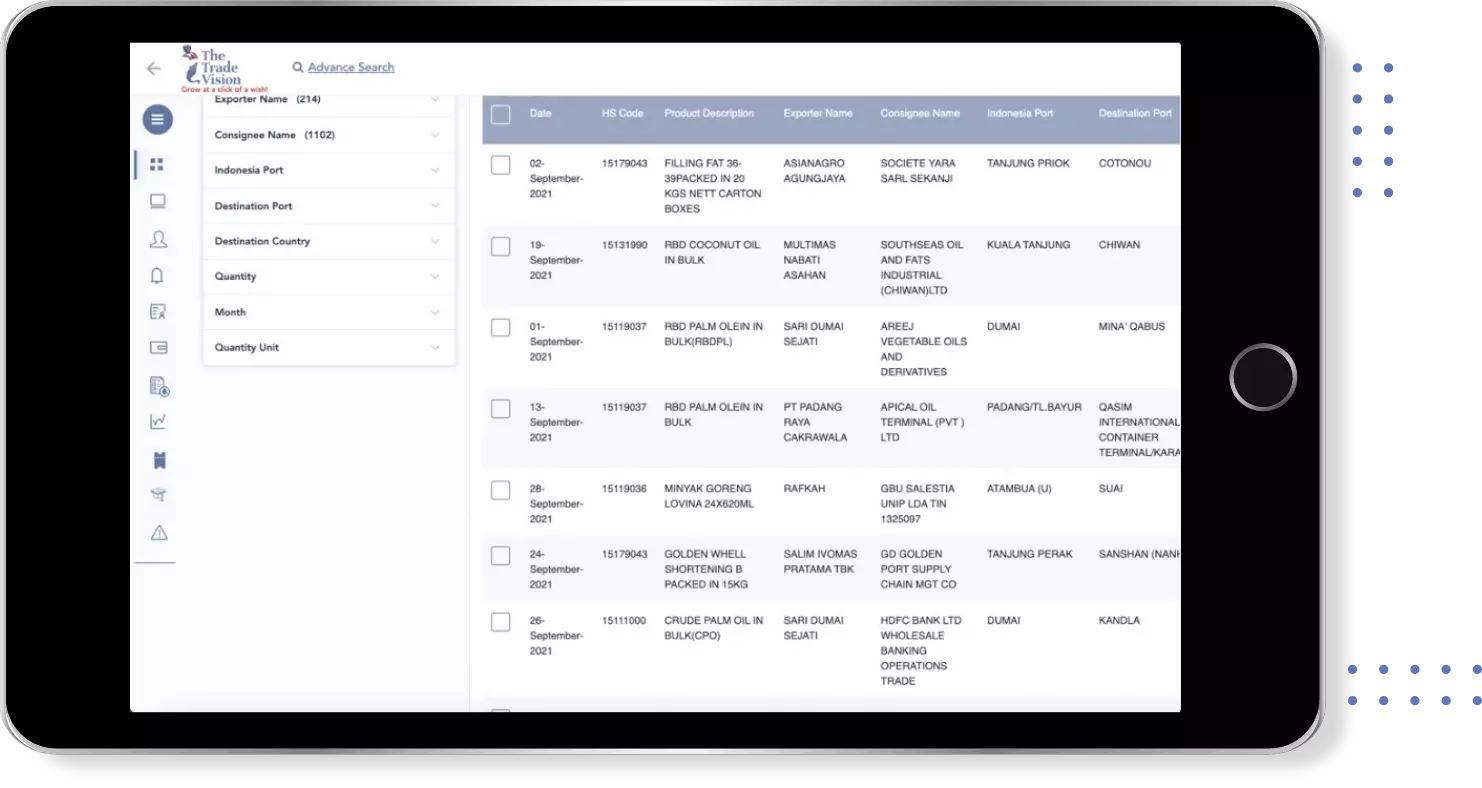
Find verified buyers and sellers of n acetyl in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- n acetyl hs code 300490 import export data
Overview
- The top 3 supplier countries for HS Code 300490 are “VIETNAM” , “UZBEKISTAN” , “SOUTH AFRICA” ,
- The top 3 Buyer countries for HS Code 300490 are “VIETNAM” , “RUSSIA” , “GERMANY” , .
- The top 3 Importing countries for HS Code 300490 are “ARGENTINA” , “SOUTH AFRICA” , “UNITED STATES OF AMERICA” , .
- The top 3 Exporting countries for HS Code 300490 are “FRANCE” , “GERMANY” , “RUSSIA” , .
- Top 1 Product Categories for HS Code 300490 are 300490,
The above summary is based on TTV’s Global Export Import data of HS Code 300490, compiled from 180+ countries export import shipments updated till .
- HS Code: 300490
Global Buyers of n acetyl under HS-Code 300490
NOVARTIS VIETNAM COMPANY LTD

GERMANY
HS-Code
Products
3004909900
ACC 200 (FACTORY FACILITY: SALUTAS PHARMA GMBH) (EACH 3G PACK CONTAINS: ACETYLCYSTEIN 200MG). BOX OF 50 PACKS; POWDER MIXED WITH ORAL SOLUTION. S?K: VN-19978-16. BATCH: LV2460. NSX: 05/2022. HSD: 05/2024
3004905900
ACC 200 (FACTORY: SALUTAS PHARMA GMBH) (ACETYLCYSTEINE 200MG). BOX OF 50 PACKS; POWDER FOR ORAL SOLUTION. REGISTRATION NUMBER: VN-19978-16. BATCH: MP3498. PRODUCER: 03/2023. EXPIRY DATE: MARCH 2025
3004905900
ACC 200 MEDICINE (FACTORY: SALUTAS PHARMA GMBH) (ACETYLCYSTEINE 200MG). BOX OF 50 PACKS; POWDER FOR ORAL SOLUTION. REGISTRATION NUMBER: VN-19978-16. BATCH: MP3503. PRODUCER: 03/2023. EXPIRY DATE: MARCH 2025
PIERRE FABRE VIETNAM CO LTD

FRANCE
HS-Code
Products
30049099
NEW. MEDICINE: TANGANIL 500MG (ACETYLLEUCINE 500MG). TABLETS. BOX OF 3 BLISTERS X 10 TABLETS. LOT: 2G2DU. PRODUCER: 09/2022. HD: 09/2025. VN-22534-20.
3004909900
NEW MEDICINE: TANGANIL 500MG (ACETYLLEUCINE 500MG). TABLETS. BOX OF 3 BLISTERS X 10 TABLETS. LOT:3G2T1 .NSX: 03/2023 HD: 03/2026. VN-22534-20.
3004909900
NEW MEDICINE: TANGANIL 500MG (ACETYLLEUCINE 500MG). TABLETS. BOX OF 3 BLISTERS X 10 TABLETS. LOT: 3G0EX. PRODUCER: 02/2023 HD: 02/2026. VN-22534-20.
NOVARTIS VIETNAM CO LTD

GERMANY
HS-Code
Products
3004909900
ACC 200 (FACTORY FACILITY: SALUTAS PHARMA GMBH) (EACH 3G PACK CONTAINS: ACETYLCYSTEIN 200MG). BOX OF 50 PACKS; POWDER MIXED WITH ORAL SOLUTION. S?K: VN-19978-16. BATCH: LM5289. NSX: 10/2021. HSD: 10/2023
3004909900
ACC 200 (FACTORY FACILITY: SALUTAS PHARMA GMBH) (EACH 3G PACK CONTAINS: ACETYLCYSTEIN 200MG). BOX OF 50 PACKS; POWDER MIXED WITH ORAL SOLUTION. S?K: VN-19978-16. BATCH: LM5290. NSX: 10/2021. HSD: 10/2023
3004909900
ACC 200 (FACTORY FACILITY: SALUTAS PHARMA GMBH) (EACH 3G PACK CONTAINS: ACETYLCYSTEIN 200MG). BOX OF 50 PACKS; POWDER MIXED WITH ORAL SOLUTION. S?K: VN-19978-16. BATCH: LM5293. NSX: 10/2021. HSD: 10/2023
LUKOLL SAC

COLOMBIA
HS-Code
Products
3004902900
RINOFLUIMUCIL 10MG/ML NASAL SOLUTION RINOFLUIMUCIL S/M LOT 06 - 21 DUE DUE. 11/30/2023 FOR HUMAN USE; ACUTE RHINITIS AND SINUSITIS NASAL SOLUTION BOX WITH GLASS BOTTLE TYPE II AMBER COLOR X 10 ML WITH DOSING VALVE ACETYLCYSTEINE / CODE 712002
3004902900
RINOFLUIMUCIL 10MG/ML NASAL SOLUTION RINOFLUIMUCIL S/M LOT 06 - 21 DUE DUE. 11/30/2023 USE: ACUTE RHINITIS AND SINUSITIS FOR HUMAN USE BOX WITH GLASS BOTTLE TYPE II AMBER COLOR X 10 ML WITH DISPENSING VALVE ACETYLCYSTEINE / CODE: 712002
3004902900
RINOFLUIMUCIL 10MG/ML NASAL SOLUTION RINOFLUIMUCIL S/M LOT 02-22 EXPIRATION 08/30/2024 CODE: 712011 MEDICAL SAMPLE USE: ACUTE RHINITIS AND SINUSITIS FOR HUMAN USE BOX WITH GLASS BOTTLE TYPE II AMBER COLOR X 10 ML WITH DOSING VALVE ACETYLCYSTEINE
Global Suppliers of n acetyl under HS-Code 300490
SOUTH DRUGS SA

ARGENTINA
HS-Code
Products
300490241000
O-ACETYLSALICYLIC ACID. OTHERS. MEDICINES (EXCEPT PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTED BY MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKED FOR SALE
300490241000
O-ACETYLSALICYLIC ACID. OTHERS. MEDICINES (EXCEPT PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTED BY MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKED FOR SALE
300490241000
O-ACETYLSALICYLIC ACID. OTHERS. MEDICINES (EXCEPT PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTED BY MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKED FOR SALE
BAYER S A

CHILE
HS-Code
Products
300490241000
O-ACETYLSALICILIC ACID. OTHERS. MEDICINES (EXCEPT FOR PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTING MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKAGED FOR SALE
300490241000
O-ACETYLSALICILIC ACID. OTHERS. MEDICINES (EXCEPT FOR PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTED OF MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKAGED FOR SALE
300490241000
O-ACETYLSALICILIC ACID. OTHERS. MEDICINES (EXCEPT FOR PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTED OF MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKAGED FOR SALE
BAYER SOCIEDAD ANONIMA

CHILE
HS-Code
Products
300490241000
O-ACETYLSALICILIC ACID. OTHERS. MEDICINES (EXCEPT FOR PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTING MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKAGED FOR SALE
300490241000
O-ACETYLSALICILIC ACID. OTHERS. MEDICINES (EXCEPT FOR PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTING MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKAGED FOR SALE
300490241000
O-ACETYLSALICILIC ACID. OTHERS. MEDICINES (EXCEPT FOR PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTING MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKAGED FOR SALE
DROGUERIA SUR SA

ARGENTINA
HS-Code
Products
300490241000
O-ACETYLSALICILIC ACID. OTHERS. MEDICINES (EXCEPT FOR PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTING MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKAGED FOR SALE
300490241000
O-ACETYLSALICILIC ACID. OTHERS. MEDICINES (EXCEPT FOR PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTING MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKAGED FOR SALE
300490241000
O-ACETYLSALICILIC ACID. OTHERS. MEDICINES (EXCEPT FOR PRODUCTS OF HEADING NOS. 3002 3005 OR 3006) CONSTITUTING MIXED OR UNMIXED PRODUCTS PREPARED FOR THERAPEUTIC OR PROPHYLACTIC USES DOSED OR PACKAGED FOR SALE
Global Export Import Trade Data for n acetyl under HS-Code 300490
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 ARGENTINA ARGENTINA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SOUTH AFRICA SOUTH AFRICA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SOUTH AFRICA SOUTH AFRICA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 GERMANY GERMANY | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
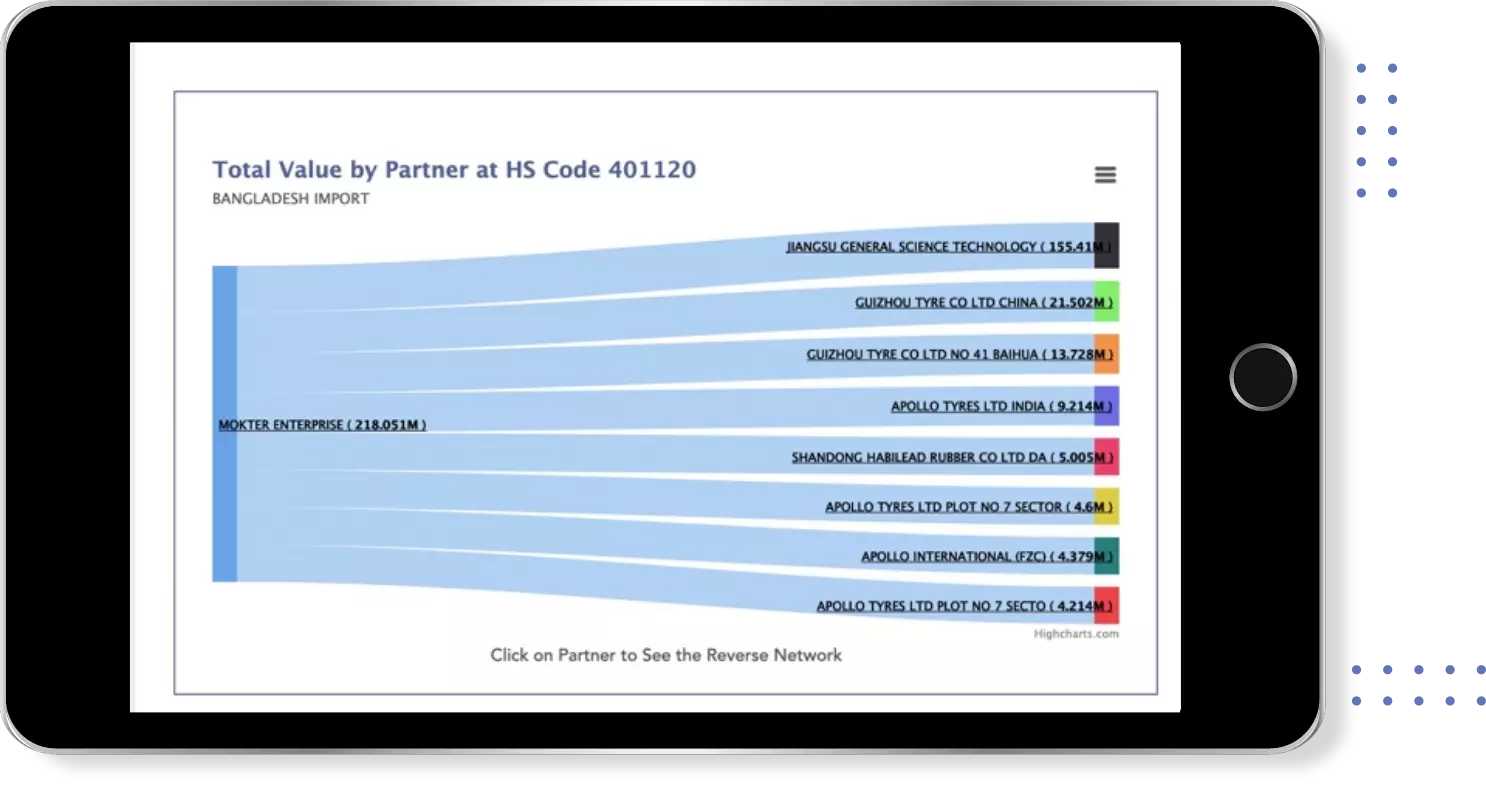
Get most profitable Buyers & Suppliers
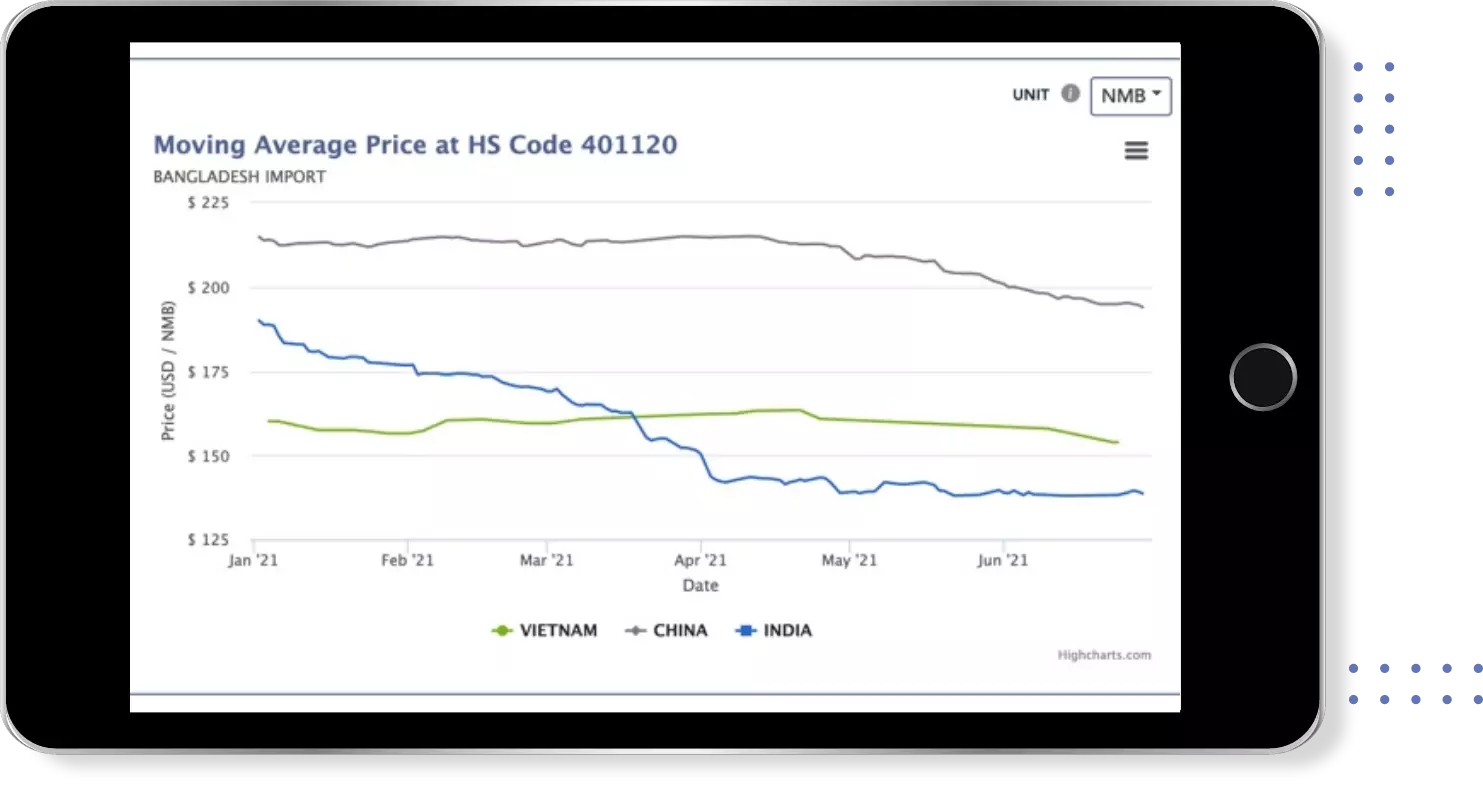
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries