIPHTHERIA TETENIUS PERTONUS (CELL-FREE) HEPATITIS B (RECOMBINANT RDNA) POLIOMYELITIS (INACTIVATED) AND HEMOPHILIUS INFLUENZAE TYPE B CONJUGES ROVED. SUSPENSION FOR INJECTION.
Global Export Import Trade Data for vaccine for polio
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UKRAINE UKRAINE | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 FRANCE FRANCE | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 FRANCE FRANCE | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UNITED ARAB EMIRATES UNITED ARAB EMIRATES | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
Get most profitable Buyers & Suppliers
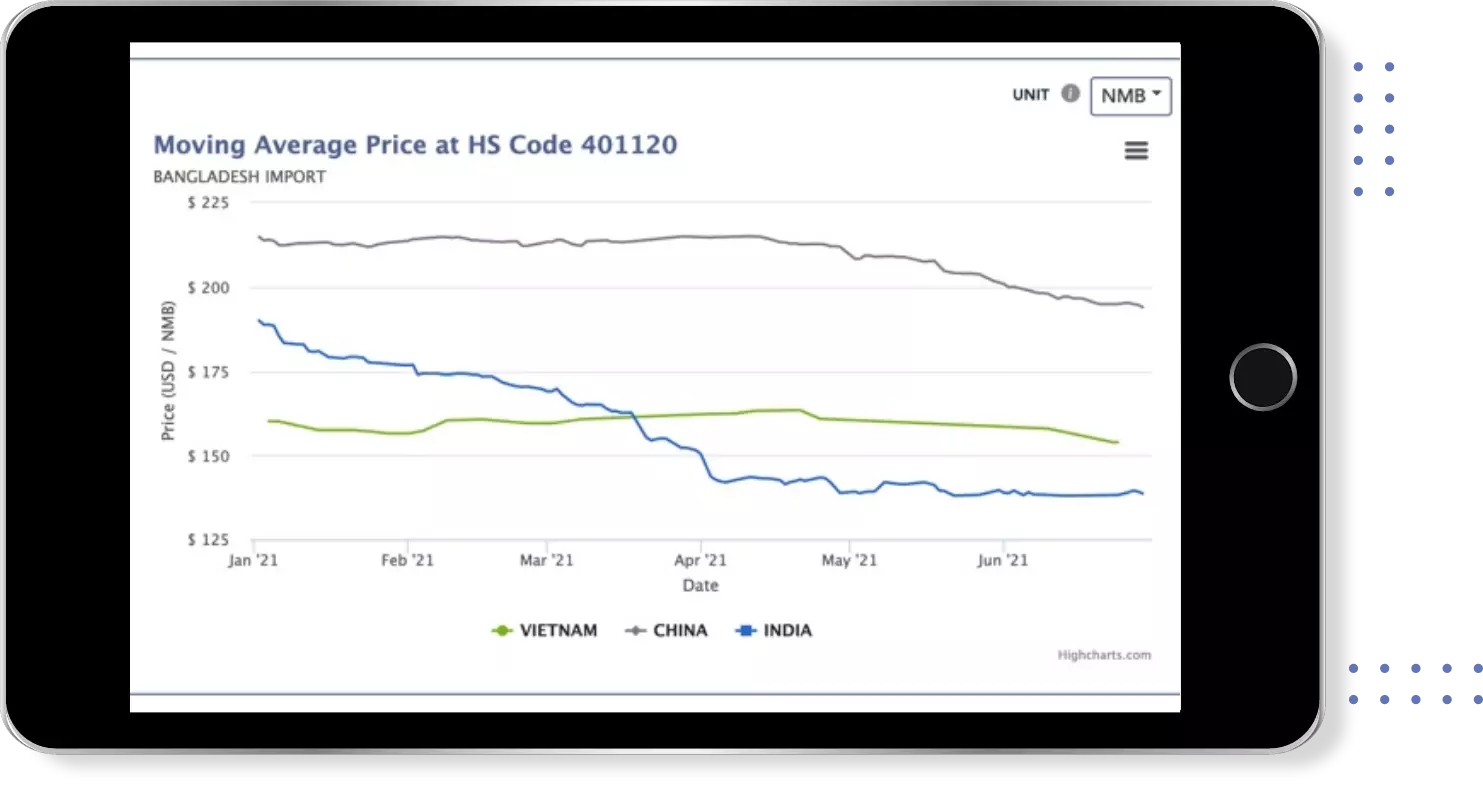
Instant insights on Price , Quantity & Value trends
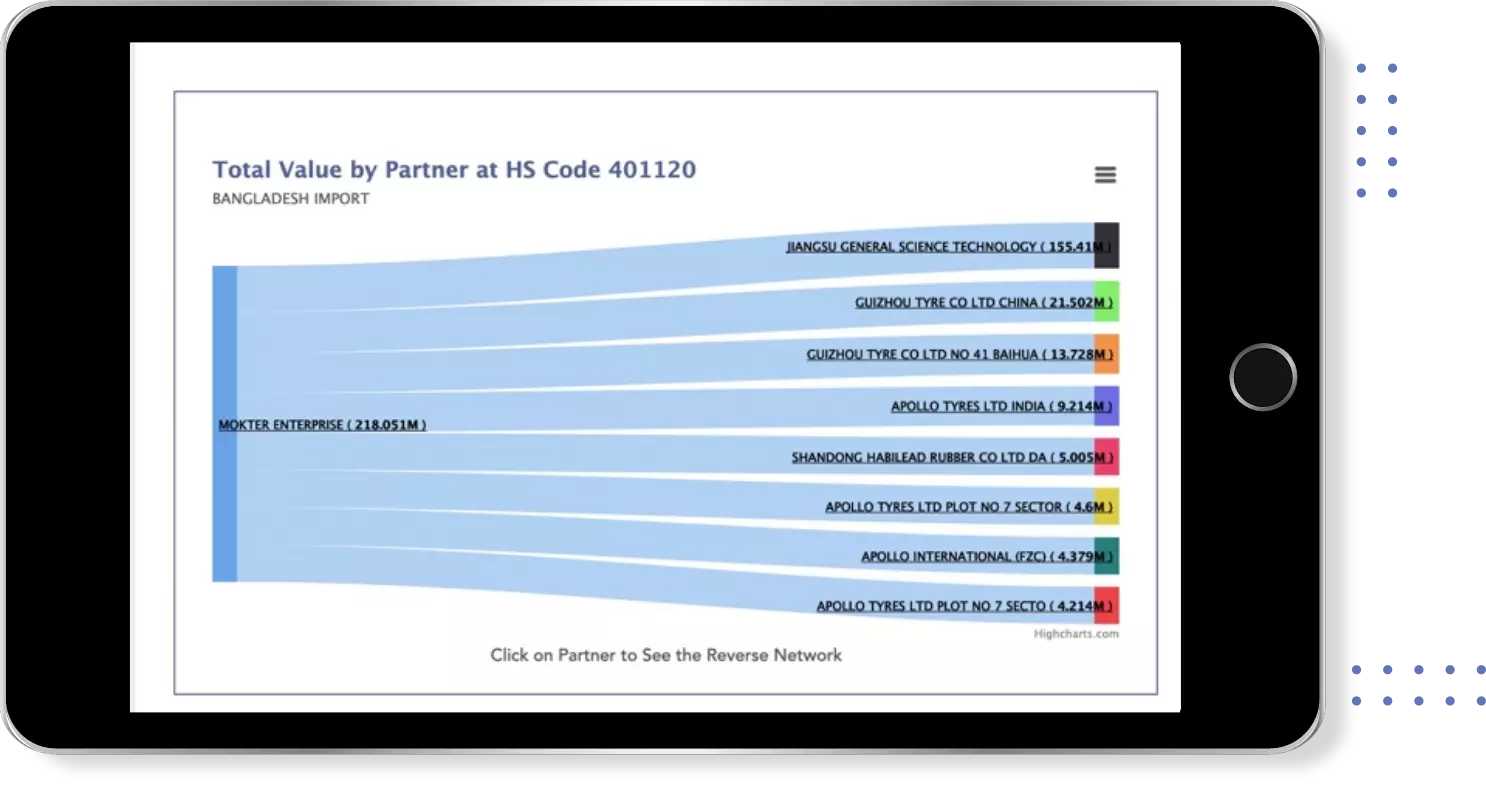
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries