Find verified buyers and sellers of ha tablet in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- ha tablet hs code 300490 buyers in vietnam
- HS Code: 300490
- COD Vietnam
Vietnam Buyers of ha tablet under HS-Code 300490
BACH KHANG MEDICINE COMPANY LTD

VIETNAM
HS-Code
Products
3004908900
RADIOPLASTY ISOTOPE I-131 (SODIUM IODIDE NA131 I) OPEN SOURCE HARD CAPSULES 12 TABLETS 100MCI/TABLET 1MCI = 1 UNIT USED FOR SCREENING DIAGNOSIS TREATMENT OF THYROID CANCER TREATMENT MANUFACTURER: POLATOM
3004908900
RADIOISOTOPE IODINE-131 (SODIUM IODIDE NA131 I) THE SOURCE OPEN HARD CAPSULES 12 TABLETS 100MCI / PARK 1MCI = 1 UNIT FOR SCREENING DIAGNOSIS AND TREATMENT OF DISEASES OF THYROID CANCER MANUFACTURER: POLATOM
3004908900
RADIOACTIVE ISOTOPE I-131 (SODIUM IODIDE NA131 I) OPEN SOURCE HARD CAPSULE 8 TABLETS 50MCI / TABLET 1MCI = 1 UNIT USED TO SCREEN DIAGNOSE AND TREAT THYROID CANCER MANUFACTURER: POLATOM
SOHACO TRADING AND PHARMACEUTICAL GROUP JOINT STOCK COMPANY

VIETNAM
HS-Code
Products
3004909900
CANDID V3. 890100075723. (VN-19660-16). THE MEDICINE HAS ANTIFUNGAL EFFECTS CONTAINING CLOTRIMAZOLE 200MG. BOX OF 1 BLISTER X 3 TABLETS WITH 1 INSERT; VAGINAL TABLETS. LOT NUMBER: 10232623. MANUFACTURER: OCTOBER 13 2023. EXPIRY DATE: OCTOBER 12 2027
300490590000
FEXO 60 TABLET. VN-23182-22. THE MEDICINE CONTAINS FEXOFENADINE HYDROCHLORIDE 60MG WHICH HAS ANTI-ALLERGIC EFFECTS. BOX OF 5 BLISTERS X 10 FILM-COATED TABLETS. LOT NUMBER: 3F03090;3G03259. PRODUCTION: JULY 2023. EXPIRY DATE: JUNE 2025
3004909900
PANTONIX 40 TABLETS. VN-19102-15. THE MEDICINE CONTAINS PANTOPRAZOLE (IN THE FORM OF PANTOPRAZOLE SODIUM SESQUIHYDRATE) 40MG WHICH HAS EFFECTS ON THE DIGESTIVE TRACT. BOX OF 5 BLISTERS X 10 TABLETS. LOT: 23073. MANUFACTURER: OCTOBER 2023. EXPIRY DATE: SEPTEMBER 2026. FOC
DKSH PHARMA VIETNAM CO LTD

VIETNAM
HS-Code
Products
30049099
NEW. MEDICINE: LIPANTHYL 200M (FENOFIBRATE 200MG). HARD CAPSULES. BOX OF 2 BLISTERS X 15 TABLETS. LOT: 33782. PRODUCER: OCTOBER 12 2022. HD: 10/2025. VN-17205-13. MANUFACTURER: RECIPHARM FONTAINE.
30049099
NEW MEDICINE: HAIBLOK (BISOPROLOL HEMIFUMARATE 5MG). BOX OF 3 BLISTERS X 10 TABLETS FILM COATED TABLETS. LOT: 25B014. PRODUCER: 02/2023. HD: 02/2025. VN-19731-16. MANUFACTURER: PT. NOVELL PHARMACEUTICAL LABORATORIES.
3004908900
NEW MEDICINE: COZAAR XQ 5MG/50MG. FILM-COATED TABLETS. BOX OF 3 BLISTERS X 10 TABLETS. LOT: Y002784; Y003034. MANUFACTURER: SEPTEMBER 13 2023. HD: SEPTEMBER 13 2025. REGISTRATION NUMBER: VN-17524-13. CSSX: HANMI PHARM.CO. LTD (PALTAN SITE) - KOREA.
BACH KHANG PHARMACEUTICAL COMPANY LTD

VIETNAM
HS-Code
Products
3004908900
RADIOISOTOPE I-131 (SODIUM IODIDE NA131 I) OPEN SOURCE HARD CAPSULE 20 TABLETS 50MCI/TABLET 1MCI=1 UNIT USED FOR SCREENING DIAGNOSIS AND TREATMENT OF THYROID CANCER MANUFACTURER: POLATOM
3004908900
RADIOISOTOPE I-131 (SODIUM IODIDE NA131 I) OPEN SOURCE HARD CAPSULE 9 TABLETS 100MCI/TABLET 1MCI=1 UNIT USED FOR SCREENING DIAGNOSIS AND TREATMENT OF THYROID CANCER MANUFACTURER: POLATOM
3004908900
RADIOISOTOPE I-131 (SODIUM IODIDE NA131 I) OPEN SOURCE HARD CAPSULE 9 TABLETS 50MCI/TABLET 1MCI=1 UNIT USED FOR SCREENING DIAGNOSIS AND TREATMENT OF THYROID CANCER PATHOLOGIES MANUFACTURER: POLATOM
GEDEON RICHTER VIETNAM COMPANY LTD

VIETNAM
HS-Code
Products
300490990000
REAGILA 3 MG (CARIPRAZINE (AS CARIPRAZINE HYDROCHLORIDE) 3MG) HARD CAPSULES. BOX OF 3 BLISTER PACKS X 10 TABLETS. BATCH: U36007B. PRODUCTION: JUNE 2023. EXPIRY DATE: JUNE 2026. REGISTRATION NUMBER: VN3-370-21. NEW 100%. FOC GOODS
3004909900
REAGILA MEDICINE 6MG (CARIPRAZINE (AS CARIPRAZINE HYDROCHLORIDE) 6MG) HARD CAPSULE. BOX OF 3 BLISTERS X 10 TABLETS. BATCH: U2B010B. PRODUCER: 11/2022. HSD: 11/2025. PHONE NUMBER: VN3-372-21. NEW 100%. FOC GOODS
3004909900
REAGILA MEDICINE 4.5MG (CARIPRAZINE (AS CARIPRAZINE HYDROCHLORIDE) 4.5MG) HARD CAPSULE. BOX OF 3 BLISTERS X 10 TABLETS. BATCH: U31011E. PRODUCER: 01/2023. HSD: 01/2026. PHONE NUMBER: VN3-371-21. NEW 100%. FOC GOODS
SOHACO GROUP JSC

VIETNAM
HS-Code
Products
3004909900
MALDIV TAB. VN-20963-18. MEDICINES CONTAINING 100MG TRIMEBUTIN MALEATE HAVE AN EFFECT ON THE GASTROINTESTINAL TRACT. BOX OF 10 BLISTERS X 10 TABLETS. LOT NUMBER: EU001. NSX: 06/07/2021. HSD: 05/07/2024. NEW 100%. FOC
3004908900
EBITAC 25. VN-17349-13. THE DRUG HAS THE EFFECT OF TREATING HYPERTENSION CONTAINING ENALAPRIL MALEATE 10 MG; HYDROCHLOROTHIAZIDE 25MG. BOX OF 2 BLISTERS X 10 TABLETS. LOT NUMBER: ((37+38+39+41) 0522). NSX: 05/2022. HSD: 05/2024
3004908900
SITAGIL 50.VN-22753-21. SITAGLIPTIN CONTAINERS (AS SITAGLIPTIN PHOSPHATE MONOHYDRATE) 50MG HAS A DIABETES TREATMENT EFFECT. 3 BLISTERS X10 TABLETS FILM.LOT: 21007.NSX: 10/2021. HSD: 09/2023. 100% NEW GOODS
VIATRIS VIETNAM LTD COMPANY

VIETNAM
HS-Code
Products
3004905900
CELEBREX (CELECOXIB 200MG); HARD CAPSULES; BOX OF 3 BLISTERS X 10 TABLETS; BATCH: HA8278. PRODUCTION: OCTOBER 2022; HSD: 10/2025. PHONE NUMBER: VN-23247-22. NSX: PFIZER PHARMACEUTICALS LLC
3004909900
LYRICA MEDICINE (PREGABALIN 75MG); BOX OF 4 BLISTER PACKS X 14 TABLETS; HARD CAPSULES.BATCH:HG3729. PRODUCTION: JUNE 2023. EXPIRY DATE: 06/2026. REGISTRATION NUMBER: VN-16347-13. MANUFACTURER: PFIZER MANUFACTURING DEUTSCHLAND GMBH.
3004909900
AMLOR MEDICINE (AMLODIPINE (AS AMLODIPINE BESILATE) 5MG); BOX OF 3 BLISTER PACKS X 10 TABLETS; HARD CAPSULES. BATCH:B851402. PRODUCTION: SEPTEMBER 2023. EXPIRY DATE: SEPTEMBER 2025. REGISTRATION NUMBER: 300110025623 (VN-20049-16). PRODUCER: FAREVA AMBOISE.
DKSH PHARMA

VIETNAM
HS-Code
Products
3004908900
NEW DRUG: LENVIMA 4MG (LENVATINIB (AS LENVATINIB MESILATE) 4MG). HARD CAPSULES. BOX OF 2 BLISTERS X 10 TABLETS. LOT: 11A04K. PRODUCER: AUGUST 24 2020. HD: AUGUST 23 2023.VN3-261-20. NSX: PATHEON INC. - CANADA.
3004909900
PHARMACEUTICALS: DUSPATALIN RETARD (MEBEVERINE HYDROCHLORIDE 200MG). BOX OF 3 BLISTERS X 10 TABLETS; HARD CAPSULES EXTENDED-RELEASE. LOT: 713 595; 714658.VN-21652-19. MANUFACTURERS: MYLAN LABORATORIES SAS.
3004909900
TAN PHARMACY: GAPIVELL (GABAPENTIN 300MG). BOX OF 3 BLISTERS X 10 TABLETS; HARD CAPSULES. LOT: 23B509; 23D157A. NSX: 02; 04/2021. HD: 02; 04/2023. VN-20075-16. MANUFACTURER: PT. NOVELL PHARMACEUTICAL LABORATORIES.
HOANG DUC MEDICAL SUPPLIES CO LTD

VIETNAM
HS-Code
Products
3004909900
CAPSULES 300MG MIRGY MEDICATION (GABAPENTIN 300MG). BOX OF 1 BLISTER X 10 TABLETS HARD CAPSULES. BATCH: 176C08. NSX: 03 / 2021.HSD: 03 / 2024.SDK: VN-17441-13. HSX: GETZ PHARMA (PVT) LTD.
3004909900
MIRGY CAPSULES 100MG (GABAPENTIN 100MG) BOX OF 1 BLISTER X10 TABLETS; HARD CAPSULE.BATCH:288C07.NSX: 12/2020.HSD:12/2023.SDK:VN-17440-13.HSX: GETZ PHARMA (PVT)LTD .
3004909900
MIRGY CAPSULES 400MG (GABAPENTIN 400MG). BOX OF 1 BLISTER X 10 TABLETS; HARD CAPSULE. BATCH NO:147C09. NSX:09/2020.HSD:09/2023.SDK: VN-17442-13. HSX: GETZ PHARMA (PVT) LTD.FOC
HOANG DUC PHARMACEUTICAL AND MEDICAL SUPPLIES CO LTD

VIETNAM
HS-Code
Products
3004909900
FENOGETZ CAPSULES 200MG (FENOFIBRATE 200MG). BOX OF 01 BLISTER X 10 TABLETS; HARD CAPSULES. BACTH: 299C24.NSX: NOVEMBER 2023. EXPIRY DATE: NOVEMBER 2026. TCCL:NSX.REGISTER: 896110408223 (VN-17958-14). HSX: GETZ PHARMA (PVT) LTD
3004909900
MIRGY CAPSULES 100MG (GABAPENTIN 100MG) BOX OF 1 BLISTER X 10 TABLETS; HARD CAPSULES.BATCH: 358C07.NSX:12/2023.HSD:12/2026.TCCL:USP-41.S?K:VN-17440-13.HSX : GETZ PHARMA (PVT)LTD
3004909900
MIRGY CAPSULES 100MG (GABAPENTIN 100MG) BOX OF 1 BLISTER X 10 TABLETS; HARD CAPSULES.BATCH: 358C07.NSX:12/2023.HSD:12/2026.TCCL:USP-41.S?K:VN-17440-13.HSX : GETZ PHARMA (PVT)LTD.FOC
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
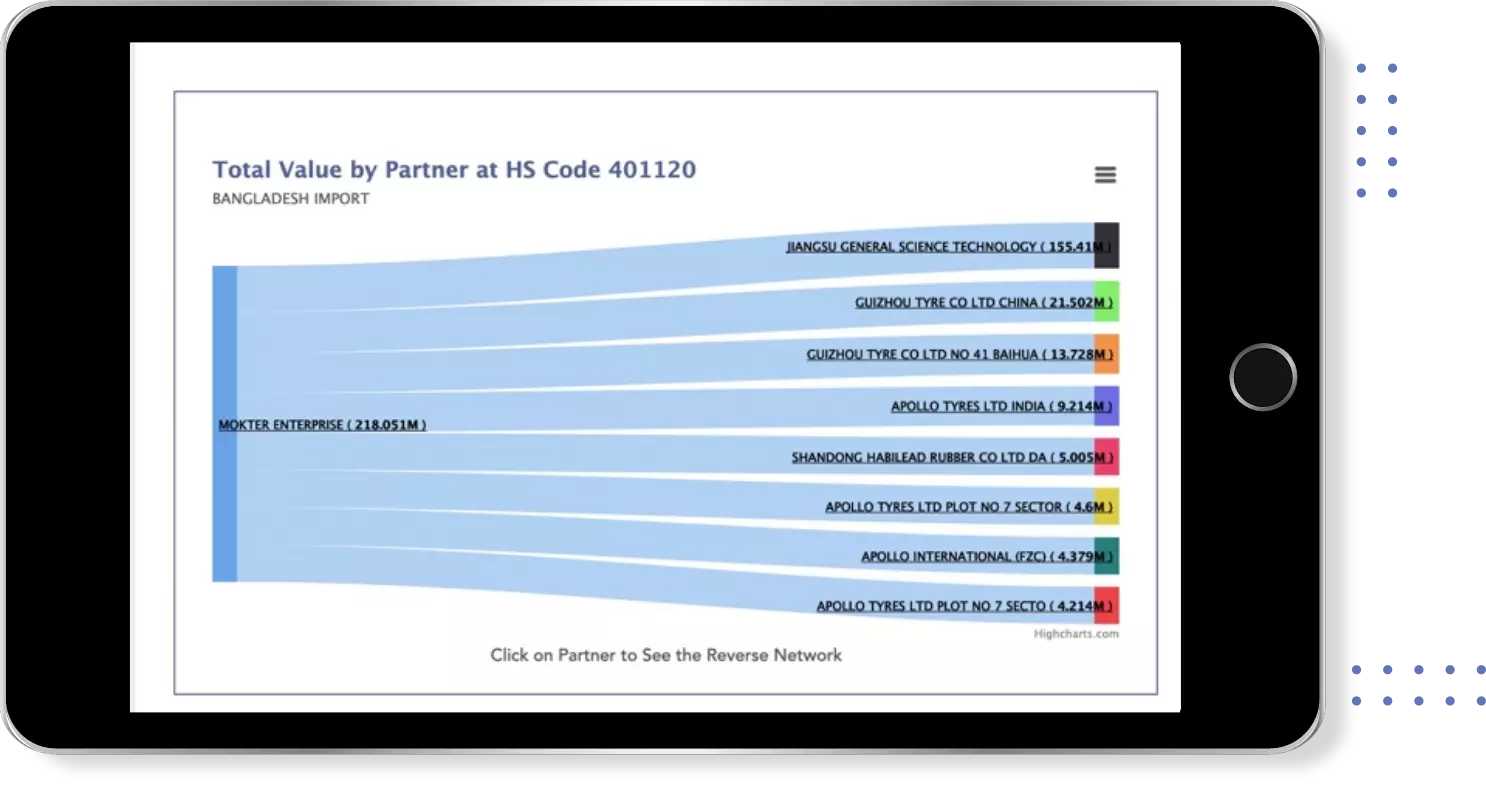
Get most profitable Buyers & Suppliers
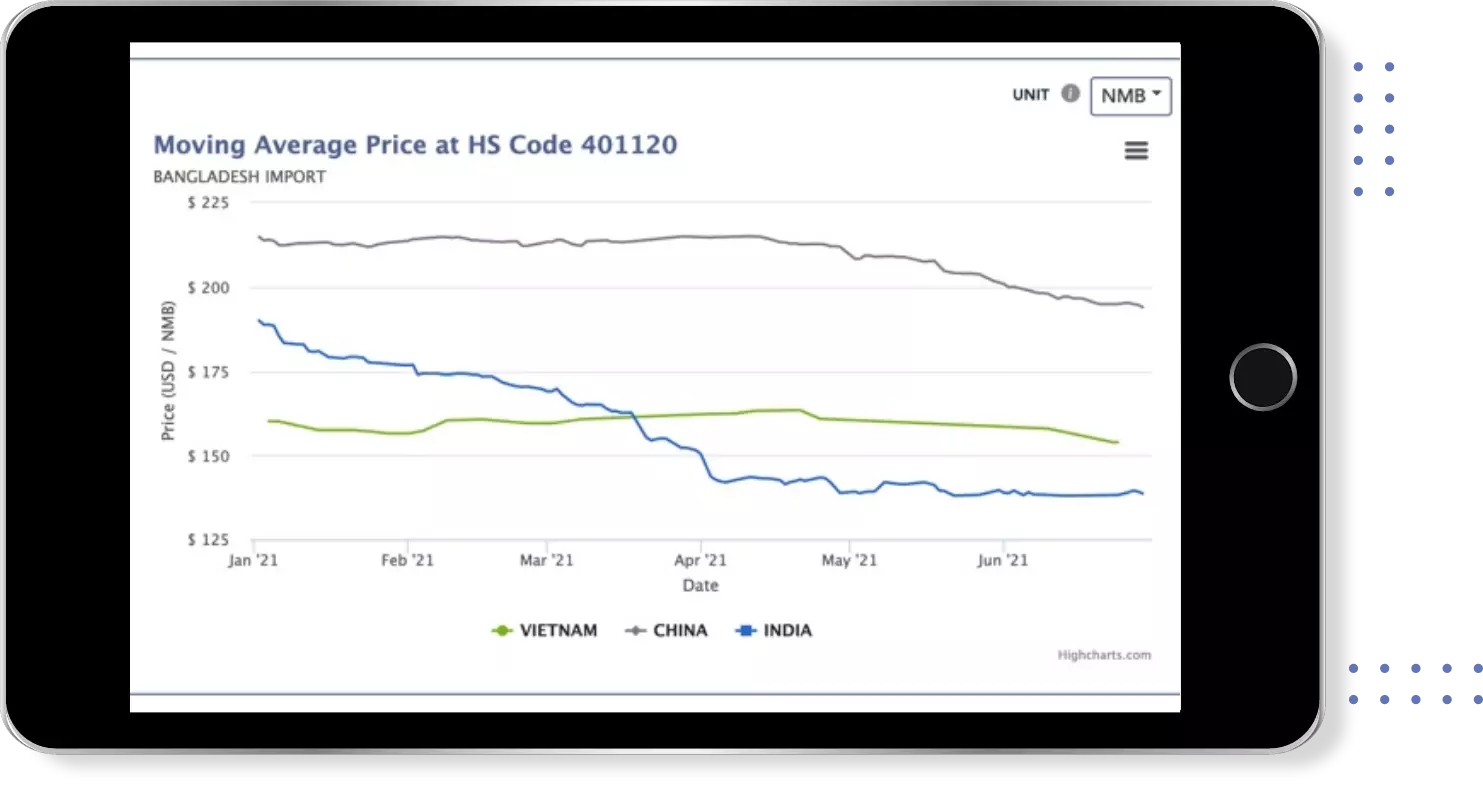
Instant insights on Price , Quantity & Value trends
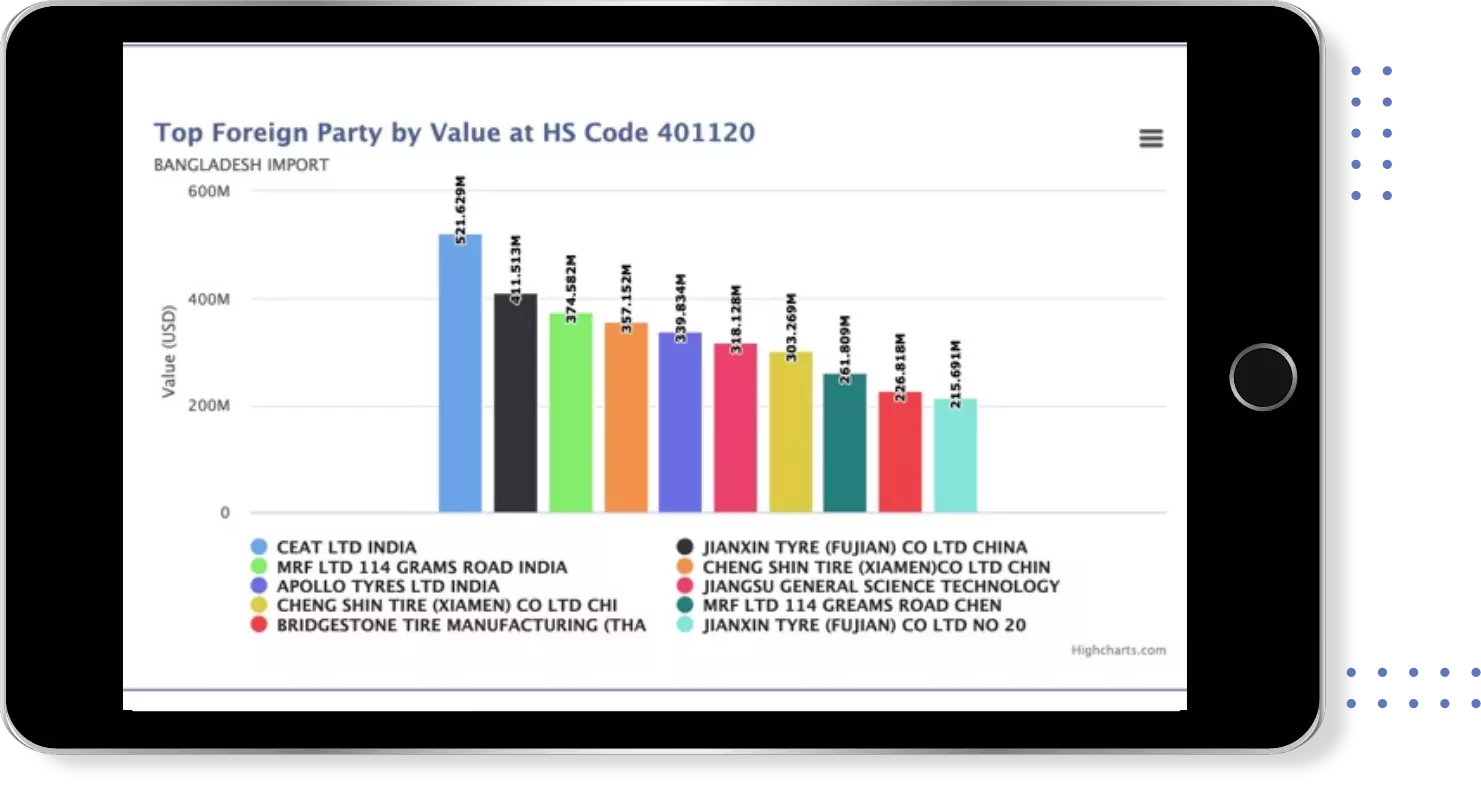
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
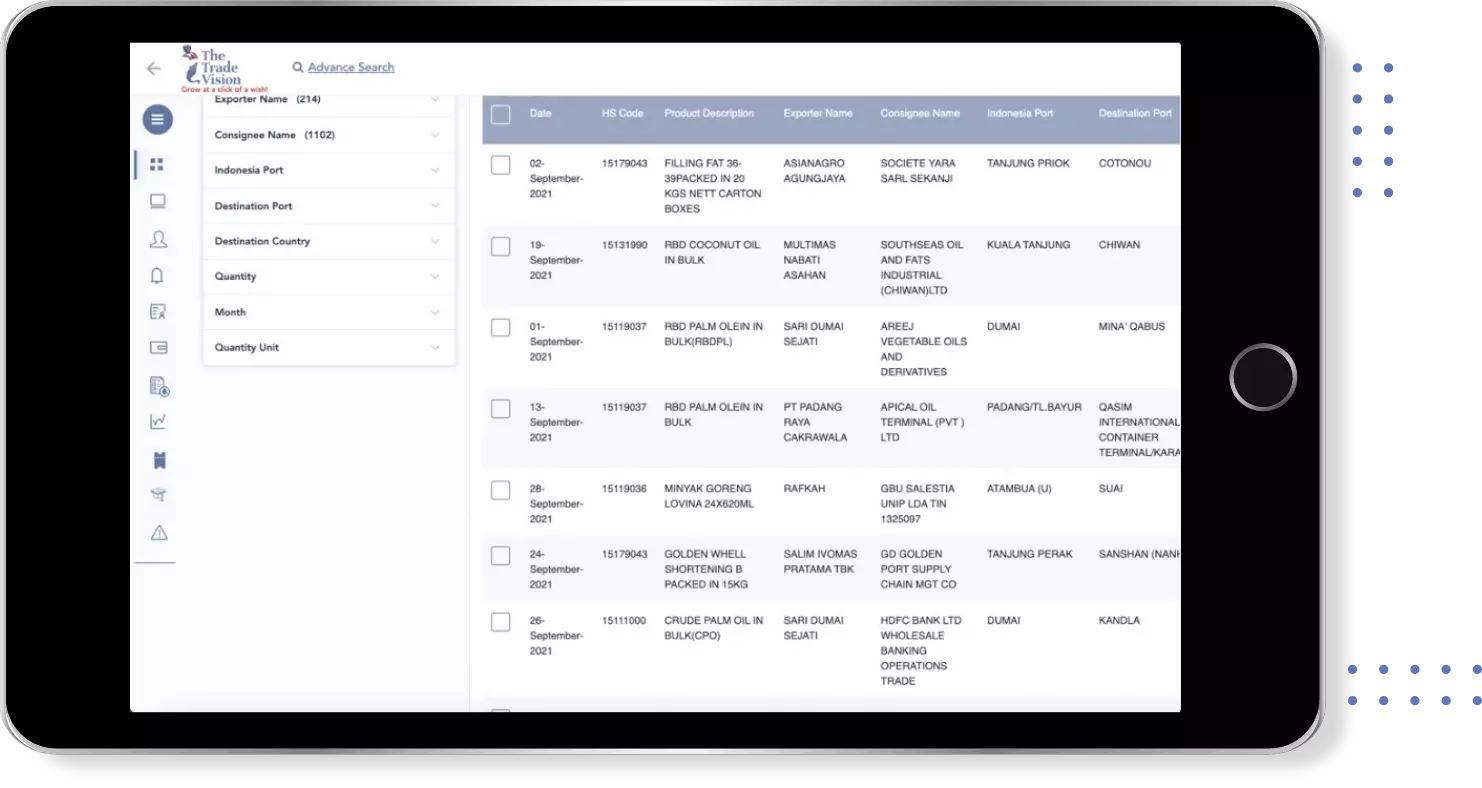
Export Import shipment Data of 180+ Countries
Hear what our clients have to say
We have given over thousands of our clients a reason to be happy with the business results they have gained by using TTV.