Find verified buyers and sellers of HS-Code 300210 in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- hs code 300210 buyers in ukraine
- COD Ukraine
Ukraine Buyers under HS-Code 300210
SM AGENCY LLC UKRAINE

UKRAINE
HS-Code
Products
3002109900
3002109900
1. RELATED MATERIALS FOR MULTICENTER CLINICAL TRIALS ACCORDING TO PROTOCOL EFC15804: GENERAL LABORATORY PRODUCTS OF NON-ANIMAL ORIGIN MODIFIED IMMUNOBIOLOGICAL PRODUCTS (MONO AND POLYCLONAL ANTIBODIES) ARE APPLIED TO TEST STRIPS THE PRINCIPLE OF WHICH IS BASED ON THE ANTIGEN-ANTIBODY REACTION: - TEST FOR ANALYSIS URINE FOR PREGNANCY TYPE QUICKVUE (25 PIECES IN A SET) - 2 SETS. MANUFACTURER: QUIDEL CORPORATION. BRAND: QUICKVUE. COUNTRY OF MANUFACTURE US.
3002109900
1. IMMUNOMODULATORY DRUGS ARE NOT REGISTERED (NOT IN AEROSOL PACKAGING) FOR HUMANS INTENDED FOR CLINICAL TRIALS UNDER THE PROTOCOL MK-3475-826 NOT FOR RETAIL NON-ANIMAL ORIGIN.- MK-3475 STERILE SOLUTION FOR INTRAVENOUS INFUSION 25 MG / ML (100 MG / 4ML ) IN A BOTTLE IN A SET ?2 - 180 SETS. PACKAGING SERIES: S031673. SHELF LIFE - 31/01 / 2021.MANUFACTURER: MSD INTERNATIONAL GMBH T / A MSD IRELAND (CARLOW). TRADEMARK - MSD. COUNTRY OF MANUFACTURE IE.
ALSI LTD

UKRAINE
HS-Code
Products
3002109900
1.LABORATORY BIOCHEMICAL REAGENTS FOR SCIENTIFIC RESEARCH. REAGENT FOR ENZYME-LINKED IMMUNOSORBENT ASSAY (ELISA). NON-MEDICAL PRODUCT. (NOT FOR USE IN VETERINARY MEDICINE AND ANIMAL HUSBANDRY). -500ML - 8PCS NET WEIGHT NET-4000G FETAL BEEFEAT NON-US ORIGIN STERILIZED BY FILTRATION FOR CULTURING CELLS - 500ML ITEM F7524-500ML - 16PCS NET WEIGHT NET-8000G LABORATORY REAGENTS - NOT FOR MEDICAL USE. SIGMA-ALDRICH . MANUFACTURER - SIGMA-ALDRICH CHEMIE GMBH . COUNTRY OF MANUFACTURE - BR.
3002109900
1. LABORATORY BIOCHEMICAL REAGENTS FOR ENZYME-LINKED IMMUNOSORBENT ASSAY FOR SCIENTIFIC RESEARCH THAT REQUIRES SPECIAL STORAGE CONDITIONS AT A TEMPERATURE OF MINUS 20-70 DEGREES CELSIUS. NON-MEDICAL PRODUCTS. (NOT FOR THERAPEUTIC PROPHYLACTIC OR DIAGNOSTIC USE. NOT FOR USE IN VETERINARY MEDICINE AND ANIMAL HUSBANDRY). ANTIBODIES TO HUMAN HGF - 100MCG ART. MAB694-100 - 1 PIECE. NET WEIGHT - 100 MCG. POLYCLONAL RABBIT ANTIBODIES OF HUMAN ERYTHROPOETIN - 1MG ART. AB-286-NA - 1PC. NET WEIGHT - 1 MG. TRADEMARK - R&D SYSTEMS. MANUFACTURER - BIO-TECHNE LTD. COUNTRY OF MANUFACTURE - US.
3002109100
1. OFFICIAL STANDARD SAMPLES OF THE EUROPEAN PHARMACOPOEIA FOR LABORATORY RESEARCH DURING QUALITY CONTROL OF MEDICINAL PRODUCTS. REQUIRE STORAGE CONDITIONS -20 DEGREES C. GLOBULINS. NOT FOR USE IN VETERINARY MEDICINE AND ANIMAL HUSBANDRY. DO NOT CONTAIN GMOS. IMMUNOGLOBULIN ANTI-B TEST ) NEGATIVE CONTROL 50MG ITEM Y0001689 - 2PCS NET NET WEIGHT-100MG. NOT INTENDED FOR HUMAN CONSUMPTION. TRADEMARK - EDQM . MANUFACTURER - EUROPEAN DIRECTORATE FOR THE QUALITY OF MEDICINES & HEALTHCARECOUNCIL OF EUROPE FRANCE. COUNTRY OF MANUFACTURE - FR.
YUVIS LLC

UKRAINE
HS-Code
Products
3002109500
1. MEDICAL DEVICES FOR IN VITRO DIAGNOSTICS: COMPLEX LABORATORY IMMUNOLOGICAL REAGENTS OF HUMAN ORIGIN OBTAINED USING BIOTECHNOLOGICAL PROCESSES: B25700 AQUIOS IMMUNO-TROL LOW CELLS - 52 PCS.
3002109500
1. MEDICAL DEVICES FOR IN VITRO DIAGNOSTICS: COMPLEX LABORATORY IMMUNOLOGICAL REAGENTS HUMAN ORIGIN OBTAINED USING BIOTECHNOLOGICAL PROCESSES: B23535 AQUIOS IMMUNO-TROL CELLS - 22 PCS. B25700 AQUIOS IMMUNO-TROL LOW CELLS - 11 PCS. B25700 AQUIOS IM TROL LOW CELLS - 2 ??. 6607098 IMMUNO-TROL LOW CELLS - 4 ??.
3002109900
1. MEDICAL DEVICES FOR IN VITRO DIAGNOSTICS: COMPLEX LABORATORY IMMUNOLOGICAL REAGENTS OF COMBINED ORIGIN OBTAINED USING BIOTECHNOLOGICAL PROCESSES: OSR6142 APO A1 - 2 PCS. OSR6142 APO A1 - 1 PC.
DIALOGUE DIAGNOSTICS LTD

UKRAINE
HS-Code
Products
3002109500
1. MEDICAL DEVICES INTENDED BY THE MANUFACTURER FOR USE IN VITRO FOR RESEARCH IN THE LABORATORY FOR INFORMATION ON PHYSIOLOGICAL OR PATHOLOGICAL CONDITION: COMPLEX LABORATORY IMMUNOLOGICAL REAGENTS OF COMBINED ORIGIN OBTAINED USING BIOTECHNOLOGICAL PROCESSES: REAGENTS COMPLEX (ANALYTICAL) DESIGNED FOR BLOOD TESTS USING IMMUNO-FERMENTER COBAS E411: CALIBRATOR FOR AUTOMATIC SYSTEMS HBA1C 3X2 ML / CFAS HBA1C 3X2MLLOT: 3197360 1 - 1 AMOUNT CALIBRATOR FOR AUTOMATIC SYSTEMS CFAS LOT: 32411601 - 1PCS. SET FOR QUALITY CONTROL PROCEDURE / PRECIPATH PUC LOT: 38363 - 1 AMOUNT SET OF CONTROL SERA FOR TEST FOR DETERMINATION OF SURFACE ANTIGEN OF HEPATITIS B VIRUS LOT: 37896201 - 1 AMOUNT TEST FOR QUANTITATIVE DETERMINATION OF PLASMA PROTEIN A ASSOCIATED WITH AGITATION IN HUMAN SERUM A LOT: 37356201 - 5 AMOUNT CALIBRATE OP FOR AUTOMATIC SYSTEMS CFAS LOT: 3241 1601 - 1PC. CALIBRATOR FOR AUTOMATIC SYSTEMS CFAS PRO
3002109900
1. MEDICAL DEVICES INTENDED BY THE MANUFACTURER FOR IN VITRO USE FOR RESEARCH IN THE LABORATORY TO OBTAIN INFORMATION ON PHYSIOLOGICAL OR PATHOLOGICAL CONDITION: COMPLEX LABORATORY IMMUNOLOGICAL REAGENTS OF COMBINED ORIGIN OBTAINED USING BIOTECHNOLOGICAL PROCESSES: REAGENTS COMPLEX (ANALYTICAL) INTENDED FOR USE IN BLOOD ANALYSIS OMNI C ROCHE OMNI S COBAS B 121 COBAS B 221 LOT: 21492111- 1 PC.
3002109900
1. MEDICAL PRODUCTS DESIGNED VYROBNYKOMDLYA USE FOR IN VITRO RESEARCH LABORATORY FOR OBTAINING IN INFORMATSIYISTOSOVNO PHYSIOLOGICAL OR PATHOLOGICAL CONDITION SOPHISTICATED LABORATORY IMMUNOLOGIC COMBINED REAGENTS ORIGIN DERIVED FROM USING BIOTEHNOLOHICHNYHPROTSESIV REAGENTS COMPLEX (THINK) DESIGNED TO ANALYZE KROVIZ USE ENZYME IMMUNO-ANALYZER COBAS E411: TEST SYSTEM FOR DETERMINATION OF ANTIBODIES TO SURFACE ANTIGENE OF HEPATITIS IN ANTI-HBS ELECSYS COBAS E 100 LOT: 43369003 - 1 ? ?.
LLC TINITERIA MED

UKRAINE
HS-Code
Products
3002109900
1. MEDICAL DEVICES. SETS OF VITASSAY REAGENTS FOR DIAGNOSTICS IN VITRO IN MEDICAL LABORATORIES.-ART.7355020 VITASSAY H.PYLORI (25 TESTS IN A SET) -133 SETS; -ART.7355034 VITASSAY GIARDIOSIS (GIARDIASIS) (25 TESTS IN A SET) - 59 SETS; -ART.7355008 VITASSAY ROTAVIRUS (25 TESTS IN A SET) -2 SETS; -ART.7355038 VITASSAY STREPT A (25 TESTS IN A SET) -2SET; -ART.7355004 VITASSAY CALPROTECTIN (25 TEST SETS) -1 SET; -ART.7355001 VITASSAY FOB BLOOD IN FECES (25 TESTS IN A SET) - -1 SET. THE GOODS ARE NOT GMO AND ARE NOT MADE WITH USE OF GMO. THE VITASSAY TRADEMARK IS MADE BY VITASSAY HEALTHCARE SLU THE COUNTRY OF PRODUCTION ES
3002109900
1. PRODUCTS OF MEDICAL PURPOSE. REAGENTS FOR DIAGNOSTICS IN VITRO IN MEDICAL LABORATORIES. SETS OF REAGENTS FOR THE ENZYME IMMUNOSORBENT ANALYSIS OF ELISA: - ART. K6923 IDK SET HELICOBACTER PYLORI ELISA ANTIGEN - 9 PIECES; - ART. K6915 IDK SET - PANCREATIC; .K6967 IDK SET CALPROTECTIN ELISA -32PCS; -ART.K8500 IDK DAO SET (DIAMINE OXIDASE) ELISA -2PCS; -ART.K5601 IDK SET ZONULIN ELISA -1PC; -ART.KIF005 SET ID-VIT FOLIC ACID -1PC. GMOS BUT NOT PRODUCED WITH THE USE OF GMOS. TRADEMARK IMMUNDIAGNOSTIKMANUFACTURER IMMUNDIAGNOSTIK AGCOUNTRY OF PRODUCTION DE
3002109900
1. SETS OF REAGENTS FOR LABORATORY DIAGNOSTICS IN VITRO USED FOR RESEARCH ON ANALYZERS IN IMMUNOASSAY IN THE SYSTEM: -ART. 07P4820 SET OF REAGENTS ALINITY AND TSH REAGENT KIT - 5 PIECES - AN ART. 07P5120 SET OF REAGENTS ALINITY AND TOTAL-HCG REAGENT KIT -7PCS. BRAND: ALINITYCOUNTRY OF PRODUCTION: IEMANUFACTURER: ABBOTT IRELAND DIAGNOSTICSDIVISION LISNAMUCK LONGFORD CO.LONGFORD IRELAND
LLC INTELMED

UKRAINE
HS-Code
Products
3002101000
1.DIAGNOSTIC REAGENTS USED IN SYSTEMS FOR CLINICAL CHEMICAL AND IMMUNOLOGICAL TESTS FOR HOSPITAL LABORATORIES - ANTISERA WHICH ARE INTENDED ONLY FOR DIAGNOSIS IN VITRO NOT ON A SUBSTRATE CONTAIN CONTROL MATERIAL PACKAGED IN DISPOSABLE BOTTLES IN THE RANGE ( ONLY 1 PIECE SET): DM CHEM I CALIBRATOR DC18C CATALOG NUMBER DC18C - 1 PIECE ARTICLE: DC18C BRAND: SIEMENS . MANUFACTURER: SIEMENS HEALTHCARE DIAGNOSTICS INC. 511 BENEDICT AVENUE TARRYTOWN NEW YORK 10591 COUNTRY OF ORIGIN: US (UNITED STATES). COUNTRY OF ORIGIN: USA
3002101000
1.DIAGNOSTIC REAGENTS USED IN SYSTEMS FOR CLINICAL CHEMICAL AND IMMUNOLOGICAL TESTS FOR HOSPITAL LABORATORIES - ANTISERA WHICH ARE INTENDED ONLY FOR DIAGNOSIS IN VITRO NOT ON A SUBSTRATE CONTAIN CONTROL MATERIAL PACKAGED IN DISPOSABLE BOTTLES IN THE RANGE ( ONLY 1 PIECE SET): ALB / TP CALIBRATOR 6X2ML / PK CATALOG NUMBER DC31 - 1 PIECE ARTICLE: DC31 TRADEMARK: SIEMENS . MANUFACTURER: SIEMENS HEALTHCARE DIAGNOSTICS INC. 511 BENEDICT AVENUE TARRYTOWN NEW YORK 10591 USA. COUNTRY OF ORIGIN: US (USA). COUNTRY OF ORIGIN: USA
3002101000
1. DIAGNOSTIC REAGENTS USED IN SYSTEMS FOR CLINICAL CHEMICAL AND IMMUNOLOGICAL TESTS FOR HOSPITAL LABORATORIES - ANTISERA WHICH ARE INTENDED ONLY FOR DIAGNOSIS IN VITRO NOT ON A SUBSTRATE CONTAIN CONTROL MATERIAL PACKAGED IN DISPOSABLE BOTTLES IN THE RANGE (TOTAL 62 PIECES OF SETS): IML.2000 ACTH KIT 200T CATALOG NUMBER L2KAC2 - 1 PIECE. ARTICLE: L2KAC2IML.2000 ANTI-TG KIT 200T CATALOG NUMBER L2KTG2 - 2PCS. ARTICLE: L2KTG2IML.2000 ANTI-TPO KIT 200T CATALOG NUMBER L2KTO2 - 18PCS. ARTICLE: L2KTO2IMMULITE 2000 CALCITONIN KIT 200T CATALOG NUMBER L2KCL2 - 1PC. ARTICLE: L2KCL2IMMULITE 2000 FERRITIN KIT 200T CATALOG NUMBER L2KFE2 - 3PCS. ARTICLE: L2KFE2IML.2000 FOLIC ACID KIT 200T CATALOG NUMBER L2KFO2 - 1PC. ARTICLE: L2KFO2IML2000 FSH 200T CATALOG NUMBER L2KFS2 - 2PCS. ARTICLE: L2KFS2IML.2000 INSULIN KIT 200T CATALOG NUMBER L2KIN2 - 5PCS. ARTICLE: L2KIN2IML2000 LH 200T CATALOG NUMBER L2KLH2 - 2PCS. ARTICLE: L2KLH2IML2000 PROGES
MEDICOR LTD LTD

UKRAINE
HS-Code
Products
3002109900
1. RELATED MATERIALS FOR CONDUCTING CLINICAL TRIALS ACCORDING TO THE MIN-101C07 PROTOCOL. LABORATORY DIAGNOSTIC REAGENTS CONTAINING MODIFIED IMMUNOBIOLOGICAL PRODUCTS (MONO AND POLYCLONAL ANTIBODIES): - TESTS TO DETERMINE PREGNANCY QUICK VUE HCG (BLUE / WHITE) - 84 PCS .; DOES NOT CONTAIN GMOS. DOES NOT CONTAIN PRODUCTS OF ANIMAL ORIGIN NOT FOR VETERINARY MEDICINE. NOT FOR SALE THE PRODUCT IS IMPORTED EXCLUSIVELY FOR CLINICAL TRIALS.MANUFACTURER: QUIDEL; COUNTRY OF MANUFACTURE: US;
3002109100
1. PEOPLE DRUG FOR PACKAGED RETAIL CONTAINING GLOBULIN: - OKTAHAM 10% INFUSION SOLUTION 10% 20 ML SOLUTION FOR INFUSION IN BOTTLES 1 BOTTLE IN A CARDBOARD BOX. COMPOSITION ACTIVE SUBSTANCE: 1 ML SOLUTION FOR INFUSION CONTAINS: 100 MG OF TOTAL PROTEIN CONSISTING OF IMMUNOGLOBULIN? 95% K925V8542 SERIES - 397UPAK. IS INCORPORATES CULTURE OF MICROORGANISMS ANIMAL PRODUCTS NARCOTIC DRUGS AND PSYCHOTROPIC SUBSTANCES NOT IN AEROSOL UPAKOVTSI.VYROBNYK: OCTAPHARMA PHARMAZEUTIKA PRODUKTIONSGES.MBH AUSTRIA
3002109900
1. RELATED MATERIALS FOR CONDUCTING CLINICAL TRIALS ACCORDING TO THE MIN-101C07 PROTOCOL. LABORATORY DIAGNOSTIC REAGENTS CONTAINING MODIFIED IMMUNOBIOLOGICAL PRODUCTS (MONO AND POLYCLONAL ANTIBODIES): - TESTS TO DETERMINE PREGNANCY QUICK VUE HCG (BLUE / WHITE) - 30 PCS .; DOES NOT CONTAIN PRODUCTS OF ANIMAL ORIGIN NOT FOR VETERINARY MEDICINE. DOES NOT CONTAIN GMOS. NOT FOR SALE THE PRODUCT IS IMPORTED EXCLUSIVELY FOR CLINICAL TRIALS.MANUFACTURER: QUIDEL; COUNTRY OF MANUFACTURE: US;
LLC SMO GROUP UKRAINE

UKRAINE
HS-Code
Products
3002109900
1. IMMUNOMODULATORY DRUGS ARE NOT REGISTERED (NOT IN AEROSOL PACKAGING) FOR HUMANS IN DOSAGE FORM NOT FOR SALE NOT OF ANIMAL ORIGIN FOR CLINICAL TRIALS ACCORDING TO THE RESEARCH PROTOCOL SAMSON-II: - HD204 / AVASTIN 400 MG / 16 ML (25 MG / ML) SOLUTION FOR INTRAVENOUS INFUSION IN A VIAL ?1 - 240 PCS .; PACKAGING SERIES: 366868-01. SHELF LIFE: 03 / 2020.MANUFACTURER - CATALENT PHARMA SOLUTIONS (CATALENT GERMANY SCHORNDORF GMBH.) TRADEMARK - HD204 / AVASTIN. COUNTRY OF PRODUCTION DE.
3002109900
1. IMMUNOMODULATORY DRUGS ARE NOT REGISTERED (NOT IN AEROSOL PACKAGING) FOR PEOPLE INTENDED FOR CLINICAL TRIALS UNDER THE PROTOCOL NN7415-4310 NOT FOR RETAIL NON-ANIMAL ORIGIN.- CONCIZUMAB B - SOLUTION FOR INJECTION 100 MG / ML IN CARTRIDGES OF 3 ML - 3 PIECES PACKAGING SERIES: GLDG014 PRODUCTION SERIES: B000134556 SHELF LIFE: 28.11.2019.- CONCIZUMAB B - SOLUTION FOR INJECTION OF 100 MG / ML IN A CARTRIDGE OF 3 ML - 27 PIECES PACKAGING SERIES: HLDG023 PRODUCTION SERIES: B000143035. SHELF LIFE: 10.10.2020. MANUFACTURER: NOVO NORDISK A / S. TRADEMARK - NOVO NORDISK. COUNTRY OF MANUFACTURE DK.
3002109900
1. ACCOMPANYING MATERIALS BASED ON MODIFIED IMMUNOLOGICAL PRODUCTS FOR MULTICENTER CLINICAL TRIALS ACCORDING TO PROTOCOL1 36 33. PRODUCTS OF GENERAL LABORATORY PURPOSE NON-ANIMAL ORIGIN: - TEST FOR URINE ANALYSIS FOR PREGNANCY (FOR DETERMINATION OF CHORIONIC HONODONOTH). MANUFACTURER: SA SCIENTIFIC LTD. TRADEMARK: SAS. MANUFACTURING COUNTRY: US.
PISI UKRAINE LLC

UKRAINE
HS-Code
Products
3002109900
1. MODIFIED IMMUNOBIOLOGICAL PRODUCTS (MONO- AND POLYCLONAL ANTIBODIES) DEPOSITED ON TEST STRIPS THE PRINCIPLE OF WHICH IS BASED ON THE REACTION OF ANTIGEN-BODY: TEST STRIPS FOR VAHITNOSTI25 DEFINITIONS MIU / ML (50 PIECES PER PACK) - ANIMAL PRODUCTS CONTAINING 30UPAKOVOK.NE ORIGIN. ONLY FOR USE IN CLINICAL ACTIVITIES UNDER AC-058B303 PRODUCT. MANUFACTURER: SURESCREEN DIAGNOSTICS LTD. COUNTRY OF MANUFACTURING: MANUFACTURING.
3002109900
1. MODIFIED IMMUNOBIOLOGICAL PRODUCTS (MONO- AND POLYCLONAL ANTIBODIES) APPLIED TO TEST STRIPS THE PRINCIPLE OF OPERATION AS THEY ARE BASED ON THE ANTIGEN-BODY REACTION. TEST KIT FOR ONE TEST TO DETERMINE PREGNANCY SAS ONE-STEP PREGNANCY FOR ONE PREGNANCY TEST -5 PACKAGES. FOR USE IN CLINICAL TRIALS ACCORDING TO THE RPC01-3202 PROTOCOL. MANUFACTURER: SA SCIENTIFIC LTD BRAND: SAS. COUNTRY OF MANUFACTURE: US.
3002109900
1. MODIFIED IMMUNOBIOLOGICAL PRODUCTS (MONO- AND POLYCLONAL ANTIBODIES) DEPOSITED ON TEST STRIPS THE PRINCIPLE OF WHICH IS BASED ON THE REACTION OF ANTIGEN-TILO.TEST SET FOR ONE TESTUNA PREGNANCY SAS ONE-STEPPREGNANCY FOR ODNOHOTESTU FOR PREGNANCY -10 PACKAGING. FOR USE IN THE FRAMEWORK OF CLINICAL TRIALS ACCORDING TO THE RPC01-3202 PROTOCOL. MANUFACTURER: SA SCIENTIFIC LTD TRADEMARK: SAS.
MEDKOSVISS LLC (UKRAINE)

UKRAINE
HS-Code
Products
3002109500
1.SETS OF REAGENTS FOR DIAGNOSTIC MEDICAL TESTS USED TO CONDUCT RESEARCH IN A CONTROL ENVIRONMENT OUTSIDE THE HUMAN BODY.BIO-KSEL SYSTEM TT BIO-KSEL SYSTEM FIBRYNOGEN BIO-KSEL SYSTEM CALIBRATORBIO-KSEL SYSTEM NORMAL BIO-KSEL SYSTEM ABNORMAL L BIO-KSEL SYSTEM ABNORMAL H INVITRO
3002109500
1. SETS OF REAGENTS FOR DIAGNOSTIC MEDICAL TESTS USED FOR RESEARCH IN A CONTROL ENVIRONMENT OUTSIDE THE HUMAN BODY. BIO-KSEL PT PLUS
3002109900
1. SETS OF DIAGNOSTIC REAGENTS MEDICAL TESTS USED FOR RESEARCH IN A CONTROL ENVIRONMENT OUTSIDE THE HUMAN BODY. BIO-KSEL PT PLUS BIO-KSEL SYSTEM TTIN IN VITRO
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
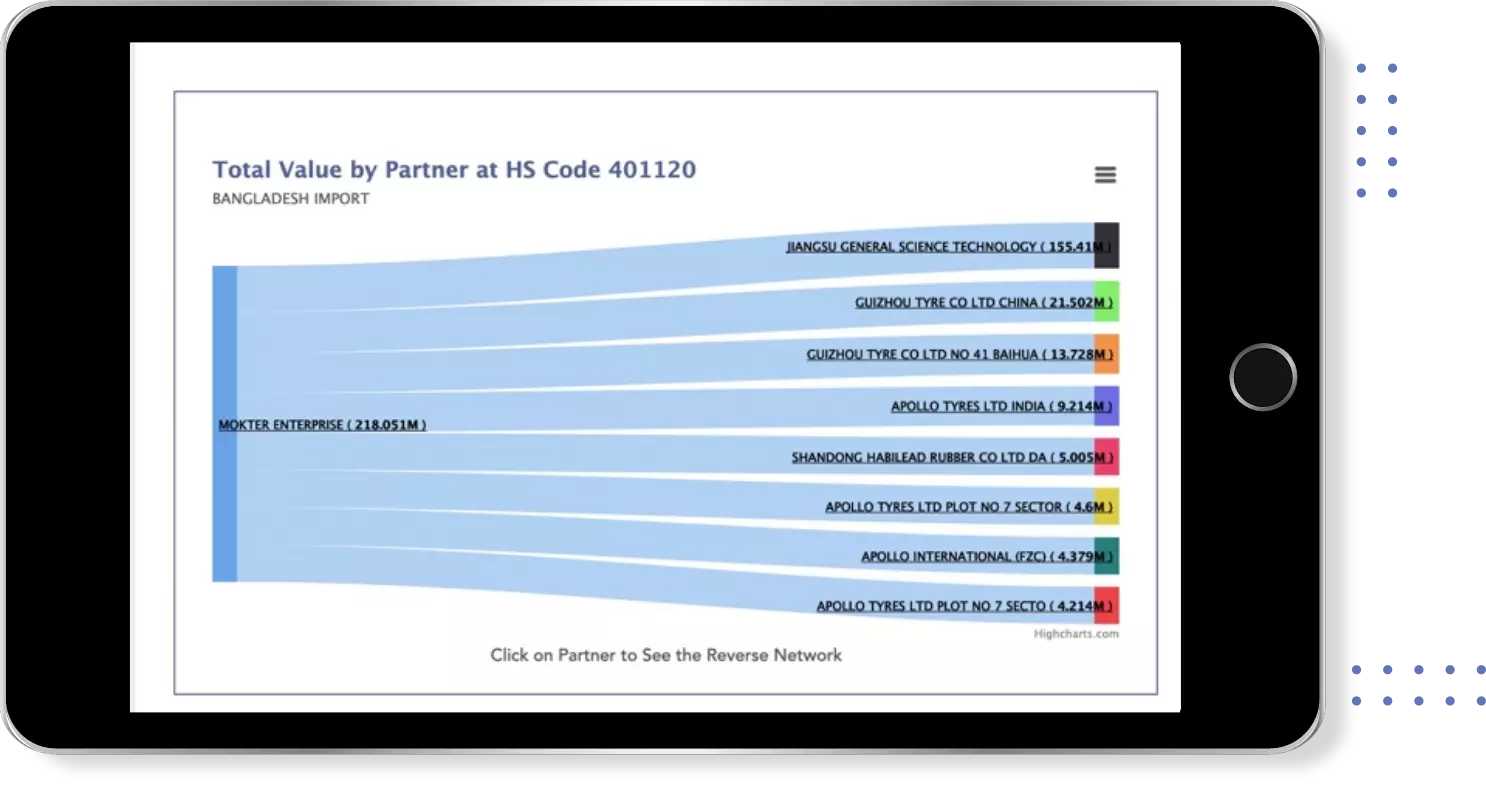
Get most profitable Buyers & Suppliers
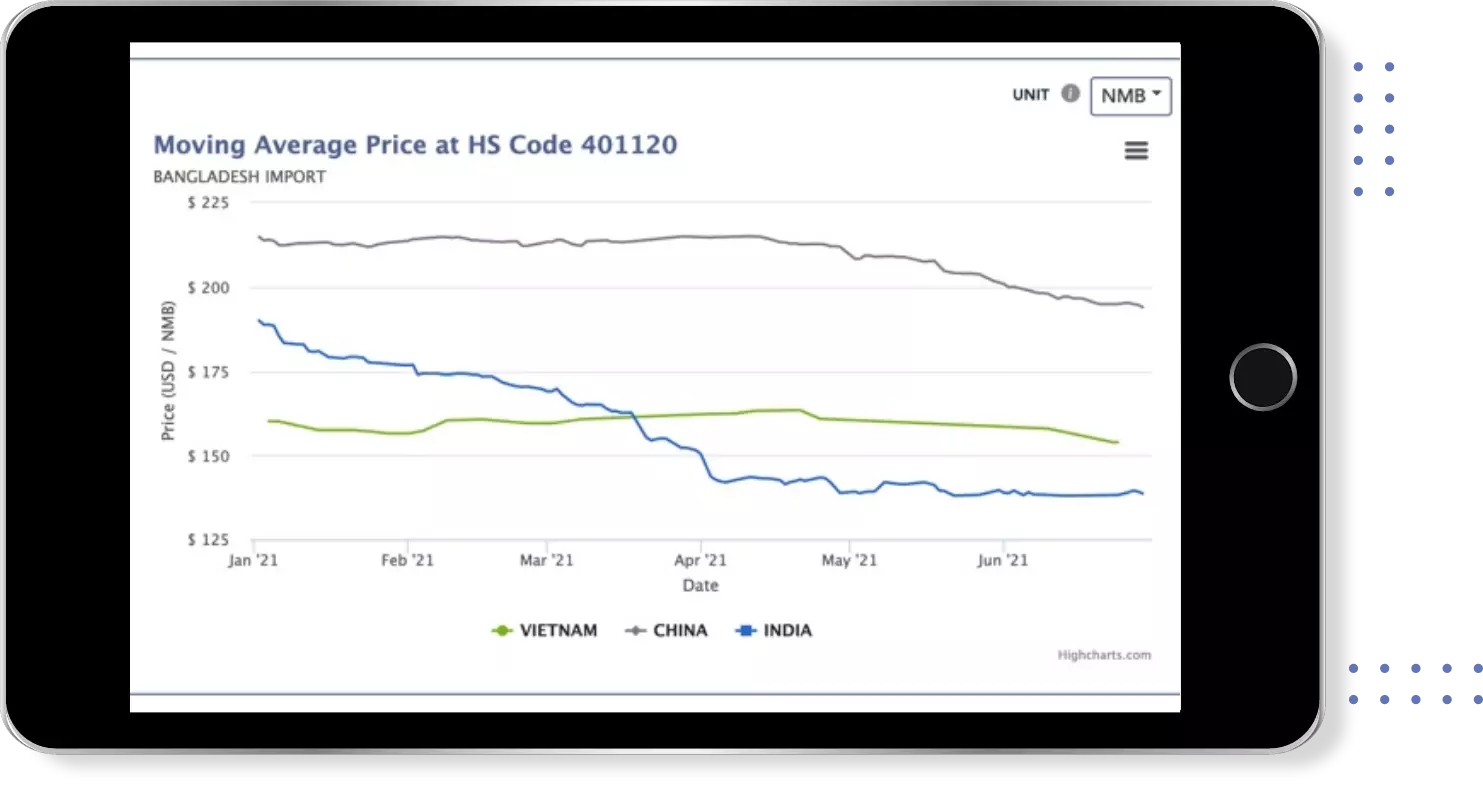
Instant insights on Price , Quantity & Value trends
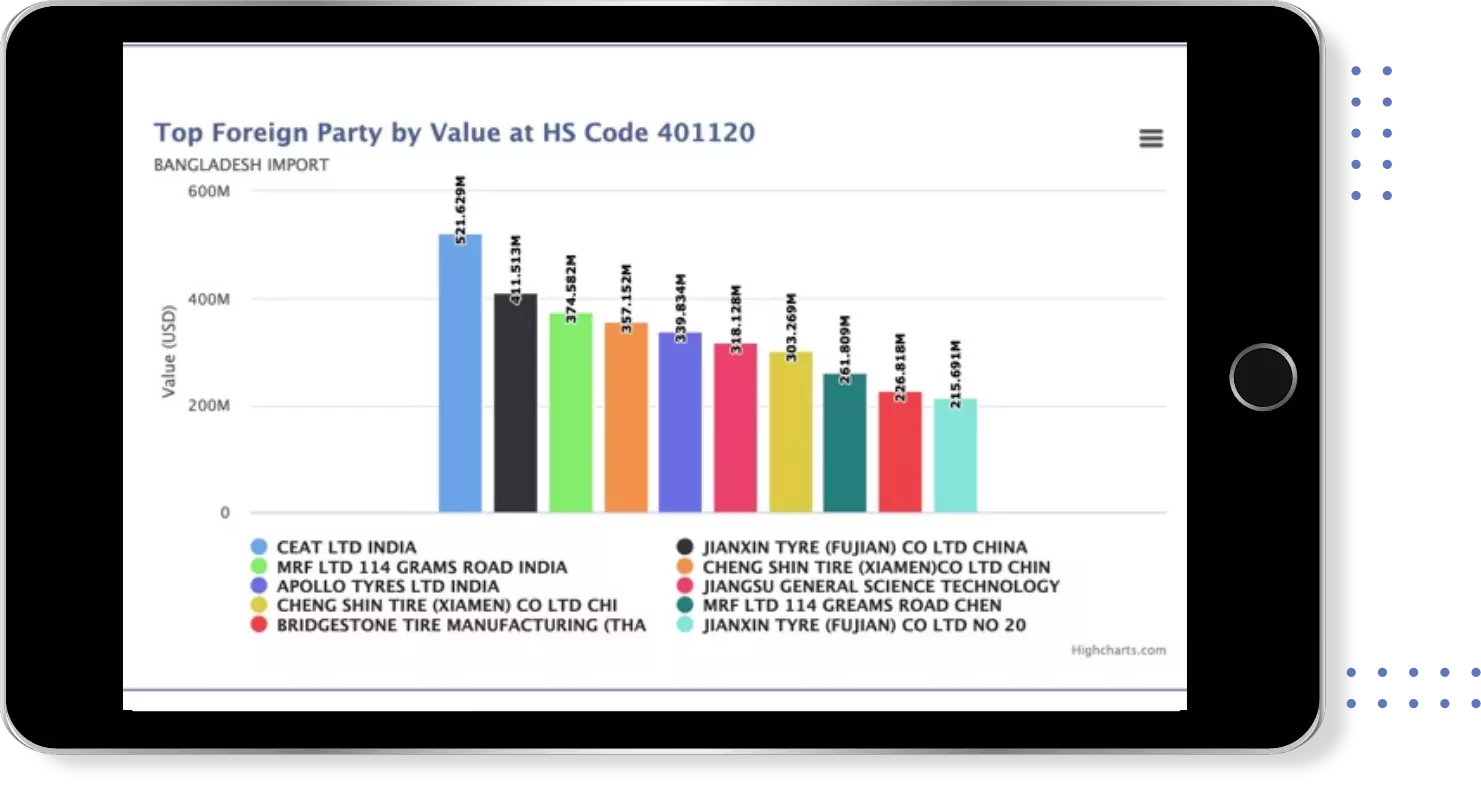
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.