Find verified buyers and sellers of paediatric co trimoxazole in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- paediatric co trimoxazole import export data
Overview
- The top 3 Buyer countries for paediatric co trimoxazole are “PAKISTAN” , “NETHERLANDS” , “PAPUA NEW GUINEA” , .
- Top 3 Product Categories for paediatric co trimoxazole are 300420, 300490, 300450,
The above summary is based on TTV’s Global Export Import data of paediatric co trimoxazole, compiled from 180+ countries export import shipments updated till .
Global Buyers of Paediatric co trimoxazole
IMRES

NETHERLANDS
HS-Code
Products
300420200000
PAEDIATRIC CO-TRIMOXAZOLE TABLETS BP 120MG (3600 X 1000) - JAR
300420200000
PAEDIATRIC CO-TRIMOXAZOLE TABLETS BP 120MG (750 X 1000) - JAR
300420200000
PAEDIATRIC CO-TRIMOXAZOLE TABLETS BP 120MG (400 X 1000) - JAR
BORNEO PACIFIC PHARMACEUTICALS LTD

PAPUA NEW GUINEA
HS-Code
Products
300420200000
PAEDIATRIC CO-TRIMOXAZOLE FOR ORAL SUSPENSION (240MG/5ML)( FORTRIM )(BATCH NO.D-060622 D-070622 D-080622 D-090622)(
300420200000
PAEDIATRIC CO-TRIMOXAZOLE FOR ORAL SUSPENSION (240MG/5ML) ( FORTRIM )(BATCH NO.D-020822 TO D-080822 D-010922 TO D-040
300420200000
PAEDIATRIC CO-TRIMOXAZOLE FOR ORAL SUSPENSION 240MG/5ML FORTRIM 10000X100ML(MLT)
PHILCO PHARMA E K

GERMANY
HS-Code
Products
300490990000
PHILCO- COTRI - 240 (PAEDIATRIC CO-TRIMOXAZOLE ORAL SUSPENSION BP 240MG/5ML ) 100 ML BT 2111063-3 MFG 01/2022 EXP 12/202
300490990000
PHILCO-COTRI-240 (PAEDIATRIC CO-TRIMOXAZOLE ORAL SUSPENSION BP 240MG/5ML)100ML:B/NO.2102015-2 2102015-3
300490990000
PHILCO-COTRI-240(PAEDIATRIC CO-TRIMOXAZOLE ORAL SUSPENSION BP 240MG/5ML)100 ML:B/NO.2102015-1 M/D 04/2021 E/D 03/2024
KARIMED PHARMACEUTICALS LTD

TRINIDAD AND TOBAGO
HS-Code
Products
300420200000
PAEDIATRIC CO - TRIMOXAZOLE ORAL SUSPENSION (240MG/5ML) (FORTRIM) (BATCH NO. BEL-230601 BEL-231001)(8000 X 100ML )
300420200000
FORTRIM / PAEDIATRIC CO-TRIMOXAZOLE FOR
300420200000
PAEDIATRIC CO - TRIMOXAZOLE ORAL SUSPENSION (240MG/5ML) (FORTRIM) (BATCH NO. BEL-230601 BEL-231001)(8000 X 100ML )
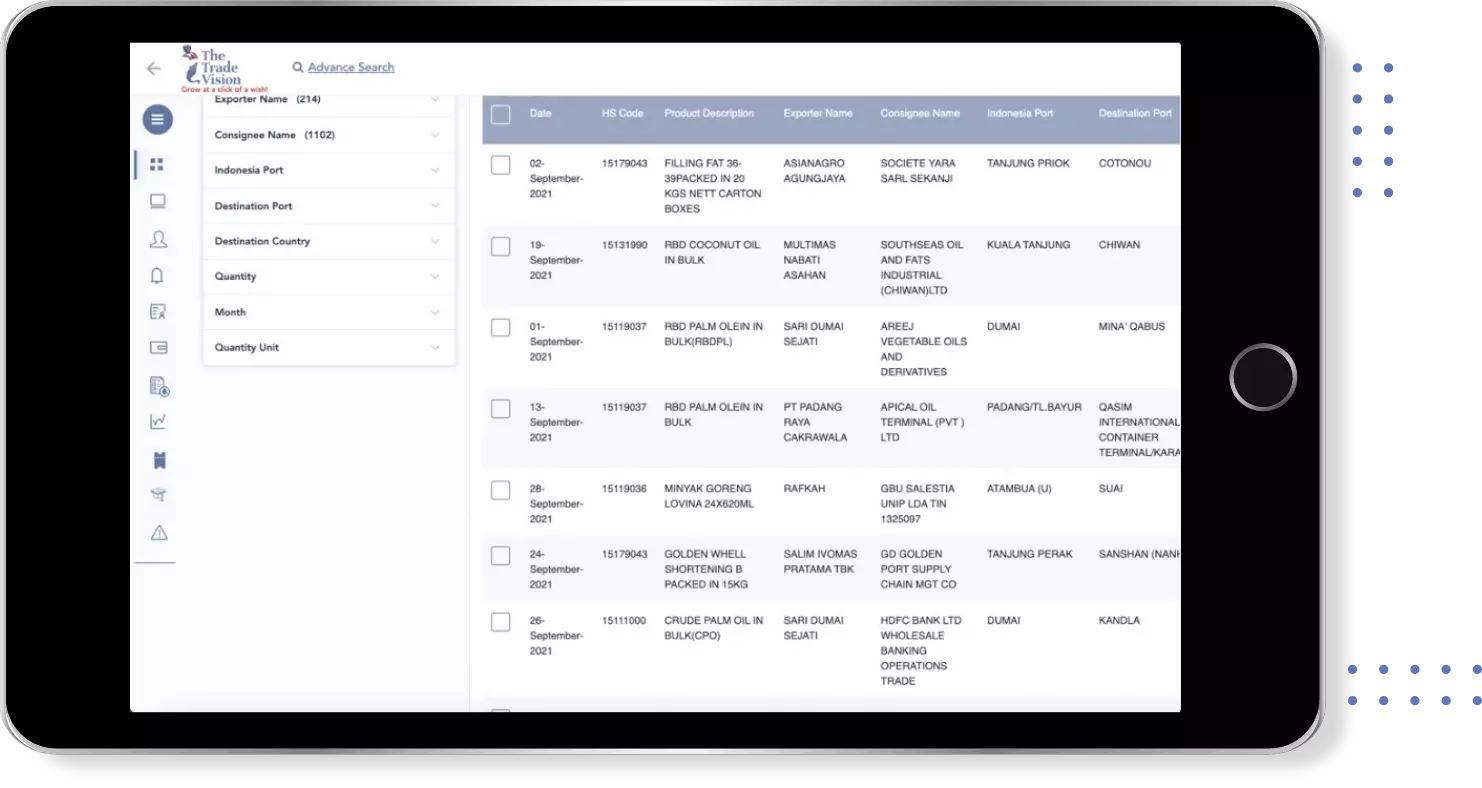
Global Export Import Trade Data for paediatric co trimoxazole
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UNITED STATES OF AMERICA UNITED STATES OF AMERICA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 PAKISTAN PAKISTAN | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 PAPUA NEW GUINEA PAPUA NEW GUINEA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 NETHERLANDS NETHERLANDS | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UNITED STATES OF AMERICA UNITED STATES OF AMERICA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
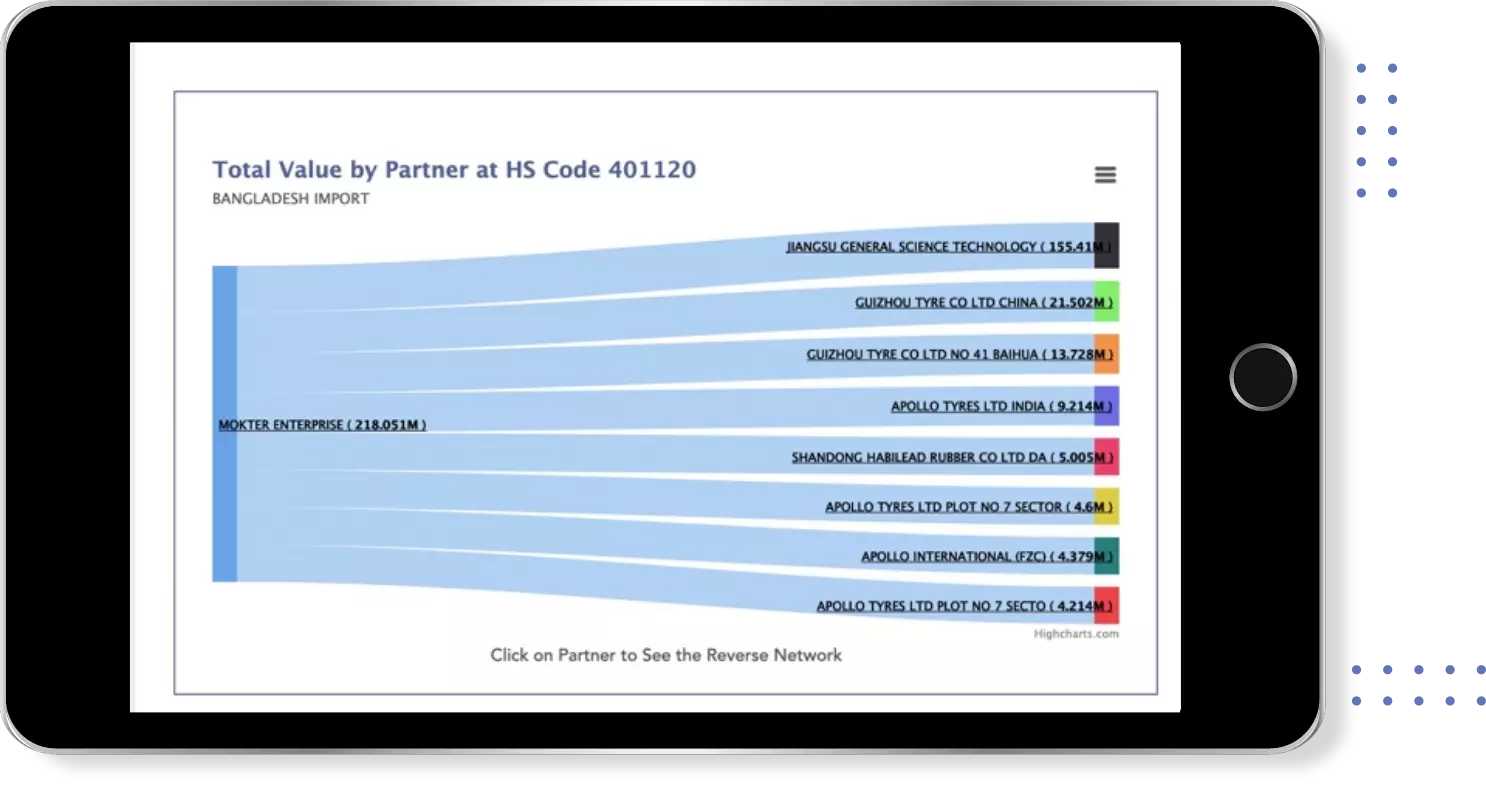
Get most profitable Buyers & Suppliers
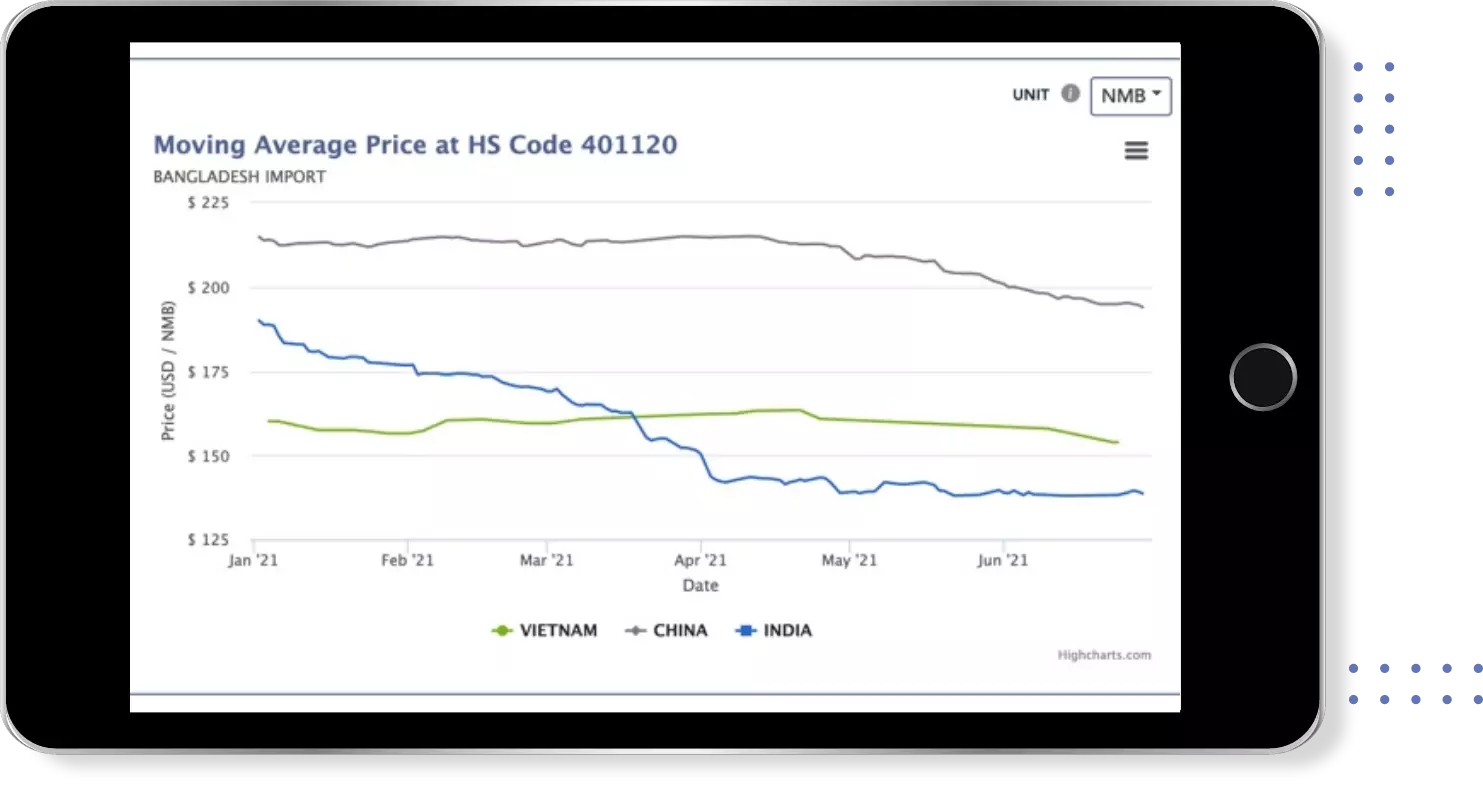
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries