Find verified buyers and sellers of o diethyl in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- o diethyl hs code 300490 buyers in ukraine
- HS Code: 300490
- COD Ukraine
Ukraine Buyers of o diethyl under HS-Code 300490
GLEDPHARM LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR THERAPEUTIC USE IN FINISHED DOSAGE FORMS NOT CONTAINING VITAMINS AND ALCOHOL PACKAGED FOR RETAIL TRADE: MONTULAR ® FILM-COATED TABLETS 10 MG (ACTIVE SUBSTANCE - 1 TABLET CONTAINS: MONTELUKAST SODIUM IN TERMS OF 10 TABLETS MONTELUKA IN A BLISTER 3 BLISTERS IN A CARDBOARD PACKAGE WITH LABELING IN UKRAINIAN AND ENGLISH - 16240 PACK. SERIES: 2002740. NEURODAR ® COATED TABLETS 500 MG (ACTIVE SUBSTANCE - 1 TABLET CONTAINS: CITICOLINE SODIUM EQUIVALENT TO CITICOLINE 10 MG) BLISTERS; 1 BLISTER IN A CARDBOARD PACKAGE; 3 PACKAGES IN A CARDBOARD BOX WITH MARKINGS IN UKRAINIAN AND ENGLISH - 9808UP. SERIES: 2002865. QUANIL COATED TABLETS 500 MG (ACTIVE SUBSTANCE - 1 TABLET CONTAINS: CITICOLIN SODIUM EQUILIVA 500 ) 10 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD PACKAGE; 3 PACKAGES IN A CARDBOARD BOX MARKED IN UKRAINIAN AND ENGLISH - 9884UP. SERIES: 2002905 .VERTINEX ® TABLETS OF 5 MG (ACTIVE INGREDIENT - 1 TABLET CONTAINS: PROCHLORPERAZINE MALEATE 5 MG) 10 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD PACKAGE; 10 CARDBOARD PACKAGES IN A CARDBOARD BOX. MARKING IN UKRAINIAN AND ENGLISH - 9754 2002862.DICLOSEF ® FORTE EMULSION GEL FOR EXTERNAL USE 2.32% (ACTIVE INGREDIENTS: 1 G OF GEL CONTAINS 23.2 MG OF DICLOFENAC DIETHYLAMINE IN TERMS OF DICLOFENAC SODIUM 20 MG) 30 G IN A TUBE 1 TUBE IN A CARDBOARD PACKAGE LABELED IN UKRAINIAN AND IN ENGLISH - 19200UP. SERIES: 2002993.DICLOSEF ® FORTE EMULSION GEL FOR EXTERNAL USE 2.32% (ACTIVE INGREDIENTS: 1 G OF GEL CONTAINS 23.2 MG OF DICLOFENAC DIETHYLAMINE IN TERMS OF DICLOFENAC SODIUM 20 MG) PER 100 G IN A TUBE; 1 TUBE IN A CARDBOARD PACKAGE WITH MARKING IN UKRAINIAN AND ENGLISH - 11300 PACK. SERIES: 2003012 2003013. TRADEMARK: KUSUM MONTULAR NEURODAR VERTINEX DICLOSEF. MANUFACTURER: KUSUM HEALTHKDV.PV.
3004900000
1. MEDICINES FOR THERAPEUTIC USE IN FINISHED DOSAGE FORMS NOT CONTAINING VITAMINS AND ALCOHOL PACKAGED FOR RETAIL TRADE: FANIGAN ® FAST GEL (ACTIVE INGREDIENTS - 1 G OF GEL CONTAINS: DICLOFENAC DIETHYLAMINE EQUIVALENT TO DICLOFENAC SODIUM 10 MG MENTHOL 50 MG MENTHOL 50 MG) 100 G IN AN ALUMINUM OR LAMINATED TUBE 1 TUBE IN A CARDBOARD PACKAGE WITH LABELING IN UKRAINIAN AND ENGLISH - 23200 UNITARY ENTERPRISE. SERIES: 2002651 2002652. NIMID ® GEL FOR EXTERNAL USE 10 MG / G (ACTIVE SUBSTANCE - 1 G OF GEL CONTAINS: NIMESULIDE 10 MG ) 30 G IN AN ALUMINUM OR LAMINATED TUBE 1 TUBE IN A CARDBOARD PACKAGE MARKED IN UKRAINIAN AND ENGLISH - 77280UP. SERIES: 2002568 2002569. SERVONEX ® COATED TABLETS 10 MG (ACTIVE SUBSTANCE - 1 TABLET CONTAINS: DONELOL 10 MG) 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD PACKAGE. MARKING IN UKRAINIAN AND ENGLISH - 3328 PACK. SERIES: 2002526. LOGUFEN ® ??FILM-COATED TABLETS 250 MG (D AND
3004900000
1. MEDICINES FOR THERAPEUTIC USE IN FINISHED DOSAGE FORMS NOT CONTAINING VITAMINS AND ALCOHOL PACKAGED FOR RETAIL TRADE: ZONIC HARD CAPSULES OF 150 MG (1 CAPSULE CONTAINS: 150 MG OF PREGABALIN) 14 CAPSULES IN A BLISTER; 2 BLISTERS IN A CARDBOARD PACKAGE WITH LABELING IN ENGLISH - 2940UP.SERIES: 2003540 DICLOSEF ® FORTE EMULSION GEL FOR EXTERNAL USE 2.32% (ACTIVE INGREDIENTS: 1 G OF GEL CONTAINS 23.2 MG OF DICLOFENAC DIETHYLAMINE IN TERMS OF DICLOFENAC SODIUM 20 MG) 30 G IN A TUBE 1 TUBE IN A CARDBOARD PACKAGE WITH MARKING IN UKRAINIAN AND ENGLISH - 18880 PACK. SERIES: 2003735. TRADEMARK: KUSUM DICLOSEF. MANUFACTURER: KUSUM HEALTHKKER PVT. LTD. INDIA.
FDI LLC GLAKSOSMITKLINE HELSKER UKRAINE TOV

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR HUMANS FOR THERAPEUTIC OR PROPHYLACTIC USE PACKAGED FOR RETAIL SALE: - COMMERCIAL NAME: VOLTAREN EMULGEL EMULGEL FOR EXTERNAL USE 1% - 24156UP. DOSAGE FORM: EMULGEL FOR EXTERNAL USE 1% ACTIVE SUBSTANCES: 1 G OF EMULGEL CONTAINS DIETHYLAMINE 11.6 MG WHICH CORRESPONDS TO 10 MG OF DICLOFENAC SODIUM PACKAGING: 100 G. 1 TUBE IN A CARDBOARD BOX MARKED IN UKRAINIAN. ART.531978 SERIES: 9T9A - 24156 UNITARY ENTERPRISE. SHELF LIFE 31.01.2024IT DOES NOT CONTAIN OZONE-DEPLETING SUBSTANCES AND FLUORINATED GREENHOUSE GASES. (NOT IN AEROSOL PACKAGING) TRADEMARK VOLTARENMANUFACTURER: GSK CONSUMER HEALTHCARE SACOUNTRY OF PRODUCTION CH.
3004900000
1. MEDICINES FOR HUMANS FOR THERAPEUTIC OR PROPHYLACTIC USE PACKAGED FOR RETAIL SALE: COMMERCIAL NAME: VOLTAREN FORTE EMULGEL FOR EXTERNAL USE 2.32% PER 100 G. IN TUBES ?1 - 48528UP. DOSAGE FORM: EMULGEL FOR EXTERNAL USE 2.32% ACTIVE SUBSTANCES: 1 G OF EMULGEL CONTAINS: DICLOFENAC DIETHYLAMINE 23.2 MG WHICH CORRESPONDS TO 20 MG OF DICLOFENAC SODIUM PACKAGING: 100 G IN A TUBE 1 TUBE IN A CARDBOARD BOX WITH THE MARK LANGUAGE. ART.60000000124495SERIES: CS5V - 24300 UNITARY ENTERPRISE. EXPIRATION DATE 29.02.2024 SERIES: CS6A - 24228 UNITARY ENTERPRISE. SHELF LIFE 29.02.2024IT DOES NOT CONTAIN OZONE-DEPLETING SUBSTANCES AND FLUORINATED GREENHOUSE GASES. (NOT IN AEROSOL PACKAGING) TRADEMARK VOLTARENMANUFACTURER: GSK CONSUMER HEALTHCARE SACOUNTRY OF PRODUCTION CH
3004900000
1. MEDICINES FOR HUMANS FOR THERAPEUTIC OR PROPHYLACTIC USE PACKAGED FOR RETAIL SALE: - COMMERCIAL NAME: VOLTAREN FORTE EMULGEL FOR EXTERNAL USE 2.32% OF 50 G. IN TUBES ?1 - 47712UP. DOSAGE FORM: EMULGEL FOR EXTERNAL USE 2.32% ACTIVE SUBSTANCES: 1 G OF EMULGEL CONTAINS: DICLOFENAC DIETHYLAMINE 23.2 MG WHICH CORRESPONDS TO 20 MG OF DICLOFENAC SODIUM PACKAGING: 50 G. 1 TUBE IN A CARDBOARD BOX MARKED IN UKRAINIAN. ART.530400 SERIES: AL4L- 47712UP. EXPIRATION DATE 30.09.2023VOLTAREN TRADEMARKMANUFACTURER: GSK CONSUMER HEALTHCARE SACOUNTRY OF PRODUCTION CH.
ORGANOSIN LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: FLAMIDES GEL GEL 40 G IN A LAMINATED TUBE; 1 TUBE IN A CARDBOARD BOX LABELED IN UKRAINIAN (ACTIVE INGREDIENTS: 1 G OF GEL CONTAINS: DICLOFENAC DIETHYLAMINE 11.6 MG (EQUIVALENT TO DICLOFENAC SODIUM 10 MG) METHYL SALICYLATE 100 MG LEVOMENTHOL 50 MG). SERIES: FG008 FG009 FG010 FG011 FG012 FG013 FG014 - 166800UP. CLOTRIMAZOLE OINTMENT 1% ON 20 G IN A TUBE; 1 TUBE IN A CARDBOARD PACK LABELED IN UKRAINIAN (ACTIVE INGREDIENTS: 1 G OF OINTMENT CONTAINS: CLOTRIMAZOLE 10 MG). SERIES: CO003 CO004 - 54000UP.MANUFACTURER: ENCUBE ETHICALS PRIVATE LIMITED INDIA.COUNTRY OF PRODUCTION: IN
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: FLAMIDES GEL GEL 100 G IN A LAMINATED TUBE; 1 TUBE IN A CARDBOARD BOX LABELED IN UKRAINIAN (ACTIVE INGREDIENTS: 1 G OF GEL CONTAINS: DICLOFENAC DIETHYLAMINE 11.6 MG (EQUIVALENT TO DICLOFENAC SODIUM 10 MG) METHYL SALICYLATE 100 MG LEVOMENTHOL 50 MG). SERIES: FG027 FG028 .FLAMIDES GEL GEL 40 G IN A LAMINATED TUBE; 1 TUBE IN A CARDBOARD BOX LABELED IN UKRAINIAN (ACTIVE INGREDIENTS: 1 G OF GEL CONTAINS: DICLOFENAC DIETHYLAMINE 11.6 MG (EQUIVALENT TO DICLOFENAC SODIUM 10 MG) METHYL SALICYLATE 100 MG LEVOMENTHOL 50 MG). SERIES: FG020 FG021 FG022 - 67120UP. FLAMIDES PHYTOPLUS OINTMENT 20 G IN A TUBE; 1 TUBE IN A CARDBOARD PACK LABELED IN UKRAINIAN (ACTIVE INGREDIENTS: 1 G OF OINTMENT CONTAINS: LEVOMENTHOL 50 MG THYMOL 10 MG METHYL SALICYLATE 50 MG EUCALYPTUS OIL (EUCALYPTUS OIL) 10 MG TURPENTINE OIL (TURPENTINEAL) 30 MG). SERIES: FP002 - 45340UP.MANUFACTURER: ENCUBE ETHICALS
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: FLAMIDES GEL GEL 100 G IN A LAMINATED TUBE; 1 TUBE IN A CARDBOARD BOX LABELED IN UKRAINIAN (ACTIVE SUBSTANCES: 1 G OF GEL CONTAINS: DICLOFENAC DIETHYLAMINE 11 6MG (EQUIVALENT TO DICLOFENAC SODIUM 10MG) METHYL SALICYLATE 100MG LEVOMENTHOL 50MG). SERIES: FG036 FG037 FG038 - 216UP. 1 G OF GEL CONTAINS: DICLOFENAC DIETHYLAMINE 11.6 MG (EQUIVALENT TO DICLOFENAC SODIUM 10 MG) METHYLSALICYLATE 100 MG LEVOMENTHOL 50 MG) SERIES: FG032 FG033 FG034 FG035 FG039 FG040 EPYTEI: 8 INDIA. COUNTRY OF MANUFACTURE: IN
GLEDPHARM LTD 02092 M KYIV VUL ALMA ATYNSKA 58

UKRAINE
HS-Code
Products
3004900000
MEDICINES FOR THERAPEUTIC USE IN FINISHED DOSAGE FORM DOES NOT CONTAIN VITAMINS AND ALCOHOL PUT UP FOR RETAIL SALE DERMAZOL R CREAM 20 MG G ACTIVE SUBSTANCE 1 G OF CREAM CONTAINS KETOCONAZOLE 20 MG PO 30G IN A TUBE LABELED UKRAYINSKOYUMOVOYU IN 1 TUBE IN A CARTON BOX MARKED UKRAINIAN AND ENGLISH 384UP 9 200 041 SERIES R DIKLOSEYF SUPPOSITORIES IN 1 00MH ACTIVE SUBSTANCE 1 SUPPOSITORY CONTAINS DICLOFENAC SODIUM 1 00MH IN 5SUPOZYTORIYIV IN A STRIP MARKED UKRAINIAN 2 STRIP MARKED UKRAINIAN AND ENGLISH AMI 3584UP SERIES 2000353 2000374 2000375 2000377 2000382 2000389 2000392 2000421 2000423 FANIGAN R FAST GEL ACTIVE SUBSTANCES 1 G GEL CONTAINS DICLOFENAC DIETHYLAMINE EQUENEQUENE
3004900000
MEDICINAL PRODUCTS FOR THERAPEUTIC USE IN FINISHED DOSAGE FORMS PACKAGED FOR RETAIL SALE DERMAZOL VAGINAL SUPPOSITORIES 400MG 10 5X2 ACTIVE INGREDIENTS 1 SUPPOSITORY CONTAINS KETOCONAZOLE 400MG 5 SUPPOSITORIES IN A STRIP WITH LABELING IN UKRAINIAN 2 STRIPS IN A CARDBOARD PACKAGE8 IN ENGLISH5 WITH BRAND 2000503 FANIGAN R FAST GEL ACTIVE INGREDIENTS 1 G OF GEL CONTAINS DICLOFENAC DIETHYLAMINE EQUIVALENT TO DICLOFENAC SODIUM 10 MG OF MENTHOL 50 MG OF METHYL SALICYLATE 100 MG OF LINSEED OIL 30 MG OF 100 G IN ALUMINUM OR LAMINATED TUBE OF 1 TUBE IN ENGLISH AND CARDBOARD PACKAGING SUBSTANCE 1 G OF GEL CONTAINS DICLOFENAC DIETHYLAMINE EQUIVALENT TO DICLOFENAC SODIUM 10 MG OF MENTHOL 50 MG OF METHYL SALICYLATE 100 MG OF LINSEED OIL 30 MG OF 30 G IN AN ALUMINUM OR LAMINATED TUBE 1 TUBE IN A CARDBOARD PACKAGE MARKED IN UKRAINIAN AND ENGLISH 320 JULY 2012 FLAKY COATED ACTIVE INGREDIENT 1 TABLET CONTAINS LOPERAMIDE HYDROCHLORIDE 2 MG SIMETHICONE 125 MG 10 TABLETS IN A BLISTER 10 BLISTERS IN A CARDBOARD BOX MARKED IN UKRAINIAN AND ENGLISH 144UP SERIES 2000426 SERRATA R COATED TABLETS SERVO TABLETS IN A STRIP OF 5 STRIPS IN A CARDBOARD PACKAGE MARKING IN UKRAINIAN AND ENGLISH 224UP SERIES 20003
3004900000
MEDICINAL PRODUCTS FOR THERAPEUTIC USE IN FINISHED DOSAGE FORMS PACKAGED FOR RETAIL SALE DERMAZOL VAGINAL SUPPOSITORIES 400MG 10 5X2 ACTIVE INGREDIENTS 1 SUPPOSITORY CONTAINS KETOCONAZOLE 400MG 5 SUPPOSITORIES IN A STRIP WITH LABELING IN UKRAINIAN 2 STRIPS IN A CARDBOARD PACKAGE 109 IN ENGLISH5 WITH BRAND 2000503 FANIGAN R FAST GEL ACTIVE INGREDIENTS 1G OF GEL CONTAINS DICLOFENAC DIETHYLAMINE EQUIVALENT TO DICLOFENAC SODIUM 10MG MENTHOL 50MG METHYL SALICYLATE 100MG LINSEED OIL 30MG 100G IN ALUMINUM OR LAMINATED TUBE 1 TUBE IN ENGLISH AND CARDBOARD PACKAGING SUBSTANCE 1 G OF GEL CONTAINS DICLOFENAC DIETHYLAMINE EQUIVALENT TO DICLOFENAC SODIUM 10 MG OF MENTHOL 50 MG OF METHYL SALICYLATE 100 MG OF LINSEED OIL 30 MG OF 30 G IN AN ALUMINUM OR LAMINATED TUBE 1 TUBE IN A CARDBOARD PACKAGE MARKED IN UKRAINIAN AND ENGLISH SERBO R FILM-COATED TABLETS ACTIVE SUBSTANCE 1 TABLET CONTAINS LOPERAMIDE HYDROCHLORIDE 2 MG SIMETHICONE 125 MG 10 TABLETS IN A BLISTER 10 BLISTERS IN A CARDBOARD BOX LABELED IN UKRAINIAN AND ENGLISH 4087UP SERIES 2000426 SERRATA R TABLETS FILM-COATED 30 TABLETS IN A STRIP 5 STRIPS IN A CARDBOARD PACKAGE MARKING IN UKRAINIAN AND ENGLISH 20780U
BADM LTD LIABILITY COMPANY

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE WITHOUT CONTENT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: TIOKTATSID ® 600 HR COATED TABLETS OF 600 MG 30 TABLETS PER VIAL 1 VIAL PER BOX CARDBOARD / 1 FILM-COATED TABLET CONTAINING 600 MG THIOCTIC (ALPHA-LIPOIC) ACID MANUFACTURED BY MEDA MANUFACTURING GMBH (MANUFACTURER RESPONSIBLE FOR THE PRODUCTION OF BULK PRODUCTS PRIMARY AND SECONDARY PACKAGING QUALITY CONTROL) GERMANY / MEDA PHARMA GMBH & CO. KG (MANUFACTURER RESPONSIBLE FOR THE RELEASE OF THE SERIES) GERMANY REPARIL®-GEL H GEL 40 G OF GEL IN AN ALUMINUM OR LAMINATE TUBE; 1 TUBE IN A CARDBOARD BOX / 10 G OF GEL CONTAINS ESCIN 100 MG (0.1 G) AND DIETHYLAMINE SALICYLATE 500 MG (0.5 G) MANUFACTURER MADAUS GMBH GERMANY;
3004900000
1. MEDICINES FOR PEOPLE WITHOUT CONTENT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: REPARIL®-GEL H GEL 40 G OF GEL IN AN ALUMINUM OR LAMINATE TUBE; 1 TUBE IN A CARDBOARD BOX / 10 G OF GEL CONTAINS ESCIN 100 MG (0.1 G) AND DIETHYLAMINE SALICYLATE 500 MG (0.5 G) / MANUFACTURER MADAUS GMBH GERMANY;
3004900000
1. MEDICINES FOR PEOPLE WITHOUT CONTENT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: TIOKTATSID ® 600 HR COATED TABLETS OF 600 MG 30 TABLETS PER VIAL 1 VIAL PER BOX CARDBOARD / 1 FILM-COATED TABLET CONTAINS 600 MG OF THIOCTIC (ALPHA-LIPOIC) ACID MANUFACTURER MEDA MANUFACTURING GMBH GERMANY / MEDA PHARMA GMBH & CO. KG (MANUFACTURER RESPONSIBLE FOR THE RELEASE OF THE SERIES) GERMANY REPARIL®-GEL H GEL 40 G OF GEL IN AN ALUMINUM OR LAMINATE TUBE; 1 TUBE IN A CARDBOARD BOX / 10 G OF GEL CONTAINS ESCIN 100 MG (0.1 G) AND DIETHYLAMINE SALICYLATE 500 MG (0.5 G); MANUFACTURER MADAUS GMBH GERMANY;
JV OPTIMA PHARM LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE. SERIES 3807691: ALERGODIL® NASAL SPRAY DOSED 1 MG / ML ON 10 ML IN A BOTTLE WITH THE SCREWED SPRAY; ON 1 BOTTLE IN A CARDBOARD PACKING - 160 UNITARY ENTERPRISE (ACTIVE INGREDIENT - AZEROLASTY ) MANUFACTURER: MANUFACTURER RESPONSIBLE FOR THE PRODUCTION OF BULK PRODUCTS PRIMARY AND SECONDARY PACKAGING QUALITY CONTROL: MEDA MANUFACTURING GMBH DE MANUFACTURER RESPONSIBLE FOR THE PRODUCTION OF THE SERIES: MEDA PHARMA GMBH AND CO.KG DE. SERIES D1801866: REPARIL®-GEL GEL ON 40 G OF GEL IN AN ALUMINUM TUBE; ON 1 TUBE IN A CARDBOARD BOX - 576 UNITARY ENTERPRISE. SERIES D1900685: REPARIL®-GEL N GEL ON 40 G OF GEL IN AN ALUMINUM TUBE; ON 1 TUBE IN A CARDBOARD BOX - 192 UNITARY ENTERPRISE. SUBSTANCE - DIETHYLAMINE SALICYLATE ESCIN) MANUFACTURER: MADAUS GMBH DE SERIES 3839411: TIOCTACID® 600 HR COATED TABLETS 600 MG 100 TABLETS IN A VIAL 1 VIAL IN A CARDBOARD BOX - 80 SUBSTANCES - ACTION. ) MANUFACTURER: MEDA MENYUFEKCHERING GMBH DE THE MANUFACTURER IS RESPONSIBLE FOR
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE. SERIES 3807691: ALERGODIL® NASAL SPRAY DOSED 1 MG / ML ON 10 ML IN A BOTTLE WITH THE SCREWED SPRAY; ON 1 BOTTLE IN A CARDBOARD PACKING - 160 UNITARY ENTERPRISE (ACTIVE INGREDIENT - AZEROLASTY ) MANUFACTURER: MANUFACTURER RESPONSIBLE FOR THE PRODUCTION OF BULK PRODUCTS PRIMARY AND SECONDARY PACKAGING QUALITY CONTROL: MEDA MANUFACTURING GMBH DE MANUFACTURER RESPONSIBLE FOR THE RELEASE OF THE SERIES: MEDA PHARMA GMBH AND CO.KG DE. SERIES D1801866: REPARIL®-GEL GEL 40 G OF GEL IN AN ALUMINUM TUBE 1 TUBE IN A CARDBOARD BOX -480 PACK (ACTIVE INGREDIENT - DIETHYLAMINE SALICYLATE ESCIN) MANUFACTURER: MADAUS GMBH DE. SERIES 3836931: TIOKTATSID® 600 HR TABLETS COATED 30 TABLETS IN A BOTTLE 1 BOTTLE IN A CARDBOARD BOX -240 PACK (ACTIVE SUBSTANCE - THIOCTIC ACID) MANUFACTURER: MEDA MANUFACTURING GMBH DE; THE MANUFACTURER IS RESPONSIBLE FOR THE RELEASE OF THE SERIES: MEDA PHARMA GMBH AND CO. KG DE.
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL. SERIES D1 900685: REPARIL®-GEL H GEL OF 40 G GEL IN ALUMINUM TUBE; ON 1 TUBE IN A CARDBOARD BOX - 3024 UP. (ACTIVE SUBSTANCE - DIETHYLAMINE SALICYLATE ESCIN) MANUFACTURER: MADAUS GMBH DE. SERIES 3839421: THIOCTACID® 600 HR COATED TABLETS 600 MG 1 00 TABLETS IN A VIAL; 1 VIAL IN A CARTON BOX - 400 UE. (ACTIVE SUBSTANCE - THIOCTIC ACID) SERIES 3841 361: THIOCTACID® 600 HR COATED TABLETS 600 MG 30 TABLETS IN A VIAL; 1 BOTTLE IN A CARTON BOX - 1 050 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - THIOCTIC ACID) MANUFACTURER: MEDA MANUFACTURING GMBH DE MANUFACTURER RESPONSIBLE FOR SERIES: MEDA PHARMA GMBH. KG DE. 8E302A SERIES: THIOCTACID® 600T SOLUTIONS FOR INJECTION (600 MG / 24 ML) 24 ML IN AMPOULE; 5 AMPOULES IN CONTOUR CELL PACKAGING; ON 1 CONTOUR CELLULAR PACKAGING IN A PACK - 1 200 UP. (ACTIVE SUBSTANCE - THIOCTIC ACID) MANUFACTURER: ALL PRODUCTION STAGES EXCEPT OF SERIES: SIEGFRIED HAMELN GM
ASINO UKRAINE LLC

UKRAINE
HS-Code
Products
3004900000
1. MEDICINAL PRODUCT FOR HUMANS. - MIORIX® PROLONGED-RELEASE CAPSULES SOLID 15MG ?14 IN A BLISTER (MYORIX® 15MG PROLONGED-RELEASE CAPSULES HARD 15MG ?14 IN BLISTER) IN A CARDBOARD BOX MARKED IN UKRAINIAN AND ENGLISH -60510UP .SERIES 475740-25775UP.PRODUCTION DATE 12.2019 EXPIRATION DATE 11.2023R.SERIES 475371-22525UP.SERIES 475738-12210UP.PRODUCTION DATE 02.2020R EXPIRATION DATE 01.2024.ACTIVE SUBSTANCES: 1 CAPSULE CONTAINS: 15 MG OF CYCLONE SUBSTANCES: SPHERICAL SUGAR OPADRY CLEAR YS-1-7006 ETHYLCELLULOSE DIETHYL PHTHALATE CAPSULE SHELL: IRON OXIDE RED (E 172) IRON OXIDE YELLOW (E 172) TITANIUM DIOXIDE (E 171) GELATIN BLUE INK - TEKPRINTTM SB 6018WITHOUT NARCOTIC DRUGS PSYCHOTROPIC SUBSTANCES AND PRECURSORS.MANUFACTURER: TAKEDA PHARMA SP.Z OO POLAND12 KSIESTWA LOWICKIEGO STR. 99-420 LYSZKOWICE POLANDADARE PHARMACEUTICALS INC. USA845 CENTER DRIVE VANDALIA OH 45377. COUNTRY OF MANUFACTURE: US
3004900000
1. MEDICINAL PRODUCT FOR HUMANS. - MIORIX® CAPSULES OF PROLONGED ACTION HARD ON 15 MG ?14 IN A BLISTER (MYORIX® 15 MG PROLONGED-RELEASE CAPSULES HARD 15 MG ? 14 IN BLISTER) IN A CARDBOARD BOX WITH MARKING IN UKRAINIAN AND ENGLISH -19310UP .SERIES 470334-7790UP.PRODUCTION DATE 12.2019 EXPIRATION DATE 11.2023.SERIES 470434-11520UP.PRODUCTION DATE 02.2020R EXPIRATION DATE 01.2024. ACTIVE SUBSTANCES: 1 CAPSULE CONTAINS: 15 MG OF CYCLOBENZAPRINE HYDROCHLORIDE. OPADRY CLEAR YS-1-7006 ETHYLCELLULOSE DIETHYL PHTHALATE CAPSULE SHELL: IRON OXIDE RED (E 172) IRON OXIDE YELLOW (E 172) TITANIUM DIOXIDE (E 171) GELATIN BLUE INK TEKPRINTTM SB-6018- MIORIX PROLONGED-RELEASE SOLID 30MG ?14 IN BLISTER (MYORIX® 30MG PROLONGED-RELEASE CAPSULES HARD 30MG ?14 IN BLISTER) IN A CARDBOARD BOX MARKED IN UKRAINIAN AND ENGLISH -20800UP. SERIES 472751-20800UP. DATE OF PRODUCTION 02.2020. EXPIRATION DATE 01.2024. ACTIVE SUBSTANCES N
3004900000
1. A DRUG FOR HUMANS. - MIORIX® CAPSULES OF PROLONGED ACTION FIRM ON 15 MG ?14 IN A BLISTER (MYORIX® 15MG PROLONGED-RELEASE CAPSULES HARD 15MG ?14 IN BLISTER) IN A CARDBOARD BOX WITH MARKING IN UKRAINIAN AND ENGLISH -47815UP. SERIES 458291-26685UP.DATE OF PRODUCTION 04.2019 EXPIRATION DATE 03.2023. SERIES 465170-21130 PACK. DATE OF PRODUCTION 12.2019 EXPIRATION DATE 11.2023. ACTIVE SUBSTANCES: 1 CAPSULE CONTAINS: 15 MG OF CYCLOBENZAPRINE HYDROCHLORIDE. EXCIPIENTS: SPHERICAL SUGAR OPADRY CLEAR Y6-1 ETHYLCELLULOSE DIETHYL PHTHALATE; CAPSULE SHELL: IRON OXIDE RED (E 172) IRON OXIDE YELLOW (E 172) TITANIUM DIOXIDE (E 171) GELATIN BLUE INK TEKPRINTTM SB-6018NO CONTAINING NARCOTIC PSYCHOTROPIC SUBSTANCES AND PRECURSORS.MANUFACTURER: TAKEDA PHARMA SP.Z O POLAND12 KSIESTWA LOWICKIEGO STR. 99-420 LYSZKOWICE POLANDADARE PHARMACEUTICALS INC. USA845 CENTER DRIVE VANDALIA OH 45377. USA TRADEMARK: TAKEDA COUNTRY OF MANUFACTURE: US
HFC BIOCON LLC

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: -FLAMIDE GEL GEL 100 G OF LAMINATED TUBE; 1 TUBE IN A CARDBOARD BOX LABELED IN UKRAINIAN (ACTIVE SUBSTANCES: 1 G OF GEL CONTAINS: DICLOFENAC IN DIETHYLAMINE 11 DICLOFENAC SODIUM 10 MG) METHYL SALICYLATE 100 MG LEVOMENTHOL 50 MG). - SERIES: FG118 FG119 FG120 FG121 -38736UP SUBSTANCES: 1 G OF OINTMENT CONTAINS: CLOTRIMAZOLE 10 MG). - SERIES: CL105 CL106 CL107 CL108-192360 UNITARY ENTERPRISE.
300490
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: -FLAMIDES GEL GEL 100 G OF LAMINATED TUBE; 1 TUBE IN A CARDBOARD BOX LABELED IN UKRAINIAN (ACTIVE INGREDIENTS: 1 G OF GEL CONTAINS: DICLOFENAC 6 DIETHYLAMINE DICLOFENAC SODIUM 10MG) METHYL SALICYLATE 100MG LEVOMENTHOL50MG) .- SERIES: FG036 FG037 FG038 -28872UP.-FLAMIDES GEL GEL 40G IN A LAMINATED TUBE 1 TUBE IN A CARDBOARD BOX CONTAINING UKRAINIAN BUTTER : DICLOFENACUDIETHYLAMINE 11.6 MG (EQUIVALENT TO DICLOFENAC SODIUM 10 MG) METHYLSALICYLATE 100 MG LEVOMENTHOL 50 MG) .- SERIES: FG032 FG033 FG034 FG035 FG039 FG040 -143760UP.PRODUCTOR: PRODUCER: MANUFACTURER:
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: -FLAMIDES GEL GEL 100 G OF LAMINATED TUBE; 1 TUBE IN A CARDBOARD BOX LABELED IN UKRAINIAN (ACTIVE INGREDIENTS: 1 G OF GEL CONTAINS: DICLOFENAC 6 DIETHYLAMINE DICLOFENAC SODIUM 10MG) METHYL SALICYLATE 100MG LEVOMENTHOL50MG) .- SERIES: FG042 FG043 FG044 FG045 FG046- 47808UP.MANUFACTURER: ENCUBE ETHICALS PRIVATE LIMITED INDIA: COUNTRY OF MANUFACTURE
DR REDDY S LABORATORY

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE: DOLAREN® TABLETS ?100 (10X10) - 10 TABLETS IN A BLISTER; 10 BLISTERS IN A CARDBOARD BOX (ACTIVE INGREDIENTS - 1 TABLET CONTAINS DICLOFENAC SODIUM 50 MG PARACETAMOL 500 MG) SERIES DA326 - 50 UNITS; DA327 SERIES - 50 UNITS; DA328 SERIES - 50 UNITARY ENTERPRISE; DA329 SERIES - 50 UNITARY ENTERPRISE; DA330 SERIES - 50 UNITS; SERIES DA331 - 50 UNITARY ENTERPRISE; SERIES DA332 - 50 UNITARY ENTERPRISE; DA333 SERIES - 50 UNITS; SERIES DA334 - 50 PACK. DOLAREN® GEL; 50 G IN A TUBE; 1 TUBE IN A CARDBOARD PACKAGE (ACTIVE SUBSTANCES - 1 G OF GEL CONTAINS DICLOFENAC DIETHYLAMINE IN TERMS OF DICLOFENAC SODIUM 10 MG MENTHOL 50 MG METHYL SALICYLATE 100 MG) SERIES DB041 - 160UP .; DB042 SERIES - 160 UNITS; SERIES DB043 - 160 UNITARY ENTERPRISE. TRADEMARK: DOLLAR MANUFACTURER: NABROS PHARMA PVT. LTD. COUNTRY OF MANUFACTURE: IN
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE: DOLAREN® TABLETS ?100 (10X10) - 10 TABLETS IN A BLISTER; 10 BLISTERS IN A CARDBOARD BOX (ACTIVE INGREDIENTS - 1 TABLET CONTAINS DICLOFENAC SODIUM 50 MG PARACETAMOL 500 MG) SERIES DA326 - 9800 UNITS; SERIES DA327 - 9800 UNITARY ENTERPRISE; DA328 SERIES - 9800 UNITS; SERIES DA329 - 9800 UNITARY ENTERPRISE; DA330 SERIES - 9800 UNITS; SERIES DA331 - 9800 UNITARY ENTERPRISE; SERIES DA332 - 9800 UNITARY ENTERPRISE; SERIES DA333 - 9800 UNITARY ENTERPRISE; SERIES DA334 - 9800 UNITARY ENTERPRISE. DOLAREN® GEL OF 50 G IN A TUBE; 1 TUBE IN A CARDBOARD PACKAGE (ACTIVE SUBSTANCES - 1 G OF GEL CONTAINS DICLOFENAC DIETHYLAMINE IN TERMS OF DICLOFENAC SODIUM 10 MG MENTHOL 50 MG METHYL SALICYLATE 100 MG) SERIES DB041 - 5810UP .; SERIES DB042 - 5810 UNITARY ENTERPRISE; SERIES DB043 - 5810 UNITARY ENTERPRISE. TRADEMARK: DOLLAR MANUFACTURER: NABROS PHARMA PVT. LTD. COUNTRY OF MANUFACTURE: IN
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE: DOLAREN® GEL 50 G IN A TUBE 1 TUBE IN A CARDBOARD PACKAGE (1 G OF GEL CONTAINS DICLOFENAC DIETHYLAMINE IN TERMS OF DICLOFENAC SODIUM 10 MG MENTHOL 50 MG METHYL SALICYLATE 100 MG - 160 MG) SERIES DB0. SERIES DB045 - 160UP. SERIES DB046 - 160UP. TRADEMARK: DOLLAR MANUFACTURER: NABROS PHARMA PVT. LTD. COUNTRY OF MANUFACTURE: IN
FDI LLC GLAKSOSMITKLINE HELSKER UKRAINE TO VV

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR HUMANS FOR THERAPEUTIC OR PROPHYLACTIC USE PACKAGED FOR RETAIL TRADE: - COMMERCIAL NAME: VOLTAREN FORTE EMULGEL EMULGEL FOR EXTERNAL USE 2.32% OF 50 G. IN TUBES ?1 - 78120UP. DOSAGE FORM: EMULGEL FOR EXTERNAL USE 2.32% ACTIVE SUBSTANCES: 1 G OF EMULGEL CONTAINS: DICLOFENAC DIETHYLAMINE 23.2 MG WHICH CORRESPONDS TO 20 MG OF DICLOFENAC SODIUM PACKAGING: 50 G. 1 TUBE IN A CARDBOARD BOX MARKED IN UKRAINIAN. ART.530400 SERIES: 5W5U-A - 78120UP. EXPIRATION DATE 30.06.22 TRADEMARK VOLTARENMANUFACTURER: NEW CONSUMER HEALTH SAKRAINA PRODUCTION CH.
3004900000
1. MEDICINES FOR HUMANS FOR THERAPEUTIC OR PROPHYLACTIC USE PACKAGED FOR RETAIL TRADE: - COMMERCIAL NAME: VOLTAREN FORTE EMULGEL EMULGEL FOR EXTERNAL USE 2.32% OF 50 G. IN TUBES ?1 - 19194UP. DOSAGE FORM: EMULGEL FOR EXTERNAL USE 2.32% ACTIVE SUBSTANCES: 1 G OF EMULGEL CONTAINS: DICLOFENAC DIETHYLAMINE 23.2 MG WHICH CORRESPONDS TO 20 MG OF DICLOFENAC SODIUM PACKAGING: 50 G. 1 TUBE IN A CARDBOARD BOX MARKED IN UKRAINIAN. ART.530400 SERIES: 5W5U - 19194UP. SHELF LIFE 30.06.22- COMMERCIAL NAME: VOLTAREN FORTE EMULGEL FOR EXTERNAL USE 2.32% PER 100 G. IN TUBES ?1 - 47376UP. DOSAGE FORM: EMULGEL FOR EXTERNAL USE 2.32% ACTIVE SUBSTANCES: 1 G OF EMULGEL CONTAINS: DICLOFENAC DIETHYLAMINE 23.2 MG WHICH CORRESPONDS TO 20 MG OF DICLOFENAC SODIUM PACKAGING: 100 G IN A TUBE 1 TUBE IN A CARDBOARD BOX WITH THE MARK . ART.530401 SERIES: 8C2S - 47376UP. SHELF LIFE 31.07.2022
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC OR PREVENTIVE APPLICATION PACKAGED FOR RETAIL: - COMMERCIAL NAME: FULL NAME: VO IN THE BOX ?1 - 46368UP.LIKARSKA FORM: EMULHEL FOR OUTDOOR USE 2.32% OF ACTIVE SUBSTANCE: 1G EMULHELYA CONTAINS: DICLOFENAC DIETHYLAMINE 23 2MH THAT MEETS DICLOFENAC SODIUM 20MG PACKING: ON 50 G. 1 TUBE IN A CARDBOARD BOX MARKED IN UKRAINIAN. ART.530400 SERIES: NV8S - 46368UP. EXPIRY DATE 30.09.2022 TRADEMARK VOLTAREN MANUFACTURER: INNOVATION CONSUMER HELS SACRAIN PRODUCTION CH.
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
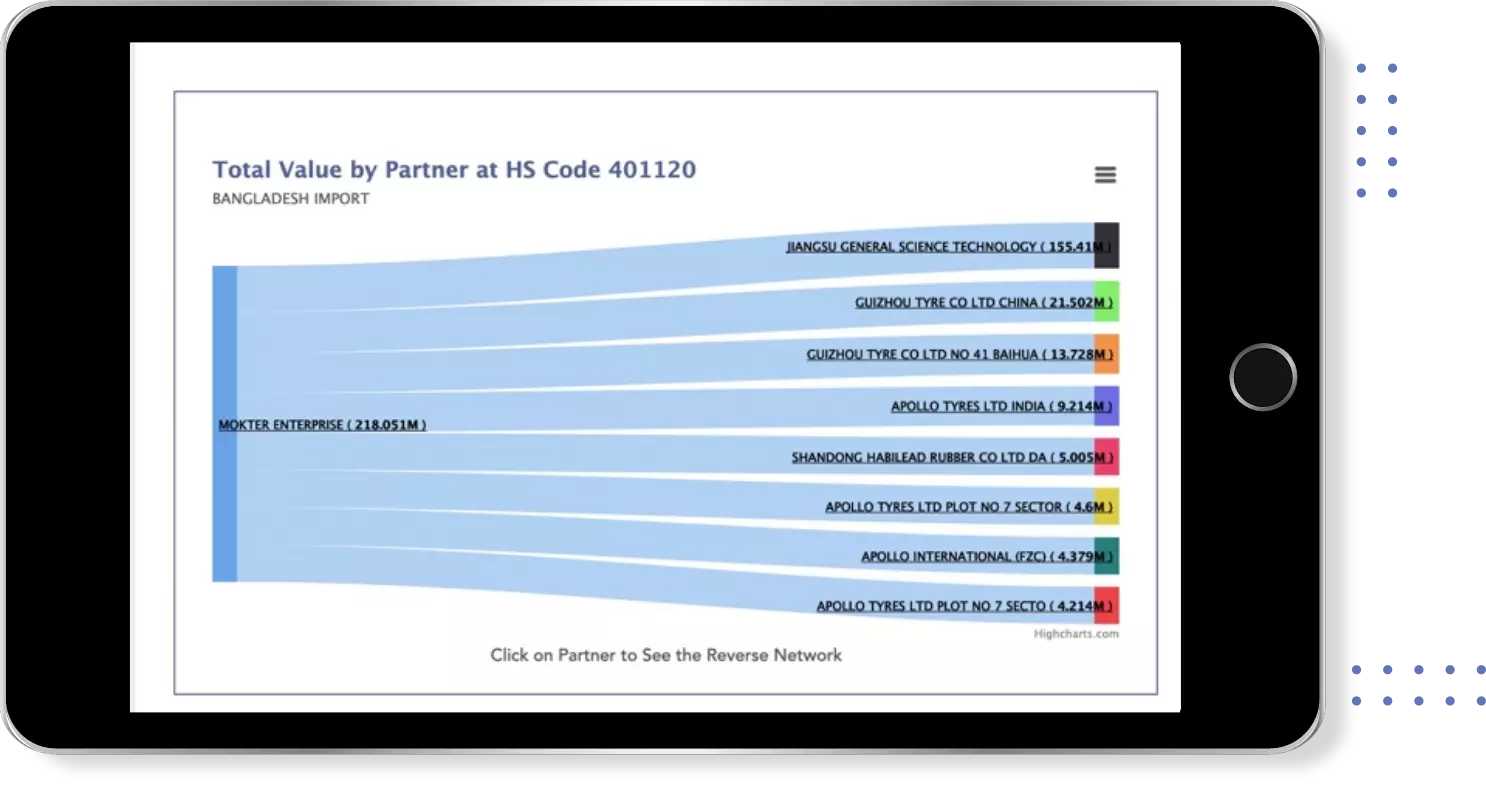
Get most profitable Buyers & Suppliers
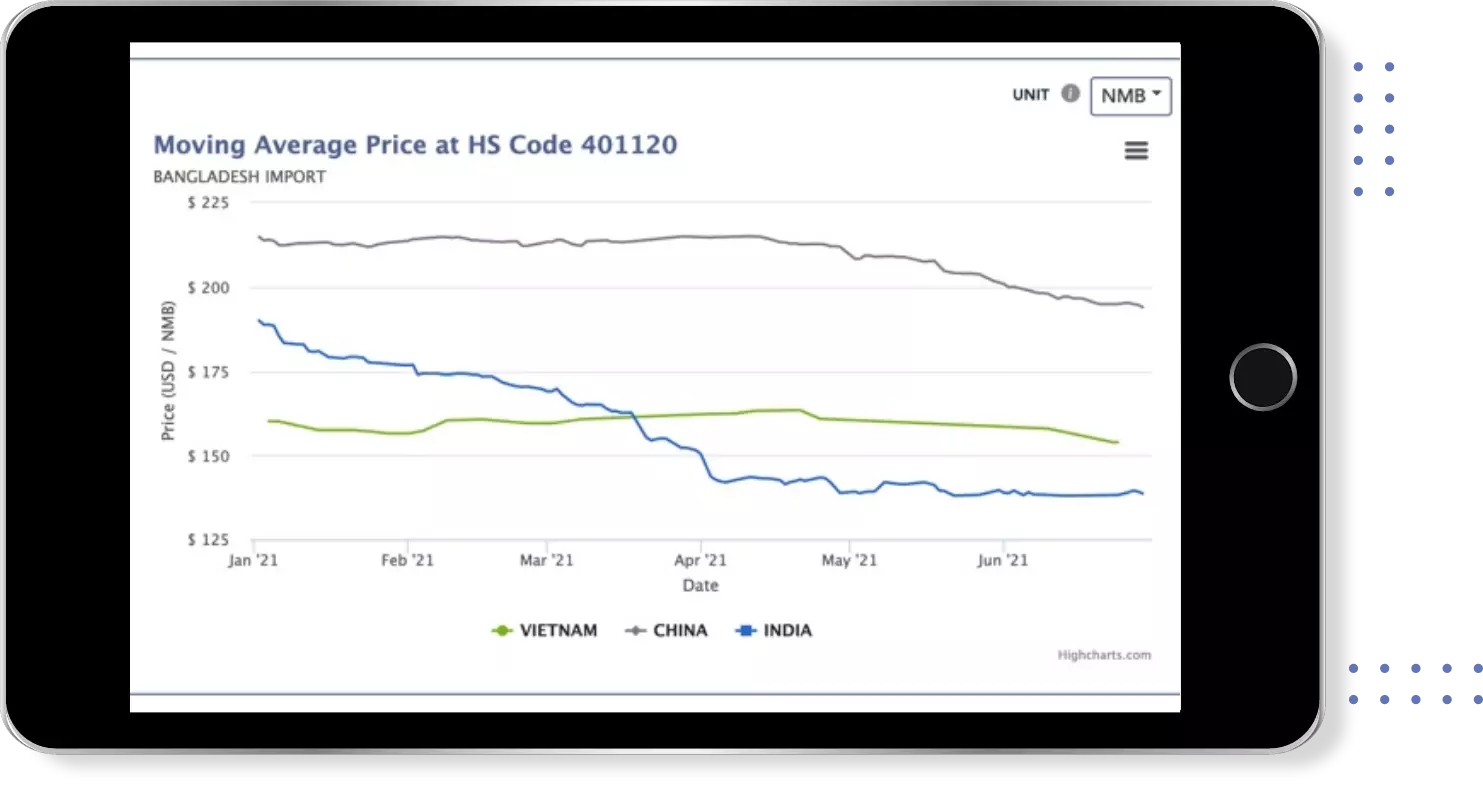
Instant insights on Price , Quantity & Value trends
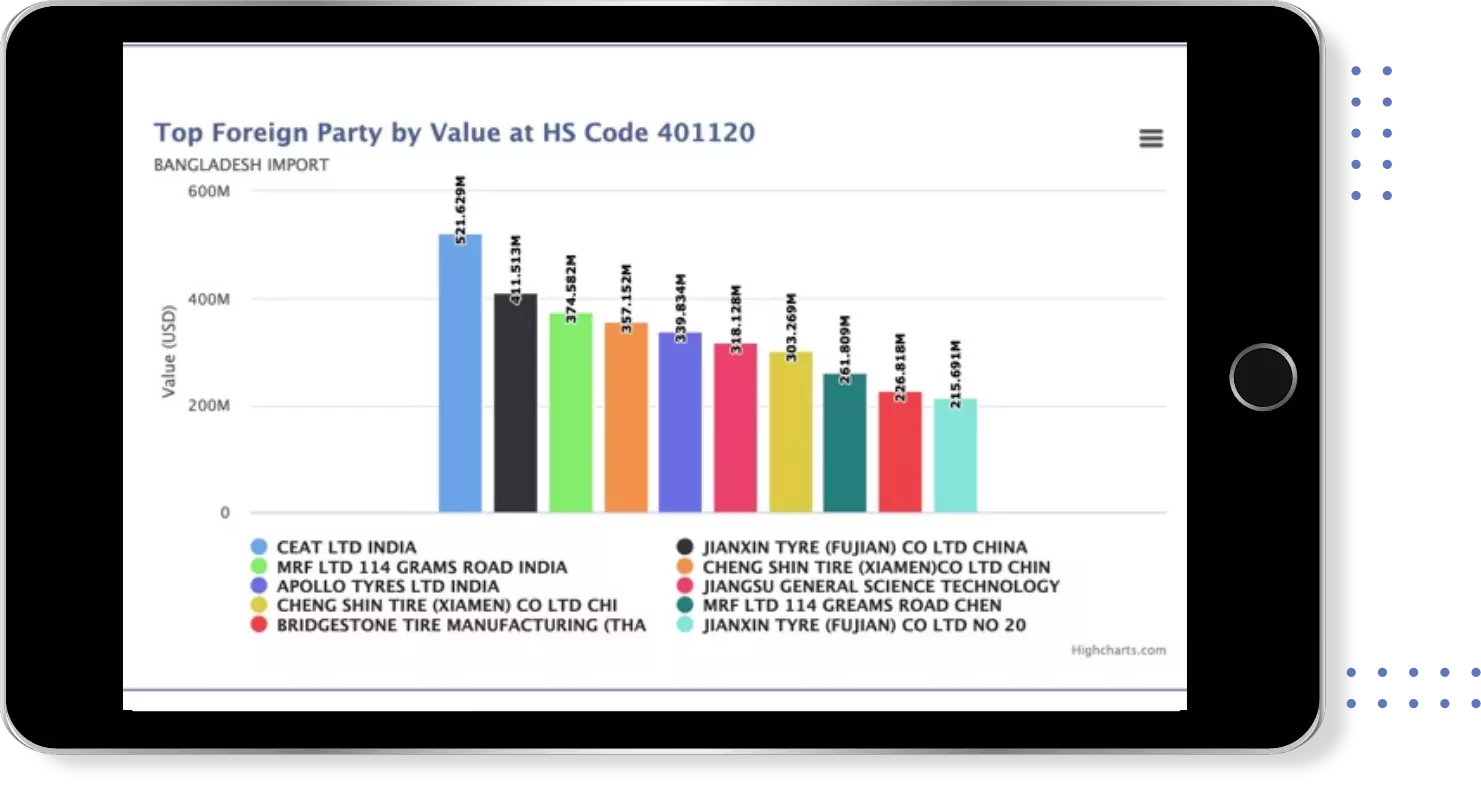
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
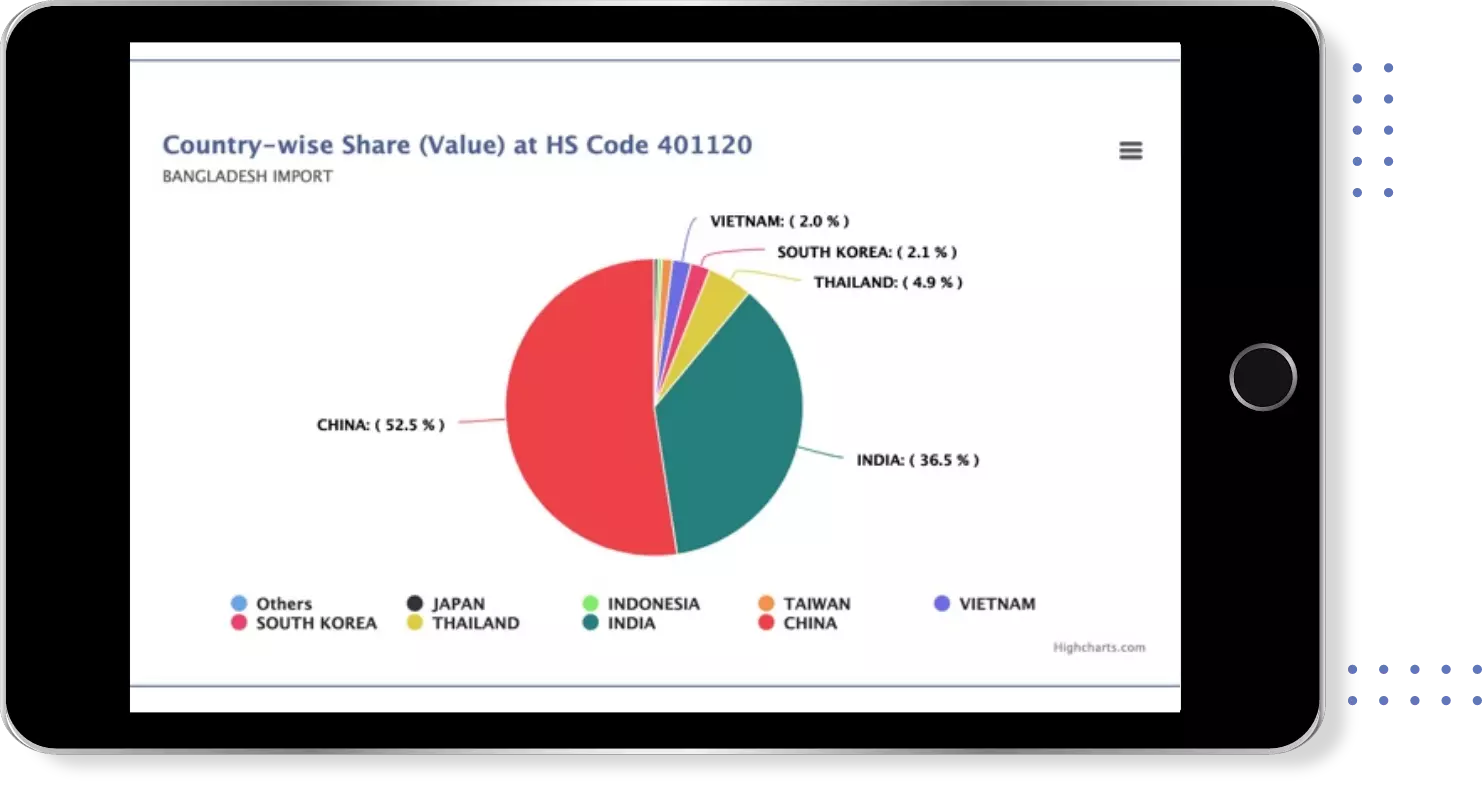
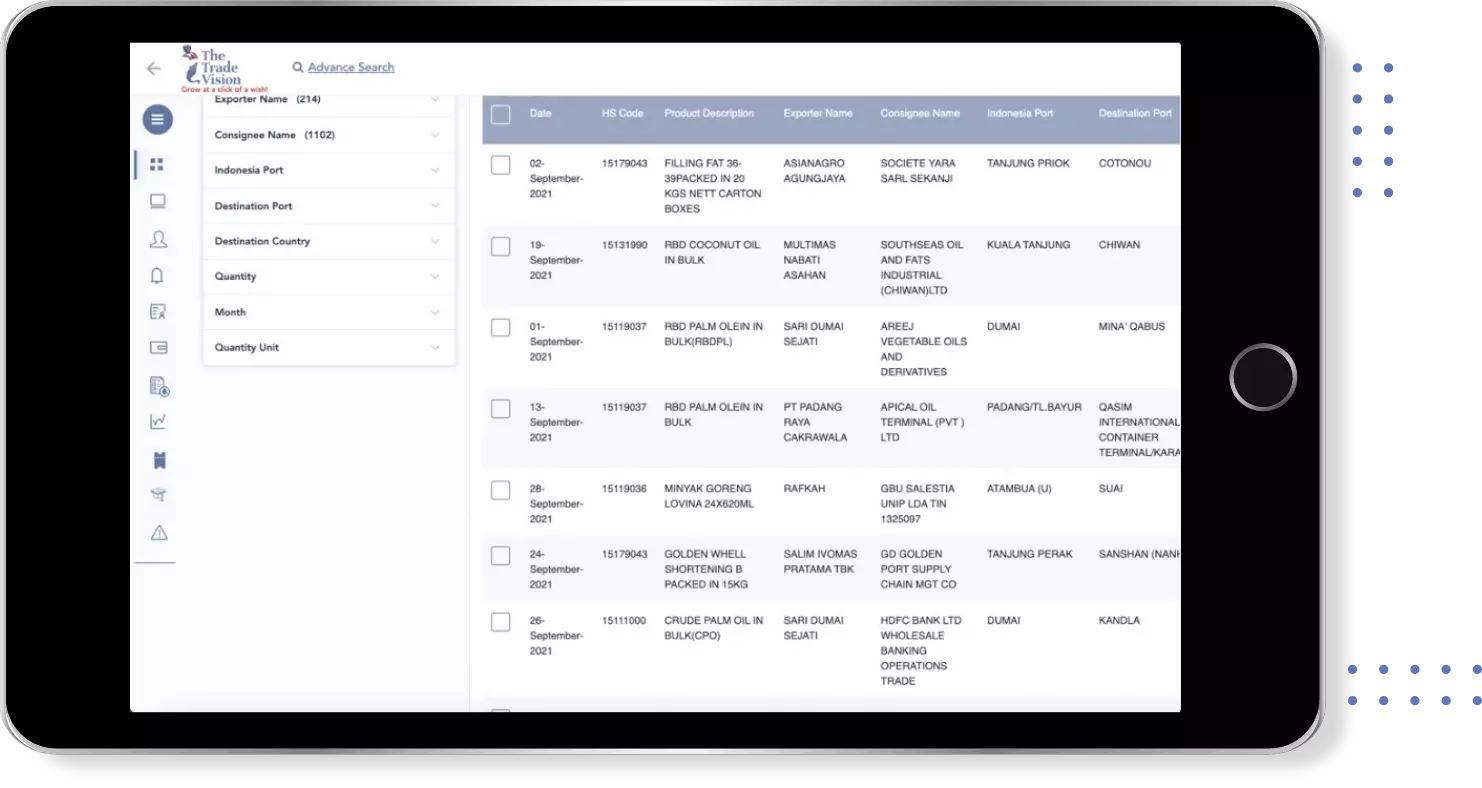
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.