Find verified buyers and sellers of HS-Code 300490 in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- hs code 300490 buyers in ukraine
- COD Ukraine
Ukraine Buyers under HS-Code 300490
JV OPTIMA PHARM LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE. SERIES 120022: ARLEVERT® TABLETS OF 20 TABLETS IN A BLISTER; 1 BLISTER IN A CARDBOARD BOX - 2160 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - DIMENHYDRINATE CINNARIZINE) SERIES 120022: ARLEVERT® TABLETS OF 25 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 900 UNITS. (ACTIVE SUBSTANCE - DIMENHYDRINATE CINNARIZINE) MANUFACTURER: HENNIG ARNZEIMITTEL GMBH & CO. KG DE SERIES 11059A: ATTENTO® 20/5 FILM-COATED TABLETS 20 MG / 5 MG; 14 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 192 UNITS. (ACTIVE SUBSTANCE - AMLODIPINE BESILATE OLMESARTAN MEDOXOMIL) MANUFACTURER: PRODUCTION IN BULK SERIES CONTROL: DAICHI SANKIO EUROPE GMBH DE PACKAGING AND PRODUCTION OF SERIES: BERLIN-CHEMI AG DE. SERIES 20001P: MG 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARDBOARD BOX - 2600 UNITS. (ACTIVE SUBSTANCE - ENALAPRIL MALEATE) MANUFACTURER: PRODUCTION IN BULK PACKAGING CONTROL AND RELEASE OF SERIES: BERLIN-CHEMI AG DE. SERIES 03520A: BERLIPRIL® 5 5 MG TABLETS 10 TABLETS IN A BLISTER; 3 BLISTERS IN A CARDBOARD BOX - 3456 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - ENALAPRIL MALEATE) MANUFACTURER: MANUFACTURER OF BULK PRODUCTION AND BATCH CONTROL: BERLIN-CHEMI AG DE MANUFACTURER OF FINAL PACKAGING: BERLIN-CHEMI AG DE MANUFACTURER OF BATCH PRODUCTION AND CONTROL : BERLIN-CHEMI AG DE. SERIES 11006: BERLITION® 600 UNITS CONCENTRATE FOR SOLUTION FOR INFUSION 600 UNITS (600 MG) / 24 ML 24 ML IN AN AMPOULE; ON 5 AMPOULES IN A CARDBOARD BOX - 420 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - THIOCTIC ACID) MANUFACTURER: SERIES RELEASE: BERLIN-CHEMI AG DE. SERIES 11507A: BROMHEXIN 8 BERLIN-CHEMI COATED TABLETS 8 MG 25 TABLETS IN A BLISTER; ON 1 BLISTER IN A CARDBOARD BOX - 4320 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - BROMHEXINE HYDROCHLORIDE) MANUFACTURER: PRODUCTION OF BULK PRODUCT BATCH CONTROL; PACKAGING CONTROL AND RELEASE OF THE SERIES: BERLIN-CHEMI AG DE SERIES 08091: GLIBOMET® FILM-COATED TABLETS OF 20 TABLETS IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 1440 UNITS. (ACTIVE SUBSTANCE - GLIBENCLAMIDE METFORMIN HYDROCHLORIDE) MANUFACTURER: PRODUCTION IN BULK PACKAGING CONTROL AND RELEASE OF SERIES: MENARINI-VON HAYDEN GMBH DE SERIES 08052: DICLOBERL® RETARD HARD CAPSULES 100 PROLONGED-RELEASE CAPSULES 10 MG PER 10 MG ; 2 BLISTERS IN A CARDBOARD BOX - 8748 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - DICLOFENAC SODIUM) MANUFACTURER: PRODUCTION OF HARD CAPSULES IN BULK SERIES CONTROL: HENNIG ARNZEIMITTEL GMBH & CO. KG DE FINAL PACKAGING SERIES RELEASE: BERLIN-CHEMI AG DE. SERIES 11010A: DICLOBER 100 MG 5 SUPPOSITORIES IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 4480 UNITS. (ACTIVE SUBSTANCE - DICLOFENAC SODIUM) SERIES 11002A: DICLOBERL® 50 SUPPOSITORIES OF 50 MG 5 SUPPOSITORIES IN A BLISTER; 2 BLISTERS IN A CARDBOARD BOX - 1920 UNITARY ENTERPRISE. (ACTIVE SUBSTANCE - DICLOFENAC SODIUM) SERIES 11003A: ESPUMIZAN® L ORAL DROPS EMULSION 40 MG / ML 30 ML IN A BOTTLE WITH A DROPPER-INSERT AND STOPPER; ON 1 BOTTLE IN A CARDBOARD BOX - 3840 UNITARY ENTERPRISE.
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL. SERIES 1 90207-01 0: MEDOPRAM TABLETS COVERED BY FILM COVER 1 0 MG 1 0 TABLETS IN BLISTER; 3 BLISTERS IN A CARDBOARD BOX - 360 UE. (ACTIVE SUBSTANCE - ESCITALOPRAM) SERIES 1 90620-003: MEDOPRAM TABLETS COVERED 20 MG 1 0 TABLETS IN BLISTER; 3 BLISTERS IN A CARDBOARD BOX - 360 UE. (ACTIVE SUBSTANCE - ESCITALOPRAM) SERIES A3G084: NEMOTAN TABLETS 30 MG 1 0 TABLETS IN BLISTER; 1 0 BLISTERS IN A CARDBOARD BOX - 1 20 UP. (ACTIVE SUBSTANCE - NIMODIPINE) MANUFACTURER: MEDOKEMI LTD (FACTORY AZ) CY. SERIES E3G052: MEDOGHER TABLETS COVERED BY 75 MG FILM COVER 1 0 TABLETS IN BLISTER; 3 BLISTERS IN A CARDBOARD BOX - 640 UE. (ACTIVE SUBSTANCE - CLOPID HEATING BISULPHATE) SERIES E3G01 8: RAMIMED 1 0 MG TABLETS 1 0 TABLETS IN BLISTER; 3 BLISTERS IN A CARDBOARD BOX - 240 UE. (ACTIVE SUBSTANCE - RAMIPRIL) MANUFACTURER: MANUFACTURE OF UNPACKAGED PRODUCT QUALITY CONTROL
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE. SERIES C63866: SINGARDI® FILM-COATED TABLETS 12.5 MG / 1000 MG 10 TABLETS IN A BLISTER; 6 BLISTERS IN A CARDBOARD BOX - 360 UNITS. SERIES C60276 : SINGARDI® FILM-COATED TABLETS 12.5 MG / 1000 MG 10 TABLETS IN A BLISTER 6 BLISTERS IN A CARDBOARD BOX - 1000 UNITS (ACTIVE SUBSTANCE - EMPAGLIFLOZIN METFORMIN HYDROCHLORIDE) MANUFACTURER: PRODUCTION QUALITY CONTROL ( EXCEPT FOR THE MICROBIOLOGICAL PURITY TEST) PRIMARY AND SECONDARY PACKAGING BATCH RELEASE: BERINGER INGELHEIM ELLAS AE GR ALTERNATIVE LABORATORY FOR THE CONTROL OF THE MICROBIOLOGICAL PURITY TEST: KACE EPE GR.
BADM LTD LIABILITY COMPANY

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE WITHOUT CONTENT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: EMEND ® CAPSULES OF 125 MG + CAPSULES OF 80 MG COMBI-PACK OF 3 CAPSULES; 1 CAPSULE OF 125 MG + 2 CAPSULES OF 80 MG IN BLISTERS IN A CARDBOARD WRAPPER; 1 CARDBOARD WRAPPER IN A CARDBOARD BOX / 1 CAPSULE CONTAINS 80 MG OR 125 MG OF APREPITANT / U011821; UA / 4525/01 / 01 22.02.2021--252UP; MANUFACTURER ALKERMES PHARMA ISLAND LTD. (PRODUCTION OF BULK PRODUCTS AND QUALITY CONTROL) IRELAND / MERCK SHARP AND DOOM BV (PRIMARY / SECONDARY PACKAGING AND BATCH AUTHORIZATION) NETHERLANDS;
3004900000
1. MEDICINES FOR HUMANS NECESSARY FOR THE IMPLEMENTATION OF MEASURES AIMED AT PREVENTING THE OCCURRENCE AND SPREAD LOCALIZATION AND ELIMINATION OF OUTBREAKS EPIDEMICS AND PANDEMICS OF CORONAVIRUS COVID-19 WITHOUT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS PACKAGED FOR RETAIL SALE IN AEROSOL PACKAGING: ESMERON ® SOLUTION FOR INJECTION 10 MG / ML 5 ML (50 MG) IN A VIAL; 10 VIALS IN A CARDBOARD BOX / 1 ML OF SOLUTION CONTAINS 10 MG OF ROCURONIUM BROMIDE / INTERNATIONAL NON-PROPRIETARY NAME: ROCURONIUM BROMIDE; MANUFACTURER SIEGFRIED HAMELN GMBH (PRODUCTION IN BULK PRIMARY PACKAGING AND QUALITY CONTROL) GERMANY / HAMELN RDS (ALTERNATIVE QUALITY CONTROL) SLOVAKIA;
3004900000
1. MEDICINES FOR PEOPLE WITHOUT CONTENTS: ANTIBIOTICS ALKALOIDS HORMONES VITAMINS FOR RETAIL NOT IN AEROSOL. AKTYLIZE ® LYOPHILISATES FOR SOLUTION FOR INFUSION 50 MG 1 VIAL WITH LYOPHILIZATE COMPLETE WITH 1 VIAL OF SOLVENT (WATER FOR INJECTION) IN 50 ML CARTON / POWDER 1 VIAL LYOPHILIZED FOR SOLUTION FOR INFUSION CONTAINS: 50 MG ALTEPLASE ; 1 SOLVENT VILLAGE CONTAINS STERILE INJECTION WATER 50 ML / PRODUCT. BERINGER INGELHEIM PHARMA GMBH & CO. KI GERMANY MIRAPEX ® 1 MG TABLETS 1 0 TABLETS IN BLISTER; 3 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS PRAMIPEXOL DIHYDROCHLORIDE MONOGHYDRATE 1 MG CORRESPONDING 0.7 MG OF PRAMIPERCO. BERINGER INGELHEIM PHARMA GMBH & CO. KG GERMANY;
SP OPTIMA FARM LTD UKRAINE M KYIV KIKVIDZE STREET 1 8 A

UKRAINE
HS-Code
Products
3004900000
MEDICINES (MEDICINES) (EXCEPT FOR GOODS INCLUDED IN HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM (INCLUDING MEDICINES IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
3004900000
MEDICINES (MEDICINES) (EXCEPT FOR GOODS INCLUDED IN HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM (INCLUDING MEDICINES IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
3004900000
MEDICINES (MEDICINES) (EXCEPT FOR GOODS INCLUDED IN HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM (INCLUDING MEDICINES IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
BADM LTD LIABILITY COMPANY 49040 DNIPROPETROVSKA OBL M DNIPRO 2 PANIKAHY STREET

UKRAINE
HS-Code
Products
3004900000
MEDICINES (MEDICINES) (EXCEPT FOR GOODS INCLUDED IN HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM (INCLUDING MEDICINES IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
3004900000
MEDICINES (MEDICINES) (EXCEPT FOR GOODS INCLUDED IN HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM (INCLUDING MEDICINES IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
3004900000
MEDICINES (MEDICINES) (EXCEPT FOR GOODS INCLUDED IN HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM (INCLUDING MEDICINES IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
TOV VENTA Â LTD

UKRAINE
HS-Code
Products
3004900000
3004900000
3004900000
TOV VENTA LTD 49000 M DNIPRO SELYANSKY UZVIZ 3A

UKRAINE
HS-Code
Products
3004900000
MEDICINES (MEDICINES) (EXCEPT FOR GOODS INCLUDED IN HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM (INCLUDING MEDICINES IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
3004900000
MEDICINES (MEDICINES) (EXCEPT FOR GOODS INCLUDED IN HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM (INCLUDING MEDICINES IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
3004900000
MEDICINES (MEDICINES) (EXCEPT FOR GOODS INCLUDED IN HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSAGE FORM (INCLUDING MEDICINES IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
HFC BIOCON LLC

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE PACKAGED FOR RETAIL TRADE: -TOTEM (ACTIVE SUBSTANCES: 1 AMPOULE (10 ML) CONTAINS: IRON (IN THE FORM OF IRON GLUCONATE) 50 MG; MANGANESE (IN THE FORM OF MANGANESE GLUCONATE) 1.33 MG; COPPER (IN THE FORM OF COPPER GLUCONATE) 0.7 MG) SOLUBLE 10 ML IN AN AMPOULE; 10 AMPOULES IN A CELL PACKAGE; 2 CELL PACKAGES IN A CARDBOARD BOX - SERIES: 59034 -12528UP MANUFACTURER: INNOTERA SHUZI FRANCE COUNTRY OF MANUFACTURE: INTERNATIONAL 2023
3004900000
1. MEDICINES FOR HUMANS PACKAGED FOR RETAIL: -HEPTRAL (R) LYOPHILIZED POWDER FOR SOLUTION FOR INJECTION OF 500 MG 5 GLASS VIALS WITH LYOPHILIZED POWDER AND 5 AMPOULES WITH SOLVENT (L-LYSINE SODIUM HYDROXIDE WATER FOR INJECTIONS) 5 ML IN A CONTOUR HONEYCOMB PACKAGE SEALED WITH ALUMINUM FOIL; 1 CONTOUR HONEYCOMB PACKAGE IN A CARDBOARD BOX (ACTIVE INGREDIENTS: 1 VIAL OF LYOPHILIZED POWDER CONTAINS: 949 MG OF ADEMETHIONINE 1 4-BUTANE DISULFONATE CORRESPONDING TO PACKAGING AND QUALITY CONTROL OF POWDER SECONDARY PACKAGING QUALITY CONTROL AND PRODUCTION OF GLZ SERIES: BIOLOGIES ITALY LABORATORIES SRL; ITALYCOUNTRY OF PRODUCTION: ITBRAND: ABBOTT HEPTRAL SHELF LIFE: 22.01.2024
3004900000
1. MEDICINES FOR HUMANS PACKAGED FOR RETAIL: -HEPTRAL (R) LYOPHILIZED POWDER FOR SOLUTION FOR INJECTION OF 500 MG 5 GLASS VIALS WITH LYOPHILIZED POWDER AND 5 AMPOULES WITH SOLVENT (L-LYSINE SODIUM HYDROXIDE WATER FOR INJECTIONS) 5 ML IN A CONTOUR HONEYCOMB PACKAGE SEALED WITH ALUMINUM FOIL; 1 CONTOUR HONEYCOMB PACKAGE IN A CARDBOARD BOX (ACTIVE SUBSTANCES: 1 VIAL OF LYOPHILIZED POWDER CONTAINS: 949 MG OF ADEMETHIONINE 1 4-BUTANE DISULFONATE CORRESPONDING TO 500 MG OF PACKAGING AND QUALITY CONTROL OF POWDER SECONDARY PACKAGING QUALITY CONTROL AND PRODUCTION OF GLZ SERIES: DELFARM SAINT-REMY SOLVENT: FAMAR AVE ALIMOS PLANT 63 AGIT DIMITRIOU STREET FRANCECOUNTRY OF PRODUCTION: FRCOMPANY: ABBOTT HEPTRAL EXPIRY DATE: 13.02.2024
TOV VENTA LTD

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. REVOLAD ™ FILM-COATED TABLETS 25 MG 7 TABLETS IN A BLISTER; 4 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS 25 MG ELTROMBOPAG (IN THE FORM OF ELTROMBOPAG OLAMINE) / GLAXO OPERATIONS UK LTD (PRODUCTION QUALITY CONTROL) UNITED KINGDOMGLAXO WELLCOME SA (MANUFACTURER FOR PRIMARY AND SECONDARY PACKAGING AND SERIES RELEASE) SPAIN. VOTRIENT ™ FILM-COATED TABLETS 400 MG 30 TABLETS IN A VIAL 1 VIAL IN A CARDBOARD BOX / 1 TABLET CONTAINS 400 MG OF PAZOPANIB (AS PAZOPANIB HYDROCHLORIDE) / GLAXO OPERATIONS (MANUFACTURER OF BULK PRODUCTS) UNITED KINGDOMGLAXO WELLCOME SA (MANUFACTURER FOR PACKAGING AND BATCH PRODUCTION) SPAIN
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. SPIRIVA® POWDER FOR INHALATION HARD CAPSULES 18 MCG 10 CAPSULES WITH POWDER FOR INHALATION IN A BLISTER 3 BLISTERS COMPLETE WITH A HANDY HEILER® DEVICE IN A CARDBOARD BOX / 1 CAPSULE CONTAINS TIOTROPIUM BROMIDE MONOHYDRATE 22.5 MCG CORRESPONDING TO TIOTROPIUM 18 MCG / PRADAXA CAPSULES 10 HARD CAPSULES 110 MG BLISTERS 6 BLISTERS IN A BOX / 1 CAPSULE CONTAINS DABIGATRAN ETEXILATE (AS MESYLATE) 110 MG / PRADAXA® HARD CAPSULES OF 150 MG 10 CAPSULES IN A BLISTER 6 BLISTERS IN A CARDBOARD BOX / 1 CAPSULE CONTAINS 150 MG OF DABIGATRAN ETEXILATE IN THE FORM OF MESYLATE / SPIRIVA® RESPIMAT® SOLUTION FOR INHALATION 2.5 ?G / INHALATION 4 ML IN A CARTRIDGE (60 INHALATIONS) 1 CARTRIDGE COMPLETE WITH 1 INHALER RESPIMAT® IN A CARDBOARD BOX / 1 INHALATION CONTAINS TIOTROPIUM BROMIDUMO HYDRATE 3 124 MCG CORRESPONDING TO 2.5 MCG TIOTROPIUM / IN AEROSOL PACKAGING. BEROTEK® H AEROSOL DOSED 100 MCG / DOSE 10 ML (200 DOSES) IN A METAL CAN WITH A DOSING VALVE; 1 CAN IN A CARDBOARD BOX / 1 DOSE CONTAINS FENOTEROL HYDROBROMIDE 100 MCG / BERINGER INGELHEIM PHARMA GMBH AND CO. KG GERMANY.
3004900000
1. MEDICINES FOR PEOPLE FOR THERAPEUTIC USE PACKAGED FOR RETAIL TRADE (NOT CONTAINING PENICILLINS ANTIBIOTICS HORMONES ALKALOIDS VITAMINS IODINE COMPOUNDS) NOT IN AEROSOL PACKAGING. BETASERK® TABLETS OF 16 MG: 15 TABLETS IN A BLISTER 2 BLISTERS IN A CARTON / 1 TABLET CONTAINS BETAHISTINE DIHYDROCHLORIDE 16 MG / MYLAN LABORATORIES CAC FRANCE. DRIPTAN® TABLETS OF 5 MG 30 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD BOX / 1 TABLET CONTAINS OXYBUTYNIN HYDROCHLORIDE .DUSPATALIN® FILM-COATED TABLETS 135 MG 15 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD BOX / 1 TABLET CONTAINS MEBEVERINE HYDROCHLORIDE 135 MG / MYLAN LABORATORIES CAC FRANCEPHYSIOTENS® FILM-COATED TABLETS 0.2 MG FILM-COATED 28 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD BOX / 1 TABLET CONTAINS MOXONIDINE 0.2 MG / MYLAN LABORATORIES CAC (FULL CYCLE PRODUCTION) FRANCE.PHYSIOTENS® FILM-COATED TABLETS YU 0.4 MG 28 TABLETS IN A BLISTER 1 BLISTER IN A CARDBOARD BOX / 1 TABLET CONTAINS MOXONIDINE 0.4 MG / MYLAN LABORATORIES CAC (FULL CYCLE PRODUCTION) FRANCE.RITMONORM® FILM-COATED TABLETS 150 MG OF 10 TABLETS IN A BLISTER; 5 BLISTERS IN A CARDBOARD BOX / 1 TABLET CONTAINS PROPAFENONE HYDROCHLORIDE 150 MG / FAMAR LYON FRANCE.
TOV BADM

UKRAINE
HS-Code
Products
3004900000
1. MEDICINES FOR PEOPLE WITHOUT CONTENT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS PACKAGED FOR RETAIL TRADE NOT IN AEROSOL PACKAGING: FLUTAMIDE TABLETS OF 250 MG 21 TABLETS IN A BLISTER; 4 BLISTERS IN A CARDBOARD BOX / FLUTAMIDE /; MANUFACTURER: ORION CORPORATION
3004900000
1. MEDICINES FOR PEOPLE WITHOUT CONTENT: VITAMINS HORMONES ALKALOIDS ANTIBIOTICS FOR RETAIL TRADE NOT IN AEROSOL PACKING. PHASLODEX SOLUTION FOR INJECTIONS 250 MG / 5 ML ON 5 ML OF SOLUTION IN THE PRE-FILLED GLASS SYRINGE WITH CONTROL OF THE FIRST OPENING 2 PRE-FILLED SYRINGES IN A CONTOUR HONEYCOMB PACKAGE WITH TWO SAFETY NEEDLES BD SAFETYGLIDE SEALED WITH POLYPROPYLENE FILM 1 CONTOUR HONEYCOMB PACKAGE IN A CARDBOARD BOX / FULVESTRANT /; MANUFACTURER: VETTER PHARMAGERT. MANUFACTURER OF IN BULK PRIMARY PACKAGING) GERMANY / ASTRAZENECA UK LIMITED (MANUFACTURER RESPONSIBLE FOR SECONDARY PACKAGING QUALITY CONTROL BATCH RELEASE) GREAT BRITAIN;
3004900000
1.LIKY FOR PEOPLE FREE OF: ANTIBIOTICS ALKALOIDS HORMONES VITAMINS RETAIL NOT AEROSOL UPAKOVTSI.AKTYLIZE ® LYOPHILISATES FOR SOLUTION FOR INFUSION 50 MG 1 VIAL WITH LYOPHILIZATE COMPLETE WITH 1 VIAL OF SOLVENT (WATER FOR INJECTION) IN 50 ML CARTON / POWDER 1 VIAL LYOPHILIZED FOR SOLUTION FOR INFUSION CONTAINS: 50 MG ALTEPLASE ONE VIAL OF SOLVENT CONTAINS STERILE WATER FOR INJECTION 50 MG / VYROBN.BERINHER INGELHEIM PHARMA GMBH AND CO . KG GERMANY;
BADM LTD LIABILITY COMPANY UKRAINE 49040 DNIPROPETROVSKA OBL M DNIPRO SOBORNYI R N STREET

UKRAINE
HS-Code
Products
3004900000
MEDICINAL PRODUCTS (DRUGS) (EXCLUDING GOODS OF HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSED FORM (INCLUDING MEDICINAL PRODUCTS IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
3004900000
MEDICINAL PRODUCTS (DRUGS) (EXCLUDING GOODS OF HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSED FORM (INCLUDING MEDICINAL PRODUCTS IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
3004900000
MEDICINAL PRODUCTS (DRUGS) (EXCLUDING GOODS OF HEADING 3002 3005 OR 3006) CONSISTING OF MIXED OR UNMIXED PRODUCTS FOR THERAPEUTIC OR PROPHYLACTIC USE IN DOSED FORM (INCLUDING MEDICINAL PRODUCTS IN THE FORM OF TRANSDERMAL SYSTEMS) OR PACKAGED FOR RETAIL TRADE: - OTHERS
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
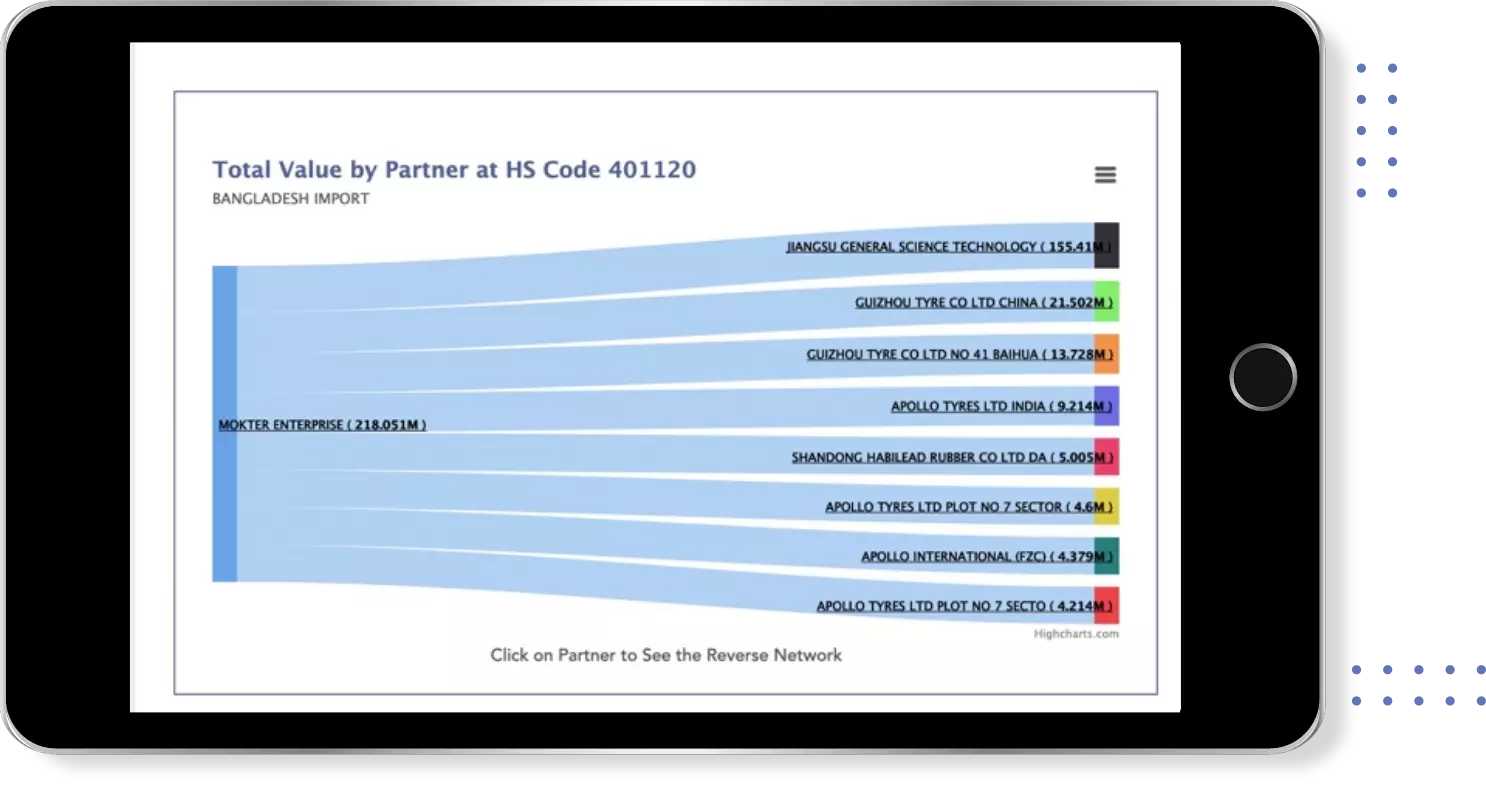
Get most profitable Buyers & Suppliers
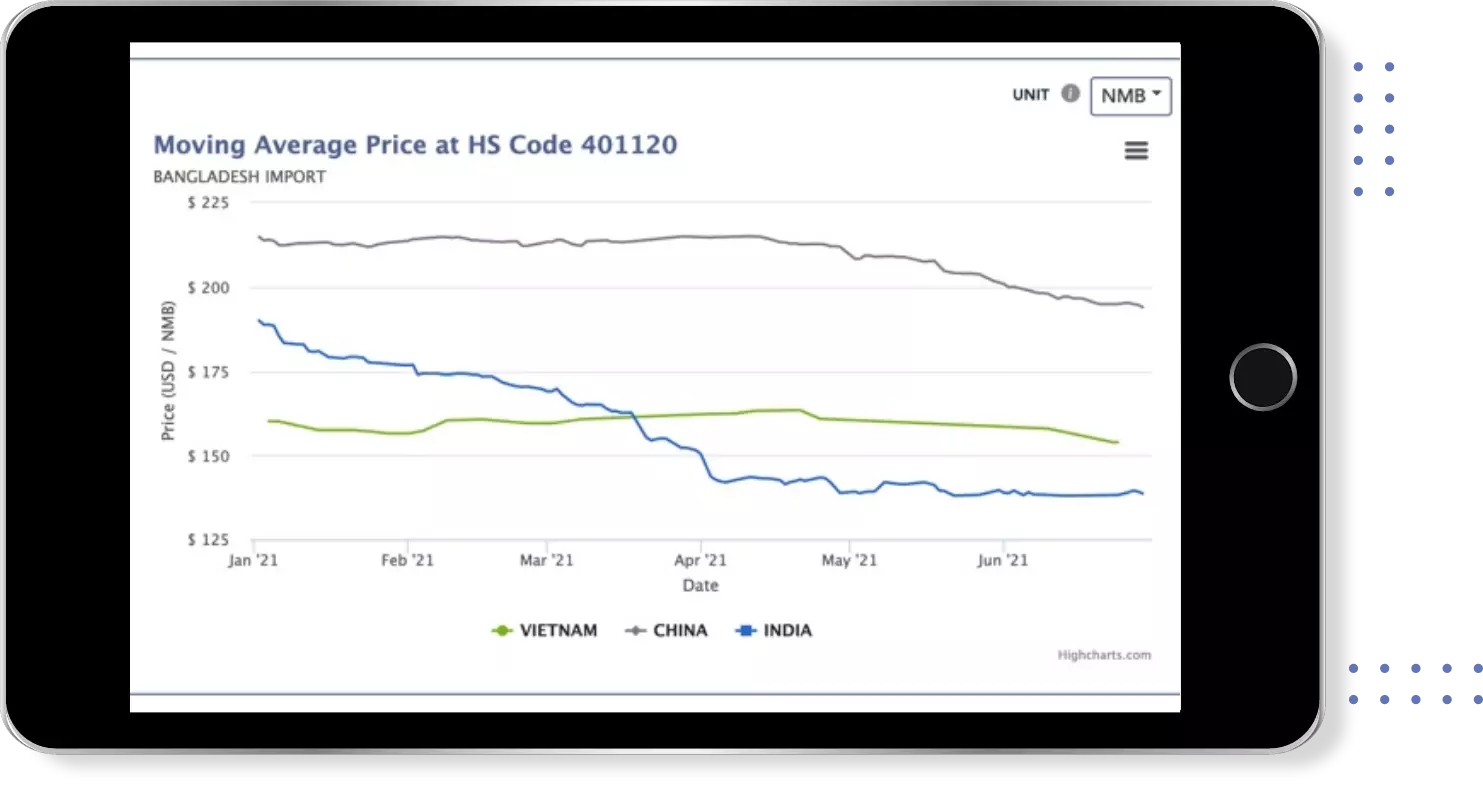
Instant insights on Price , Quantity & Value trends
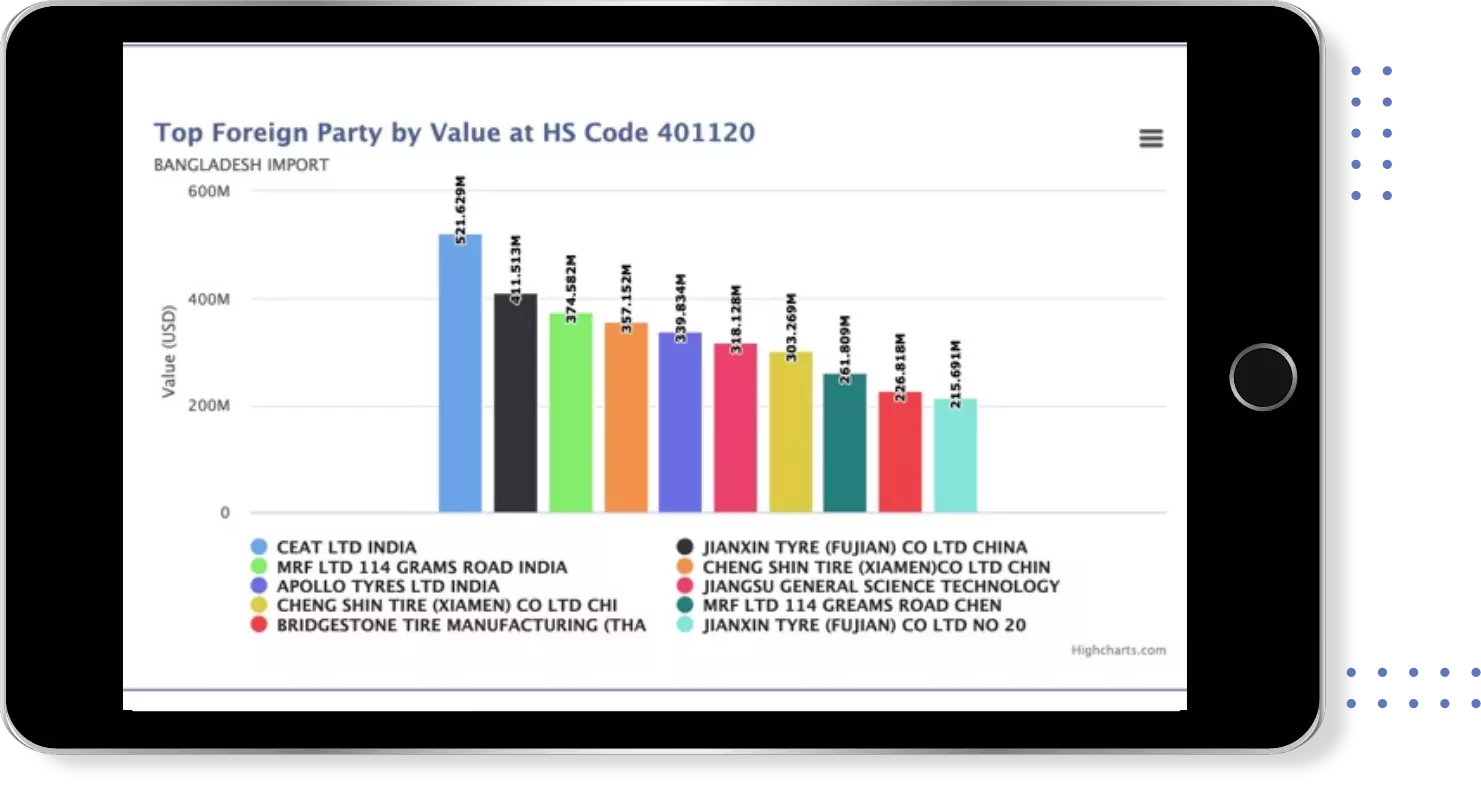
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
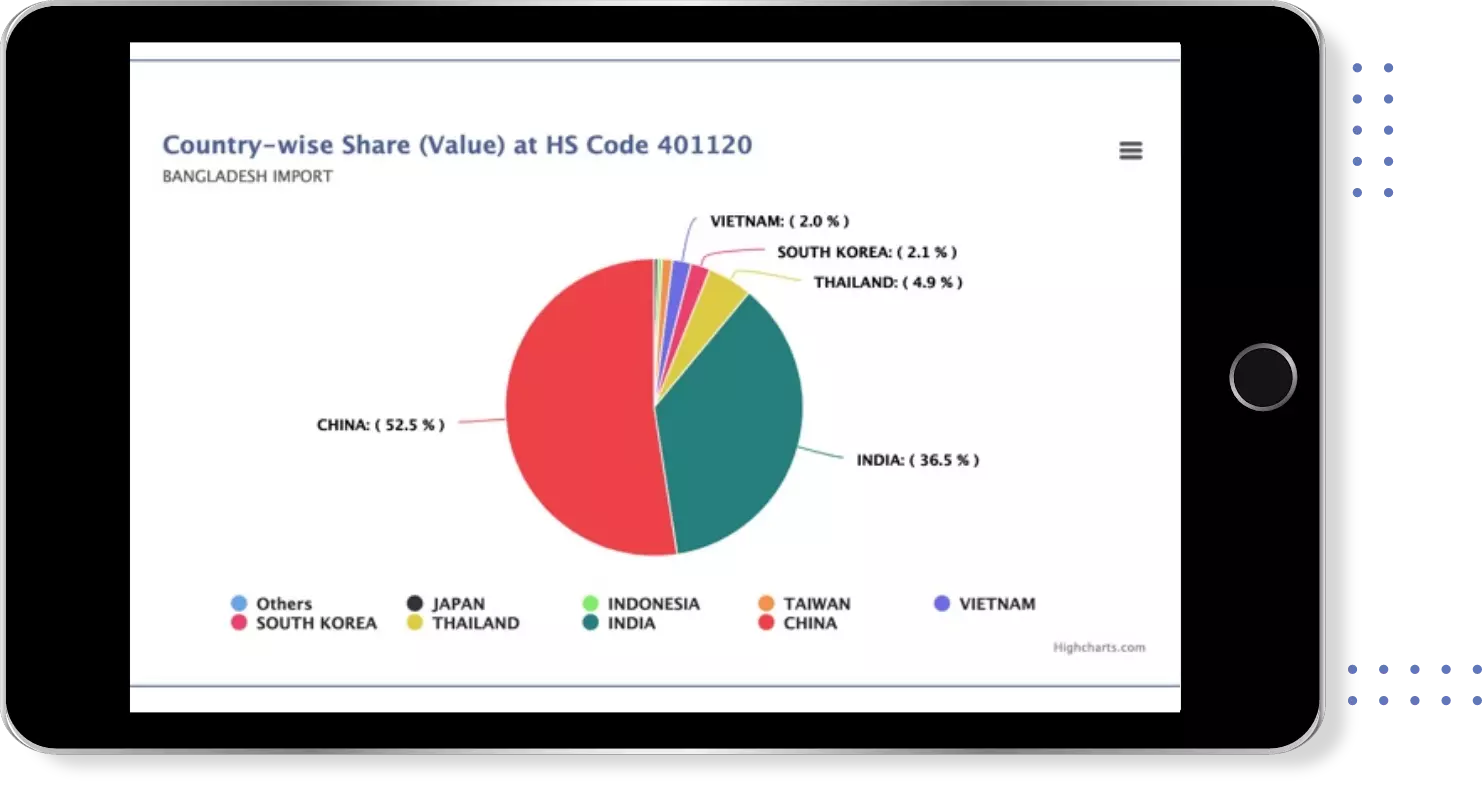
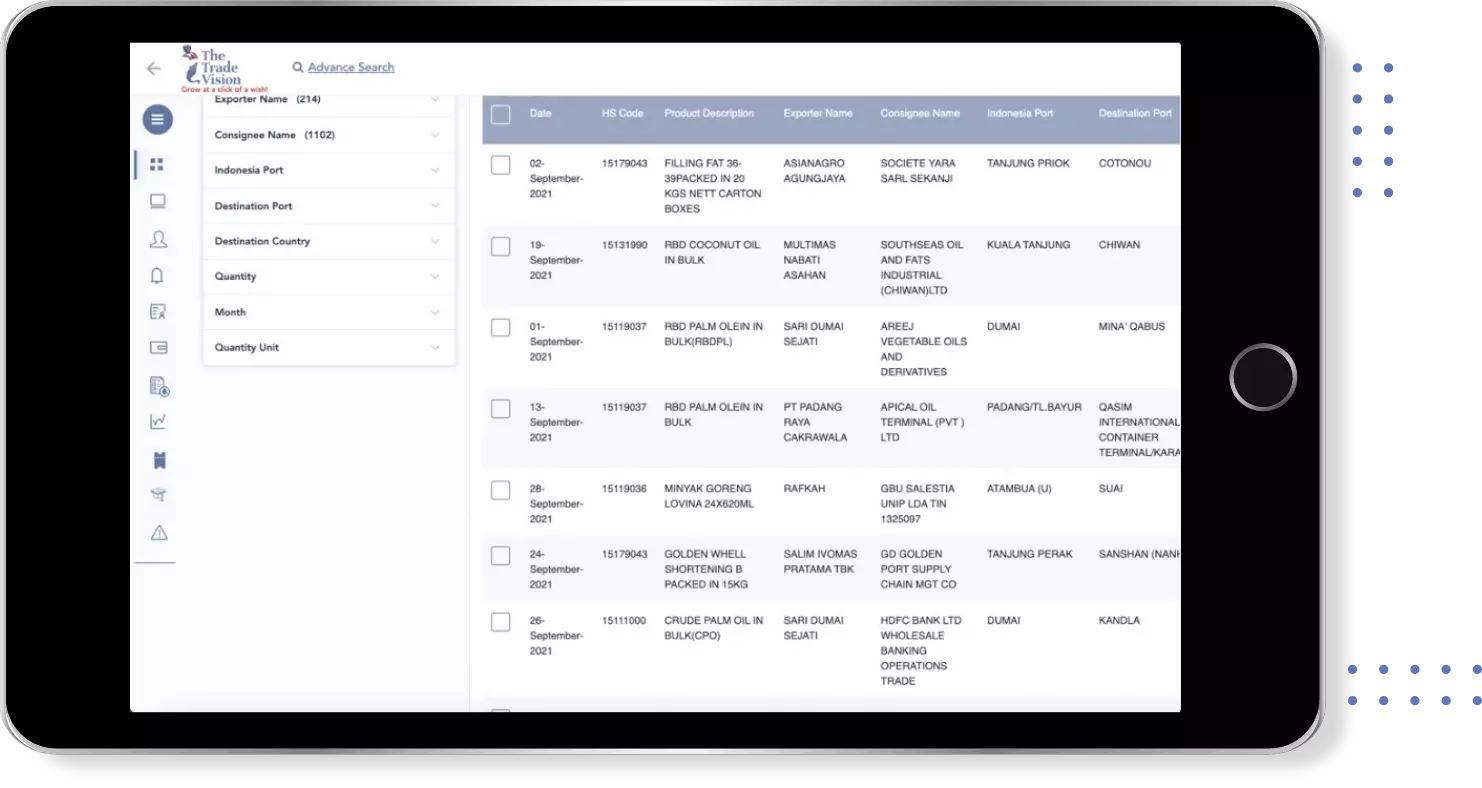
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.