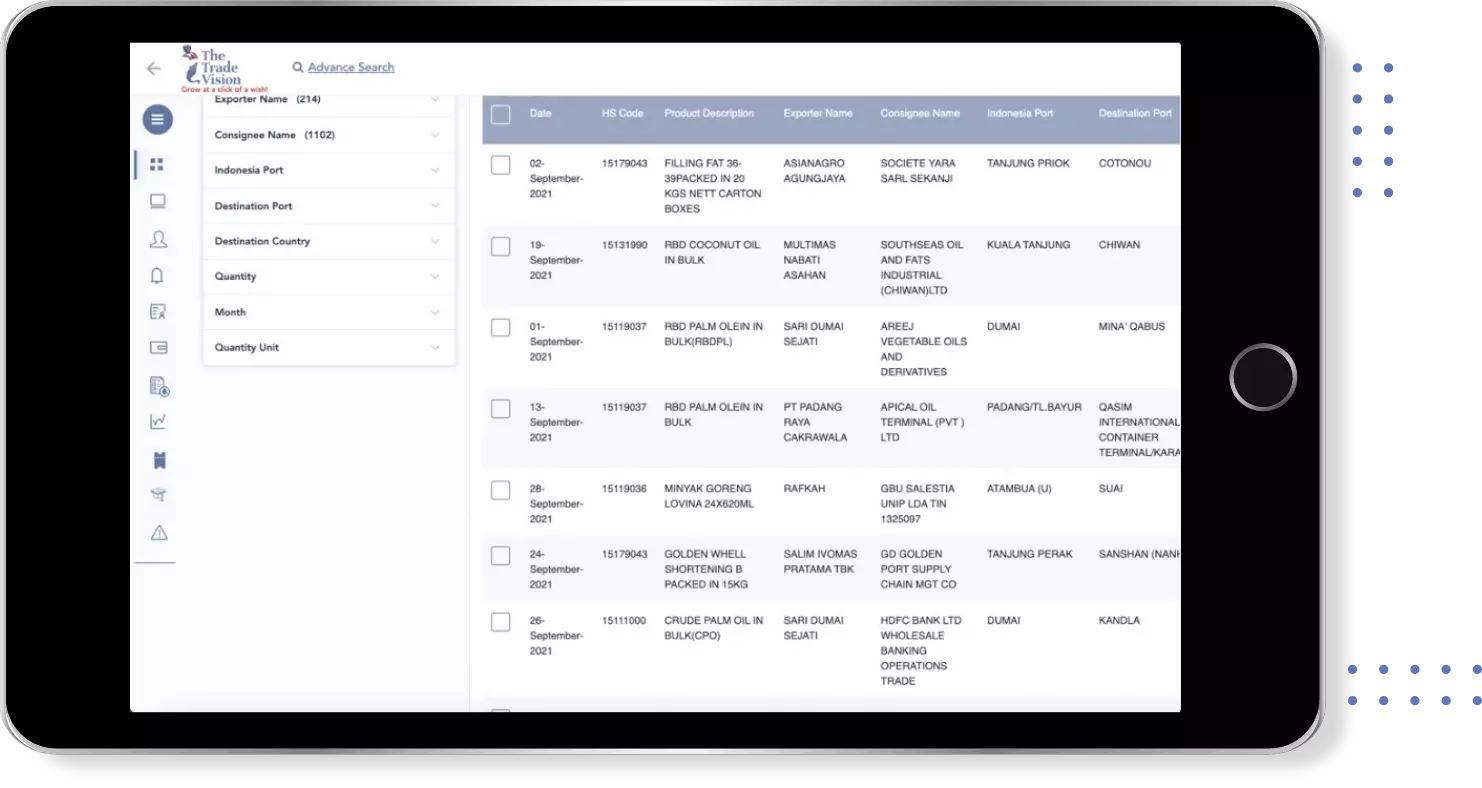
Find verified buyers and sellers of abacavir tab in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- abacavir tab import export data
Overview
- The top 3 supplier countries for abacavir tab are “PERU” , “ECUADOR” , “UNITED STATES OF AMERICA” ,
- The top 3 Buyer countries for abacavir tab are “UKRAINE” , “PERU” , “ECUADOR” , .
- Top 3 Product Categories for abacavir tab are 300490, 300410, 300420,
The above summary is based on TTV’s Global Export Import data of abacavir tab, compiled from 180+ countries export import shipments updated till .
Global Suppliers of Abacavir tab
N A

UNITED KINGDOM
HS-Code
Products
300490410000
ANTIVIRAL||ACTIVE PRINCIPLE DOLUTEGRAVIR ABACAVIR LAMIVUDINE FOR HUMAN USE || TRIUMEQ|| TABLET 50/600 / 300MG 1X30
300490410000
ANTIVIRAL||ACTIVE PRINCIPLE DOLUTEGRAVIR ABACAVIR LAMIVUDINE FOR HUMAN USE || TRIUMEQ|| TABLET 50/600 / 300MG 1X30
300490440000
ANTIVIRAL||ACTIVE PRINCIPLE DOLUTEGRAVIR ABACAVIR LAMIVUDINE FOR HUMAN USE ||TRIUMEQ||TABLETS 50/600/300MG 1X30
KUEHNE NAGEL LLC

UNITED ARAB EMIRATES
HS-Code
Products
300490000000
ABACAVIR/LAMIVUDINE 120/60 MG DISPERSIBLE TABLET
300490000000
ABACAVIR/LAMIVUDEDINE120/60 MG DISPERATE TABLET
300490000000
ABACAVIR/LAMIVUDINE 120/60 MG DISPERSIBLEN TABLETS
ESPEE BIOPHARMA AND FINE CHEM LLC

UNITED STATES OF AMERICA
HS-Code
Products
300490990000
TRIUMEQ (ABACAVIR DOLUTEGRAVIR AND LAMIVUDINE) (600 MG/50 MG /300 MG) (8 X 30240 TABS) ( FOR R & D TESTING PURPOSE )
300490990000
TRIUMEQ (ABACAVIR DOLUTEGRAVIR AND LAMIVUDINE) (600MG/50MG/300MG) (30 X 10300 TABLETS)(FOR R & D TESTING PURPOSE)
300490990000
TRIUMEQ PD (ABACAVIR SULFATE 60MG + DOLUTEGRAVIR SODIUM 5MG+LAMIVUDINE 30MG TABLET FILM COATED (R&D PURPOSE)
GLAXOSMITHKLINE R AND D LTD

UNITED KINGDOM
HS-Code
Products
300490990000
DOLUTEGRAVIR (GSK1349572)/ABACAVIR/ LAMIVUDINE DISPERSIBLE TABLETS 5 MG/60 MG/30 MG 90 TABLETS PER BOTTLE
300490990000
DOLUTEGRAVIR (GSK1349572)/ABACAVIR/ LAMIVUDINE DISPERSIBLE TABLETS 5 MG/60 MG/30 MG 90 TABLETS PER BOTTLE
300490990000
DOLUTEGRAVIR(GSK1349572)ABACAVIR/LAMIVUDINE DISPERSIBLE TABLETS 5MG/60MG/30MG(QTY: 1PACK PK SIZE:90S PRICE:GBP 49.08/PK)
Global Export Import Trade Data for abacavir tab
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 UKRAINE UKRAINE | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SOUTH AFRICA SOUTH AFRICA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 SOUTH AFRICA SOUTH AFRICA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 NETHERLANDS NETHERLANDS | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
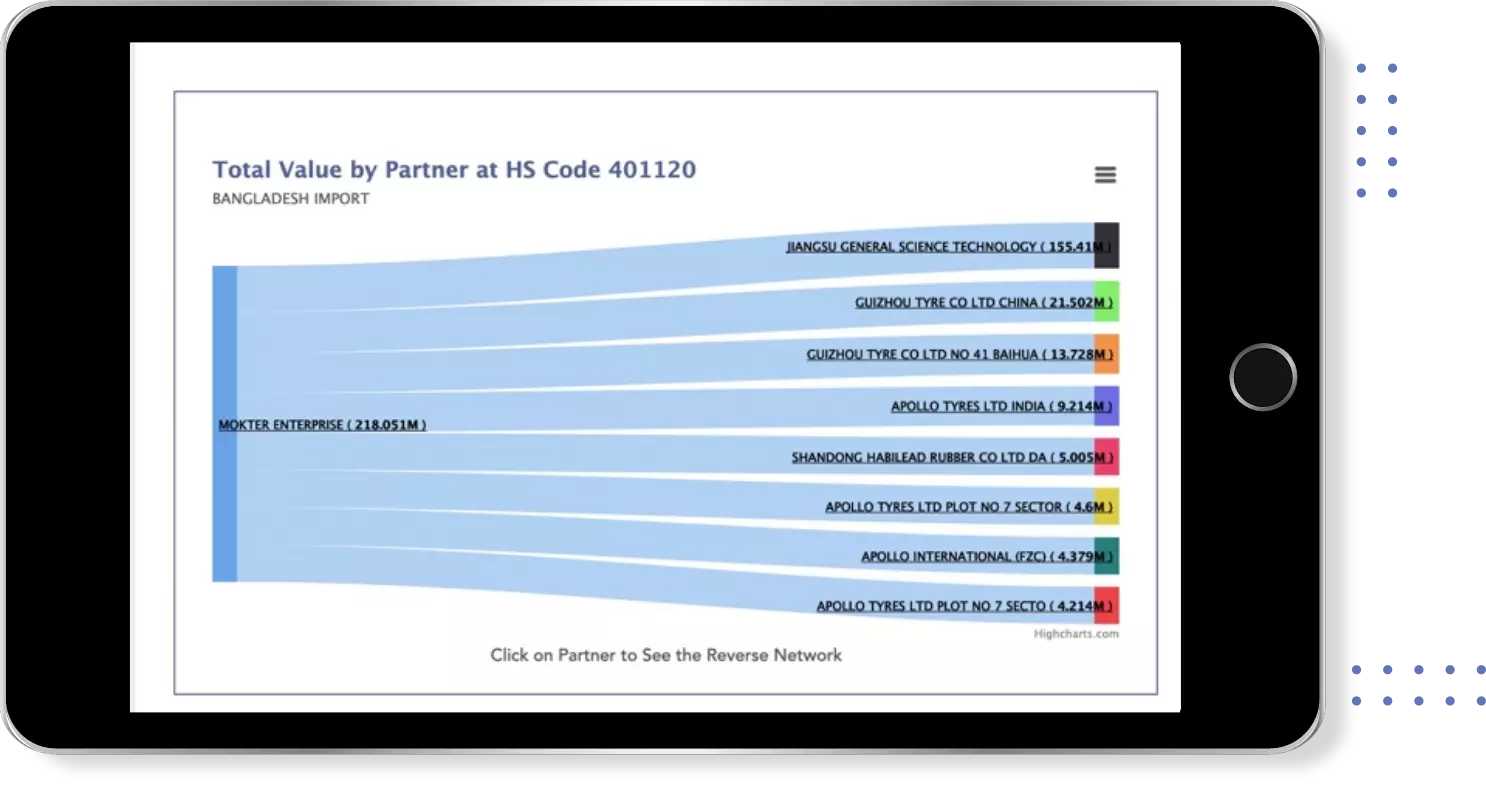
Get most profitable Buyers & Suppliers
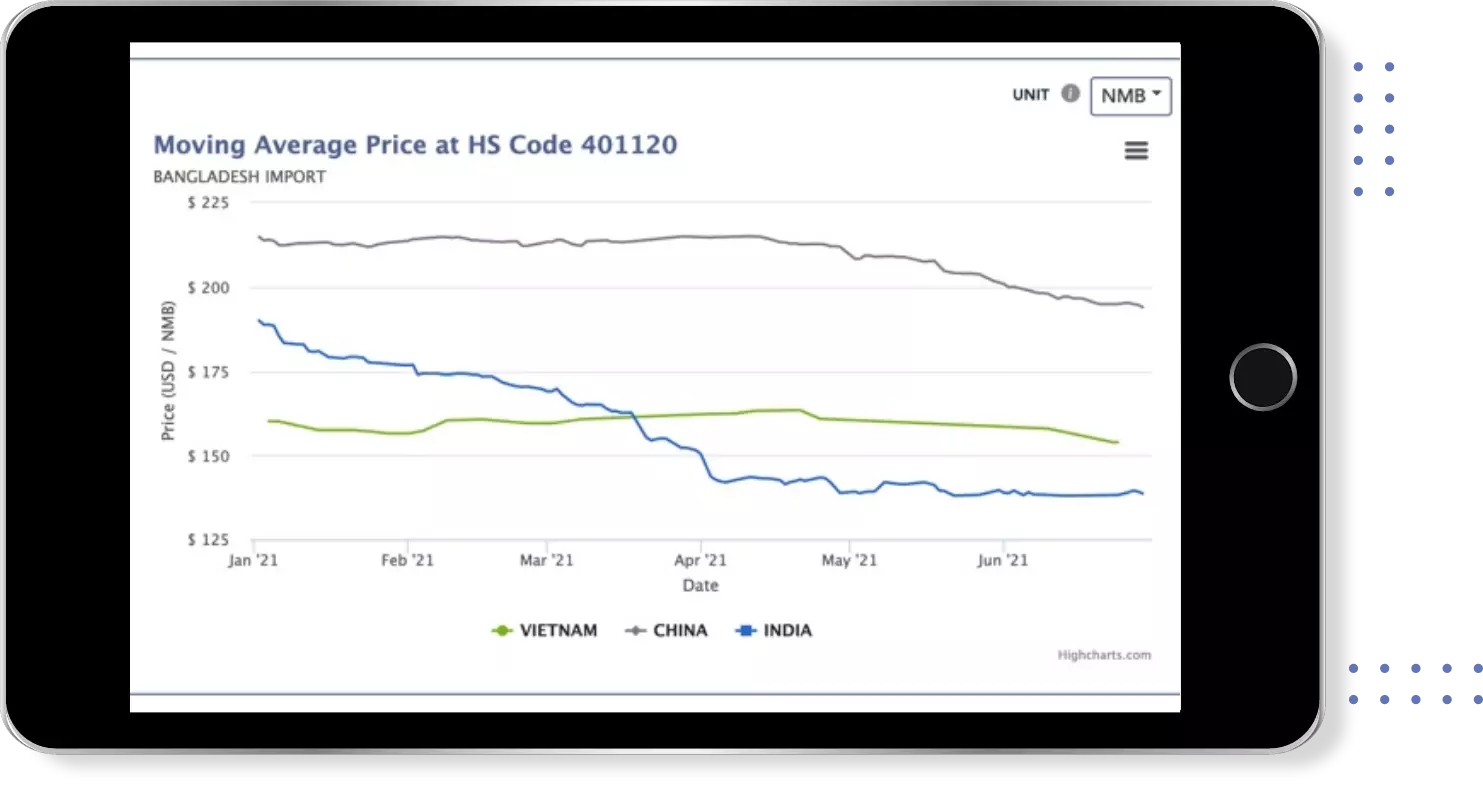
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries