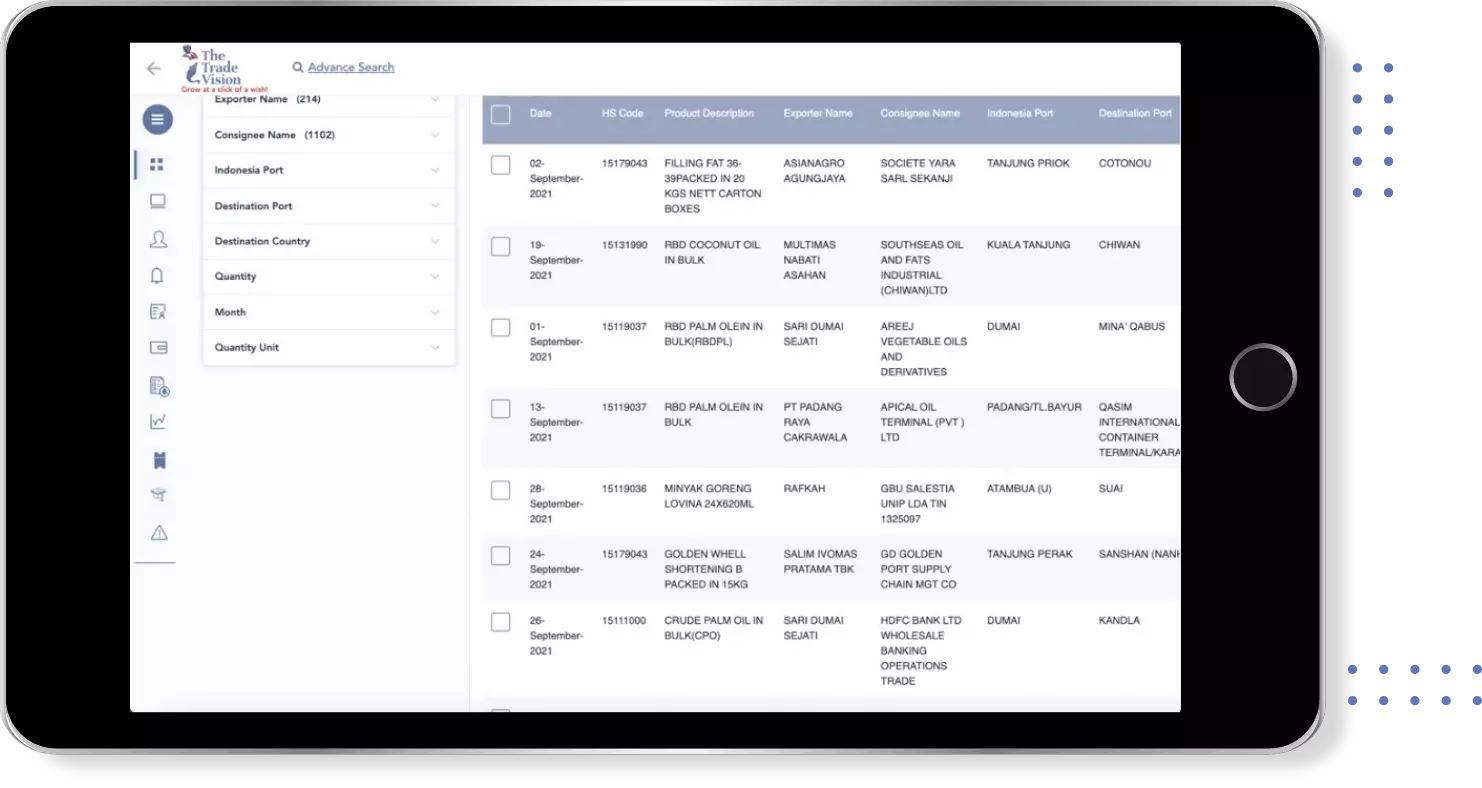
Find verified buyers and sellers of each tablet in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- each tablet hs code 300420 import export data
Overview
- The top 3 supplier countries for HS Code 300420 are “NIGERIA” , “KENYA” , “TANZANIA” ,
- The top 3 Buyer countries for HS Code 300420 are “HUNGARY” , “ARGENTINA” , “CHILE” , .
- The top 3 Importing countries for HS Code 300420 are “BELGIUM” , “CONGO (KINSHASA)” , “SOUTH AFRICA” , .
- The top 3 Exporting countries for HS Code 300420 are “CYPRUS” , “FRANCE” , “HUNGARY” , .
- Top 1 Product Categories for HS Code 300420 are 300420,
The above summary is based on TTV’s Global Export Import data of HS Code 300420, compiled from 180+ countries export import shipments updated till .
- HS Code: 300420
Global Buyers of each tablet under HS-Code 300420
ROCHE FARMA (PERU) SA

HUNGARY
HS-Code
Products
3004201900
MEDICATION S / MS / M MOPHETYL MYCOPHENOLATE 500 MG PROTOCOL CA41705 FOR USE IN CLINICAL TRIALS ONLY. MEDICINE FOR HUMAN USE AGAINST LUPUS ERYTHEMATOSUS CASE WITH THREE BLISTERS EACH OF 10 COATED TABLETS VIA ORAL 30 FILM COATED TABLETS OF MYFENAX 500 MG LOT: 3350920CT 1168805 F.VENC: 09/2023
3004201900
MEDICATION S/MS/M MYCOPHENOLATE MOFETILO 500 MG PROTOCOL CA41705 RD 163-2020-OGITT/INS DEL 05/28/2020 FOR CLINICAL TRIAL USE ONLY. MEDICATION FOR HUMAN USE AGAINST LUPUS ERYTHEMATOSUS BOX WITH THREE BLISTERS EACH OF 10 COATED TABLETS FOR ORAL USE 30 FILM COATED TABLETS OF MYFENAX 500 MG LOT: 2147332 1176698 VCTO: 11.30.2024
3004201900
MEDICATION S/MS/M MYCOPHENOLATE MOFETIL 500 MG PROTOCOL CA41705 FOR USE IN CLINICAL TRIALS ONLY. DRUG FOR HUMAN USE AGAINST LUPUS ERYTHEMATOSUS CARTON WITH THREE BLISTERS EACH OF 10 COATED TABLETS FOR ORAL USE 30 FILM COATED TABLETS OF MYFENAX 500 MG LOT: 3380521CT 1171429 EXPIRY DATE: 05.31.2024
PARTNERS IN HEALTH BRANCH PERU

GERMANY
HS-Code
Products
3004201100
DELAMANID 50 MG TABLETS S/M S/M TABLET COATED / ORAL USE MANUFACTURER: OTSUKA NOVEL PRODUCTS GMBH - GERMANY / LOT N?00027352 / VCTO. 31-JAN-25 DELAMANID 50 MG TABLETS 48 TABLETS PER BOX 672 TABLETS PER CONTAINER (14 INTERNAL BOXES EACH WITH 48 TABLETS)
3004201100
DELAMANIDA 5 MG TABLETS S/M S/M DISPERSIBLE TABLET / ORAL WAY MANUFACTURER: OTSUKA NOVEL PRODUCTS GMBH - GERMANY / LOT N?00032189 - VCTO. 31-MAR-25 DELAMANID 5 MG TABLET 48 TABLETS PER BOX 672 TABLETS PER CONTAINER (14 INTERNAL BOXES EACH WITH 48 TABLETS)
3004201100
DELAMANIDA 5 MG TABLETS S/M S/M DISPERSIBLE TABLET / ORAL WAY MANUFACTURER: OTSUKA NOVEL PRODUCTS GMBH - GERMANY / LOT N?00032189 - VCTO. 31-MAR-25 DELAMANID 5 MG TABLET 48 TABLETS PER BOX 240 TABLETS PER CONTAINER (05 INTERNAL BOXES EACH WITH 48 TABLETS)
ASTRAZENECA PERU SA

SWEDEN
HS-Code
Products
3004201100
OSIMERTINIB AZD9291 80MG ASTRAZENECA AB S / M BOTTLES WITH 35 TABLETS IN EACH ONE FOR ORAL ADMINISTRATION. LOT 25532.18 / 1 EXPIRY DATE: MAY / 31/2024 OSIMERTINIB AZD9291 80MG MEDICINE TO BE USED FOR USE IN CLINICAL RESEARCH ONLY
3004201100
OSIMERTINIB AZD9291 80MG ASTRAZENECA AB S / M BOTTLES WITH 35 TABLETS IN EACH ONE FOR ORAL ADMINISTRATION. LOT 25532.18 EXPIRY DATE: MAY / 31/2024 OSIMERTINIB AZD9291 80MG MEDICINE TO BE USED FOR USE IN CLINICAL RESEARCH ONLY
3004201100
OSIMERTINIB AZD9291 80MG ASTRAZENECA AB S / M BOTTLES WITH 35 TABLETS IN EACH ONE FOR ORAL ADMINISTRATION. LOT 25532.18 / 1 EXPIRY DATE: MAY / 31/2024 OSIMERTINIB AZD9291 80MG MEDICINE TO BE USED FOR USE IN CLINICAL RESEARCH ONLY
ANIMAL WELFARE S A

ARGENTINA
HS-Code
Products
30042099
600 000)UND OF OTHER MEDICINES FOR VETERINARY USE THAT CONTAIN OTHER ANTIBIOTICS IN; DOXYCYCLINE KUALCOS PALATABLE TABLETS 1 00 MG. 1 0 BLISTER X 1 0 TABLETS EACH. L043 V09/24
Global Suppliers of each tablet under HS-Code 300420
LABORATORIOS SAVAL SA

PARAGUAY
HS-Code
Products
30042014
ANTIBIOTIC EUROMYCIN TABLETS X 1 4 EACH TABLET CONTAINS 500 MG OF CLARITHROMYCIN DRUG FOR HUMAN USE. R-1 7
30042011
CEPHRAX ANTIBIOTIC TABLETS X 1 0 EACH TABLET CONTAINS 200 MG OF CEFPODOXIMA PROXETILMEDICAMENTO HUMAN USE. R-1 7
30042014
ANTIBIOTIC EUROMYCIN TABLETS X 1 4 EACH TABLET CONTAINS 500 MG OF CLARITHROMYCIN DRUG FOR HUMAN USE. R-1 7
SAVAL LABORATORIES S A

DOMINICAN REPUBLIC
HS-Code
Products
30042014
ANTIBIOTICOTREX TABLETS X 6 EACH TABLET CONTAINS 500 MG OF AZITROMYCIN DRUG FOR HUMAN USE. R-1 7
30042019
ANTIHYPERTENSIVE VALAXAM TABLETS X 30 EACH TABLET CONTAINS 1 60 MG VALSARTANY 1 0 MG AMLODIPIN DRUG FOR HUMAN USE. R-1 7
30042019
POLYIVITAMINIC WITH MINERALS OFFER TABLETS X 30 EACH COMP. CONT. 2MG VIT. A 200MG VIT. C 75MG VIT. E 5MG VIT. B2 30MG ZN 0.02MG SE 2MG CU 5MG MN 2MG LUTEIN / ZEAXANTINAMED
SAVAL LABORATORIES SA

ECUADOR
HS-Code
Products
30042014
ANTIBIOTICOTREX TABLETS X 6 EACH TABLET CONTAINS 500 MG OF AZITROMYCIN DRUG FOR HUMAN USE. R-1 7
30042014
ANTIBIOTICOTREX TABLETS X 3 EACH TABLET CONTAINS 500 MG OF AZITROMYCIN MEDICINE FOR HUMAN USE. R-1 7
WORLD WIDE TRADERS

SINGAPORE
HS-Code
Products
30042000
DIANE-35 TABLET (1X21 TABLET EACH BOX)(CYPROTERONE ACETATE 2MG – ETHINYLESTRADIOL 0.035MG)QTY: 6400 PCS BOXES)
30042000
DIANE-35 TABLET (CYPROTERONE ACETATE 2MG – ETHINYLESTRADIOL 0.035MG)QTY: 6000 BOXES (1X21 TABLET EACH BOX)
30042000
DIANE-35 TABLET (CYPROTERONE ACETATE 2MG – ETHINYLESTRADIOL 0.035MG)QTY: 7000 BOXES (1X21 TABLET EACH BOX)
Global Export Import Trade Data for each tablet under HS-Code 300420
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 KAZAKHSTAN KAZAKHSTAN | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 BELGIUM BELGIUM | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
.gif) CONGO (KINSHASA) CONGO (KINSHASA) | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SOUTH AFRICA SOUTH AFRICA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
Get most profitable Buyers & Suppliers
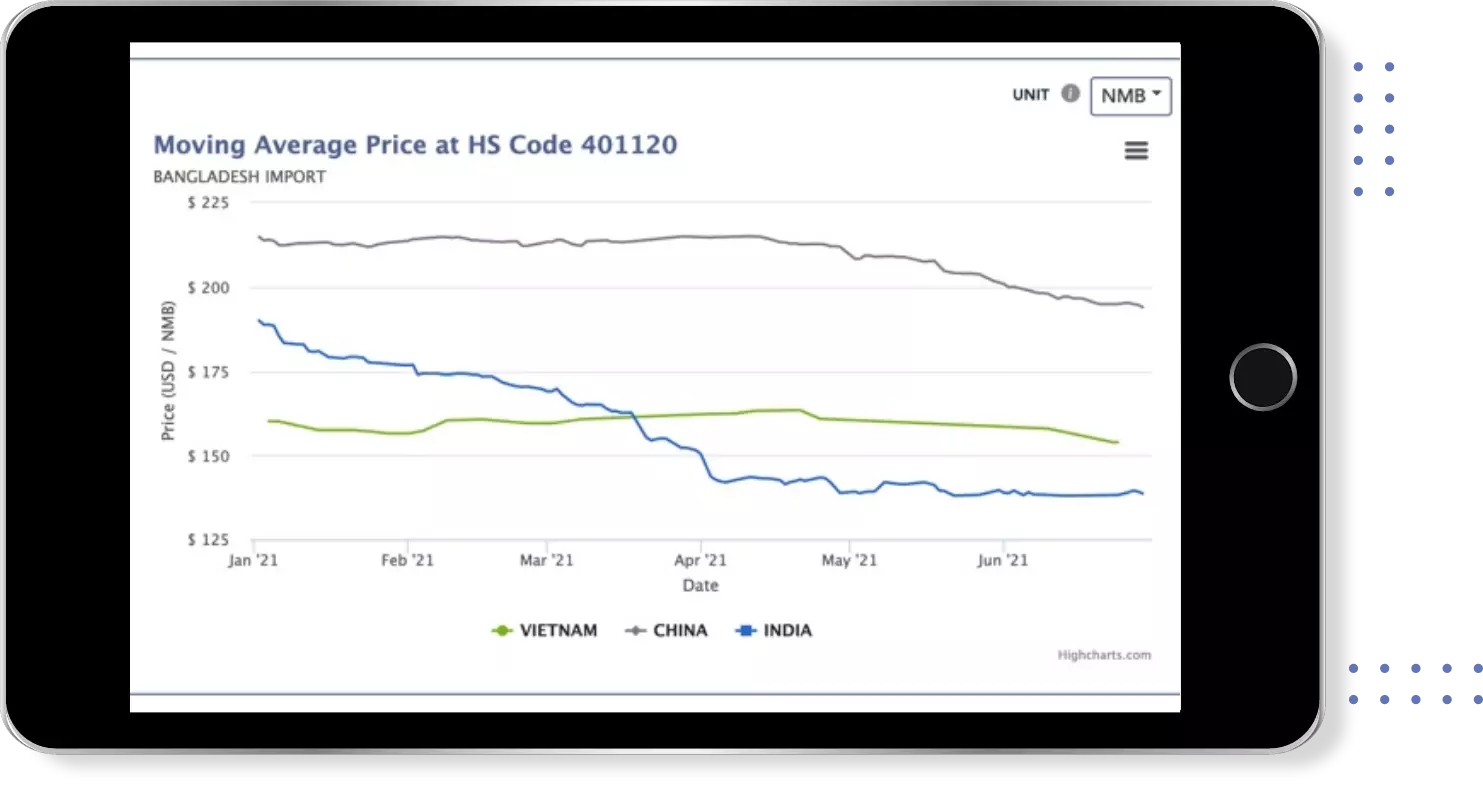
Instant insights on Price , Quantity & Value trends
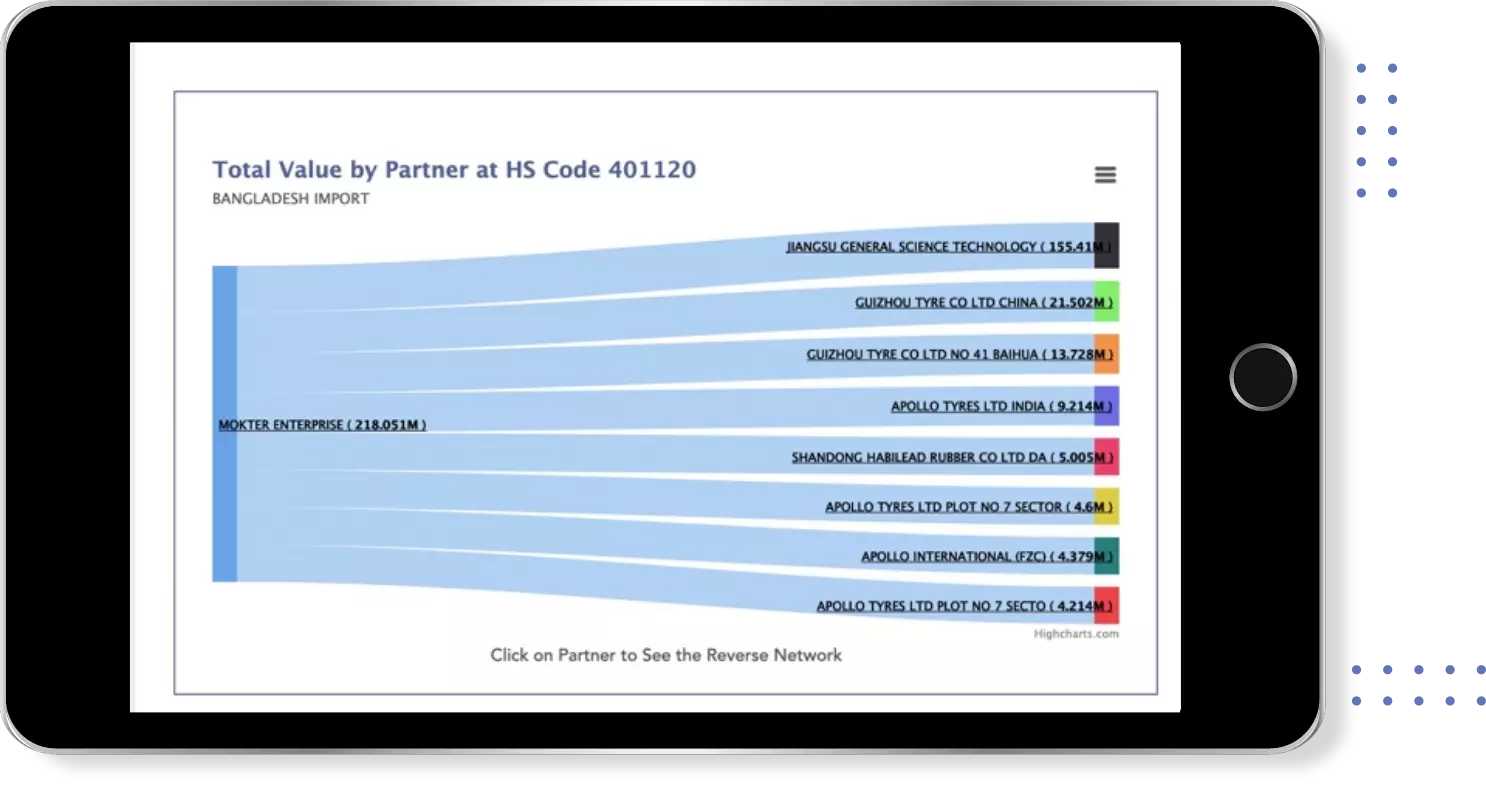
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries