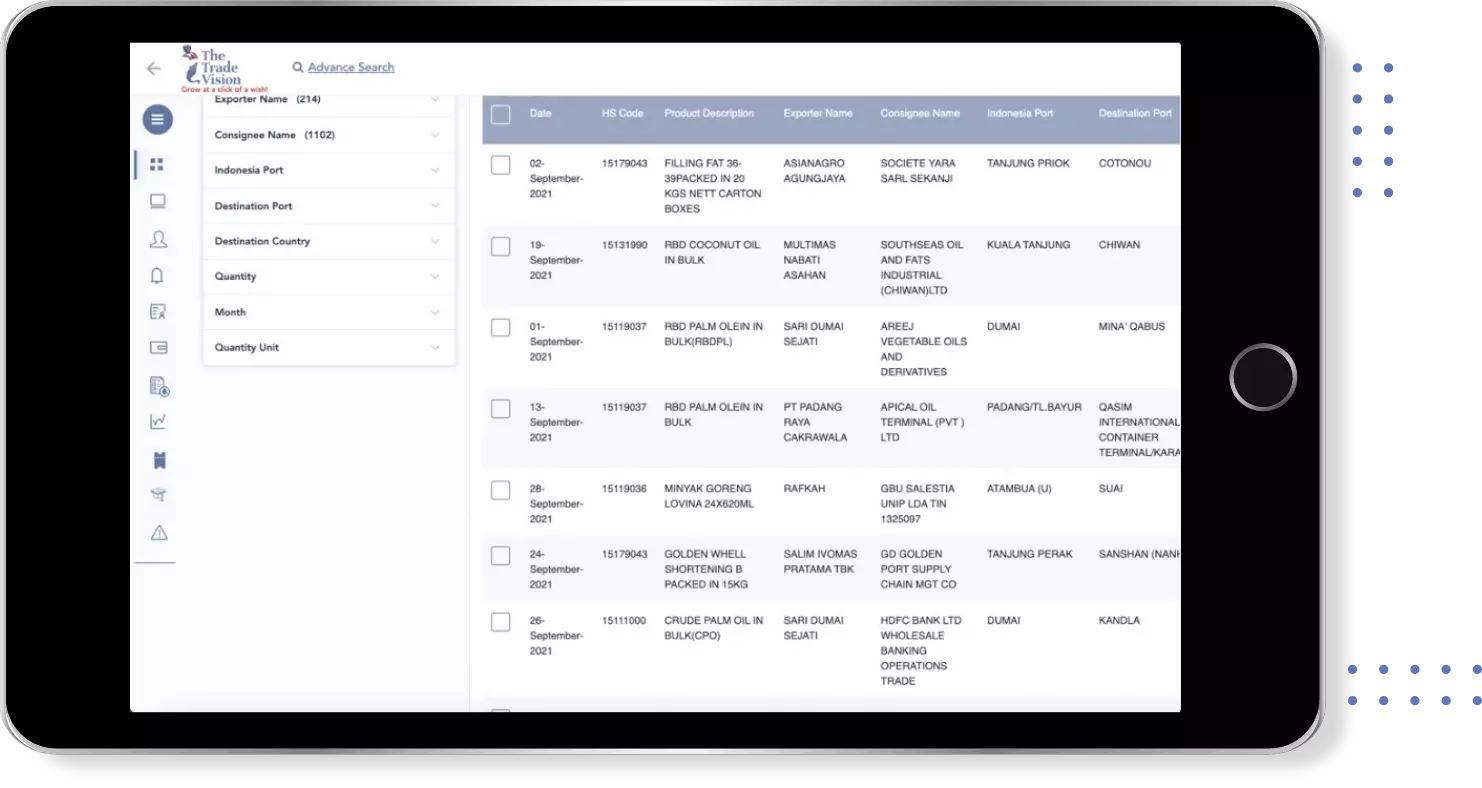
Find verified buyers and sellers of each tablet in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- each tablet hs code 300660 import export data
Overview
- The top 3 supplier countries for HS Code 300660 are “HONG KONG” , “KAZAKHSTAN” , “CHILE” ,
- The top 3 Buyer countries for HS Code 300660 are “SPAIN” , “HUNGARY” , “EUROPEAN UNION” , .
- The top 3 Importing countries for HS Code 300660 are “ZAMBIA” , “TANZANIA” , “HONG KONG” , .
- The top 3 Exporting countries for HS Code 300660 are “HUNGARY” , “SPAIN” , “EUROPEAN UNION” , .
- Top 1 Product Categories for HS Code 300660 are 300660,
The above summary is based on TTV’s Global Export Import data of HS Code 300660, compiled from 180+ countries export import shipments updated till .
- HS Code: 300660
Global Buyers of each tablet under HS-Code 300660
EXELTIS PERU S A C

SPAIN
HS-Code
Products
3006600000
DIENILLE 2MG + 0.03MG EXELTIS S/M IN BOXES - CARTON BOX X 28 COATED TABLETS IN CLEAR ALUMINUM PVC/PVDC BLISTER PACKAGE EACH BLISTER CONTAINS 21 WHITE ACTIVE TABLETS AND 7 GREEN INACTIVE TABLETS CODE 227224 - DIE/EE DIENILLE 2-0 03 1X21+7 EXELTIS PE REG. SANITARY NO. EE-04560 - LOT NO. LF30091D-FCH. OF MANUFACTURING 03-02-2023 / 03-31-2023 // EXP. DAT
3006600000
ANGIE 20 2MG/0.02 MG COATED TABLET EXELTIS S/M IN CARTON BOXES COATED TABLETS EACH BLISTER CONTAINS 24 RO COLORED ACTIVE TABLETS ORAL CONTRACEPTIVE - REG. SANITARY NO. EE-04466 - IN ALUMINUM BLISTER CODE 227237 - CMDA/EE ANGIE20 2-0 02 1X24+4 EXELTIS PE LOT NO. LF30195A - MFG DATE: 01-25-2023 / EXP. DATE: 01-2025
3006600000
LADEE 0.03MG/3MG EXELTIS S/M IN BOXES - EACH BLISTER CONTAINS 21 YELLOW ACTIVE TABLETS AND 7 INACTIVE TABLETS PHARMACEUTICAL USE - REG. SANITARY NO. EE-04503 - LADEE DROSPIRENONE 3MG* CODE 231228 - DR/EE LADEE 3-0 03MG 1X21+7 EXELTIS PE LOT NO. LF29636A - MFG DATE: 11-25-2022 / 01-24-2023 // EXP. DATE: 11-2024 - *ETNILESTRADIOL 0.03M
EXELTIS PERU SAC

SPAIN
HS-Code
Products
3006600000
DIENILLE 2MG + 0.03MG EXELTIS S/M IN CARDBOARD BOX X 28 COATED TABLETS IN COLORLESS PVC/PVDC ALUMINUM BLISTER PACKAGING* EACH BLISTER CONTAINS 21 WHITE ACTIVE TABLETS AND 7 GREEN INACTIVE TABLETS CODE 227224 - DIE/EE DIENILLE 2-0.03 1X21+7 EXELTIS PE REG. SANITARY NO. EE-04560 - LOT NO. LF31908C - FCH. FROM FAB. 07-19-2023 / FCH. OF EXP. 05-2025
3006600000
SLINDA 4MG EXELTIS S/M IN CARDBOARD BOX WITH 28 COATED TABLETS EACH BOX CONTAINS 24 COLOR ACTIVE TABLETS PHARMACEUTICAL USE - REG. SANITARY NO. EE-08512 - DROSPYRENONE 4MG ED COATED TABLETS LATAM CODE 232243 - DR SLINDA 4MG 1X24+4 EXELTIS PE BATCH NO. LF32670B / MFG DATE: 09-19-2023 / EXP. DATE: 09-2025
3006600000
ANGIE CD 2MG/0.03MG EXELTIS S/M CARDBOARD BOX OF 28 COATED TABLETS EACH BLISTER CONTAINS 21 ACTIVE TABLETS AND 7 TABLETS ORAL CONTRACEPTIVE - REG. SANITARY NO. EE-10572 CODE 238776 - CMDA/EE ANGIE CD2-0.03 1X21 +7 EXELTIS PE LOT NO. LF30952F - MFG. DATE: 04-24-2023 / EXP. DATE: 05-2025
NOT KNOWN

HUNGARY
HS-Code
Products
30066000
LEVONORGESTREL COMPOUND 0.15MG ADMINISTRATION VIA ORAL EACH BLISTER CONTAINS 28 TABLETS
30066000
LEVONORGESTREL COMPOUND 0.15MG VIAORAL ADMINISTRATION EACH BLISTER CONTAINS 28 TABLETS
30066000
LEVONORGESTREL COMPOUND 0. 1 5MG ADMINISTRATION VIA ORAL EACH BLISTER CONTAINS 28 TABLETS
COM GEDEON RICHTER CHILE LTDA

HUNGARY
HS-Code
Products
30066000
LEVONORGESTREL COMPOUND 0. 1 5MG ADMINISTRATION VIA ORAL EACH BLISTER CONTAINS 28 TABLETS
30066000
LEVONORGESTREL COMPOUND 0. 1 5MG ADMINISTRATION VIA ORAL EACH BLISTER CONTAINS 28 TABLETS
Global Export Import Trade Data for each tablet under HS-Code 300660
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 KAZAKHSTAN KAZAKHSTAN | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 PERU PERU | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SPAIN SPAIN | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 HUNGARY HUNGARY | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 TANZANIA TANZANIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
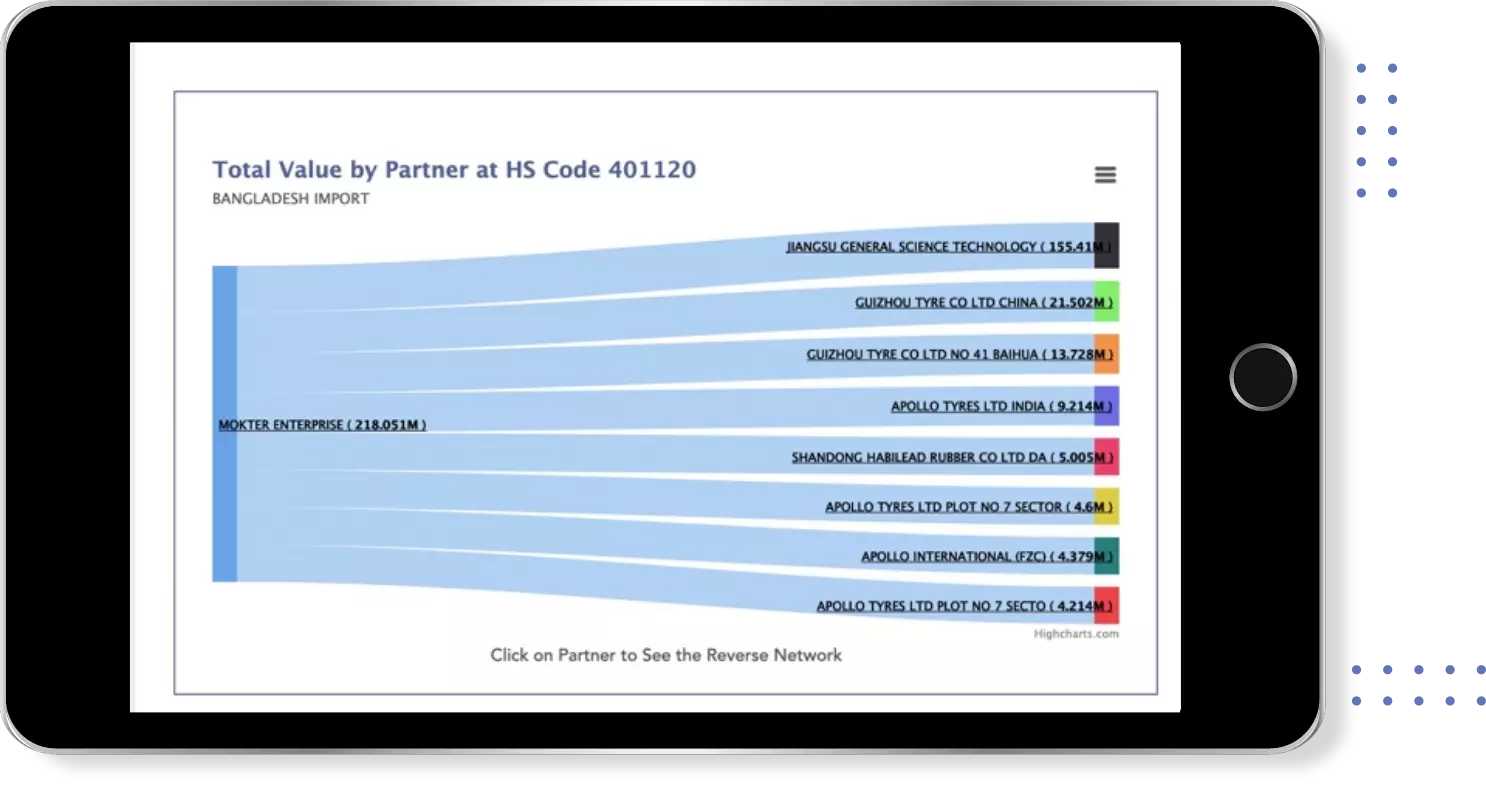
Get most profitable Buyers & Suppliers
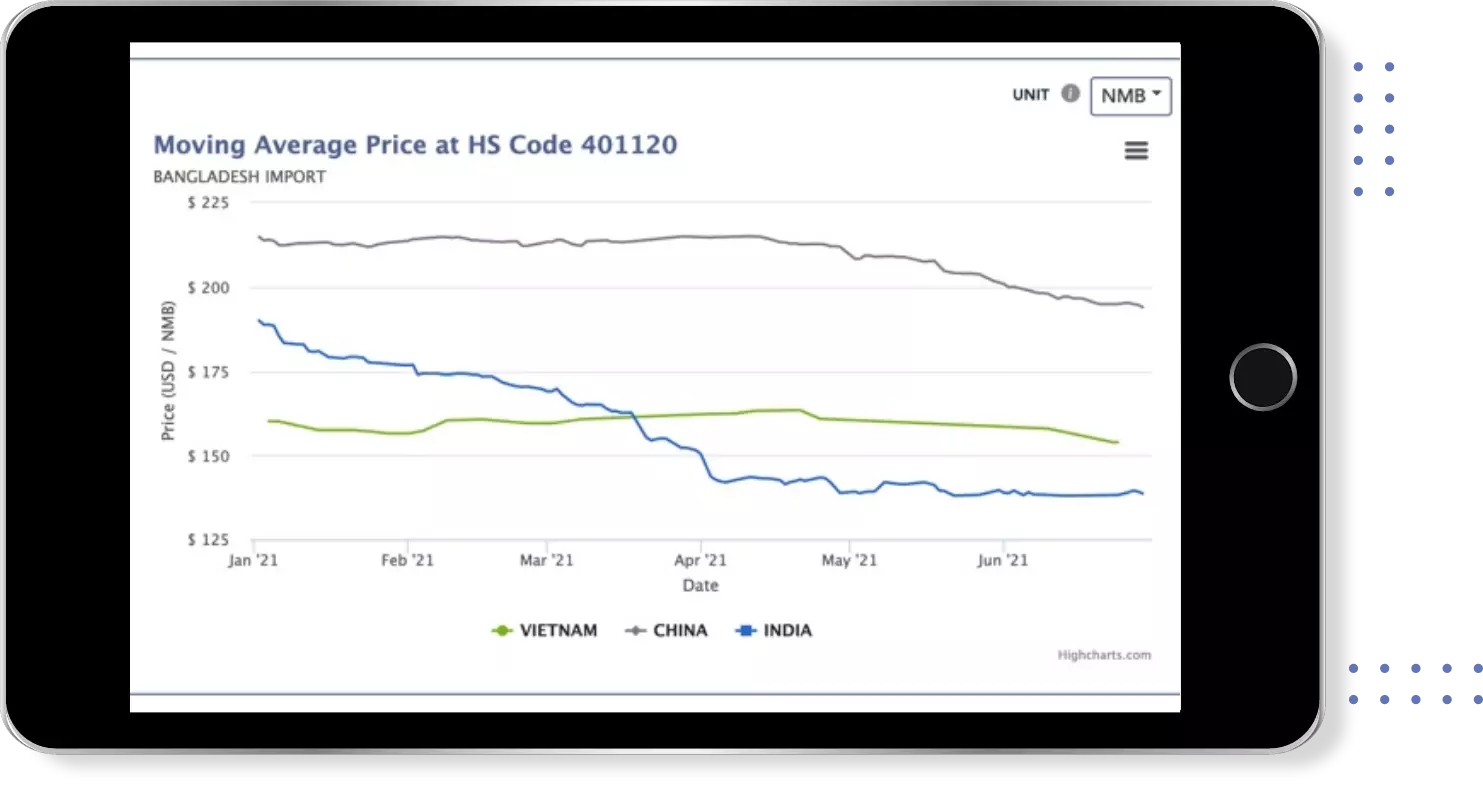
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries