Find verified buyers and sellers of azithromycin susp in 180+ countries along with their valid phone numbers and email ids.
- Home
- Global trade data
- azithromycin susp import export data
Overview
- The top 3 supplier countries for azithromycin susp are “PAKISTAN” , “UZBEKISTAN” , “VIETNAM” ,
- The top 3 Buyer countries for azithromycin susp are “UZBEKISTAN” , “UKRAINE” , “KAZAKHSTAN” , .
- Top 3 Product Categories for azithromycin susp are 300420, 300490, 294190,
The above summary is based on TTV’s Global Export Import data of azithromycin susp, compiled from 180+ countries export import shipments updated till .
Global Buyers of Azithromycin susp
TOO RATIOFARMS KAZAKHSTAN

KAZAKHSTAN
HS-Code
Products
3004200002
1 . SUMAMED® (AZITHROMYCIN DIHYDRATE) POWDER FOR ORAL SUSPENSION PREPARATION POWDER IN A VIAL 1 VIAL IN A CARDBOARD PACK 100 MG/5 ML
3004200002
1 . SUMAMED® (AZITHROMYCIN DIHYDRATE) POWDER FOR ORAL SUSPENSION PREPARATION POWDER IN A VIAL 1 VIAL IN A CARDBOARD PACK 100 MG/5 ML
3004200002
1 . SUMAMED® FORTE (AZITHROMYCIN DIHYDRATE) POWDER FOR PREPARING SUSPENSION FOR ORAL ADMINISTRATION 1 6. 7 G POWDER IN VIALS 1 VIAL IN A CARDBOARD PACK 200 MG / 5 ML
TOO RAUZA ADE

KAZAKHSTAN
HS-Code
Products
3004200002
1. AZIDRATE™ (AZITHROMYCIN MONOHYDRATE) 100 MG/5 ML POWDER FOR PREPARATION OF SUSPENSION FOR ORAL ADMINISTRATION. 17.10 G OF THE PREPARATION IN POLYETHYLENE BOTTLES. 1 BOTTLE TOGETHER WITH A MEASURING SYRINGE IN A CARDBOARD PACK. S/Y 03/2025
3004200002
1. AZIDRATE® (AZITHROMYCIN) 200MG/5ML POWDER FOR PREPARATION OF SUSPENSION FOR ORAL ADMINISTRATION. 24.80 G OF THE PREPARATION IN POLYETHYLENE BOTTLES. 1 BOTTLE TOGETHER WITH A MEASURING SYRINGE IN A CARDBOARD PACK. C/Y 12/2024
3004200002
1. AZIDRATE® (AZITHROMYCIN) 100 MG/5 ML POWDER FOR PREPARATION OF SUSPENSION FOR ORAL ADMINISTRATION. 17.10 G OF THE PREPARATION IN POLYETHYLENE BOTTLES. 1 BOTTLE TOGETHER WITH A MEASURING SYRINGE IN A CARDBOARD PACK. S/Y 03/2025
OOO GRAND PHARM TRADE

UZBEKISTAN
HS-Code
Products
3004200002
1 . AZIMAK POWDER FOR APPLICATION SUSP. FOR RECEPTION INTERNAL 200 MG/5 ML. 30ML. FL. NO. 1 SER. NO1 1 1 01 524 TO 01 . 10. 2024 - 9860 UP. INN: AZITHROMYCIN. IZG. WORLD MEDICINE ILAC SAN. VE TIC. AS TURKEY FOR GM PHARMACEUTICALS LTD GEORGIA - WEIGHT: 669.3 KG 2. SEATS 1 1 8
3004200002
1 . AZIMAK POWDER FOR APPLICATION SUSP. FOR RECEPTION INTERNAL 200 MG/5 ML. 30ML. FL. NO. 1 SER. NO1 1 1 01 528 TO 01 . 10. 2024 - 9883 UP. INN: AZITHROMYCIN. IZG. WORLD MEDICINE ILAC SAN. VE TIC. AS TURKEY FOR GM PHARMACEUTICALS LTD GEORGIA - WEIGHT: 659 KG 2. SEATS 1 1 8 1 1
3004200002
1 . AZIMAK POWDER FOR APPLICATION SUSP. FOR RECEPTION INTERNAL 200 MG/5 ML. 30ML. FL. NO. 1 SER. NO1 1 1 01 526 TO 01 . 10. 2024 - 9779 UP. INN: AZITHROMYCIN. IZG. WORLD MEDICINE ILAC SAN. VE TIC. AS TURKEY FOR GM PHARMACEUTICALS LTD GEORGIA - WEIGHT: 652.2 KG 2. SEATS 1 1 7
RIN PHARM LLP (RIN PHARM)

KAZAKHSTAN
HS-Code
Products
3004200002
1 . MEDICINE: ZIROMIN POWDER FOR PREPARING SUSPENSION FOR ORAL ADMINISTRATION 200MG/5ML NO1. PACKAGING: POWDER ON 24 G IN A BOTTLE. 1 VIAL WITH A MEASURING SYRINGE IN A CARDBOARD BOX. COMPOSITION OF THE ACTIVE SUBSTANCES: AZITHROMYCIN DIHYDRATE
3004200002
1 . MEDICINE: ZYROMIN POWDER FOR PREPARING SUSPENSION FOR ORAL ADMINISTRATION 200MG / 5ML NO. 1 IN 1 PACKAGING POWDER IN POLYETHYLENE VIAL WITH MEASURING SYRINGE ACTIVE QUESTION: AZITHROMYCIN
3004200002
1 . MEDICINE: ZYROMIN POWDER FOR PREPARING SUSPENSION FOR ORAL ADMINISTRATION 200MG / 5ML NO. 1 POWDER 24 G IN A VIAL. 1 VIAL WITH A MEASURING SYRINGE IN A CARDBOARD BOX ACTIVE QUESTION: AZITHROMYCIN DIHYDRATE
Global Suppliers of Azithromycin susp
PFIZER SERVICE CO BV

ITALY
HS-Code
Products
300420310000
MEDICINE: ZITROMAX (AZITHROMYCIN (AS AZITHROMYCIN DIHYDRATE) 200MG/5ML). BOX OF 1 VIAL 600MG/15ML; POWDER FOR ORAL SUSPENSION. MANUFACTURER: SEPTEMBER 14 2023. EXPIRY DATE: SEPTEMBER 12 2025. BATCH: D38003. REGISTRATION NUMBER:VN-21930- 19.HSX:HAUPT PHARMA LATINA SRL
300420310000
MEDICINE: ZITROMAX (AZITHROMYCIN (AS AZITHROMYCIN DIHYDRATE) 200MG/5ML). BOX OF 1 VIAL 600MG/15ML; POWDER FOR ORAL SUSPENSION. MANUFACTURER: SEPTEMBER 22 2023. EXPIRY DATE: SEPTEMBER 20 2025. BATCH: D39200. REGISTRATION NUMBER:VN-21930- 19.HSX:HAUPT PHARMA LATINA SRL
300420310000
MEDICINE: ZITROMAX (AZITHROMYCIN (AS AZITHROMYCIN DIHYDRATE) 200MG/5ML). BOX OF 1 VIAL 600MG/15ML; POWDER FOR ORAL SUSPENSION. MANUFACTURER: AUGUST 4 2023. EXPIRY DATE: AUGUST 2 2025. BATCH: D32800. REGISTRATION NUMBER:VN-21930- 19.HSX:HAUPT PHARMA LATINA SRL
PLIVA CROATIA LTD

CROATIA
HS-Code
Products
300420000200
1 . SUMAMED® FORTE (AZITHROMYCIN DIHYDRATE) POWDER FOR ORAL SUSPENSION 35.6 G POWDER IN VIALS 1 VIAL IN A CARDBOARD PACK 200 MG/5 ML
300420000200
1 . SUMAMED® FORTE (AZITHROMYCIN DIHYDRATE) POWDER FOR PREPARING SUSPENSION FOR ORAL ADMINISTRATION 29.3 G POWDER IN VIALS 1 VIAL IN A CARDBOARD PACK 200 MG / 5 ML
300420000200
1 . SUMAMED® (AZITHROMYCIN DIHYDRATE) POWDER FOR ORAL SUSPENSION PREPARATION POWDER IN A BOTTLE 1 BOTTLE IN A CARDBOARD PACK 100 MG / 5 ML
SANDOZ AG

ROMANIA
HS-Code
Products
300420310000
BINOZYT DRUG 200MG / 5ML (AZITHROMYCIN (AZITHROMYCIN MONOHYDRATE EQUIVALENT) 200MG / 5ML). BOX 1 VIAL 15 ML; POWDER ORAL SUSPENSION. BATCH: LB6790. NSX: 01/2021. HSD: 01/2024. SDK: VN-22179-19
300420310000
BINOZYT MEDICINE 200MG/5ML (AZITHROMYCIN (EQUIVALENT TO AZITHROMYCIN MONOHYDRATE) 200MG/5ML). BOX OF 1 BOTTLE OF 15 ML; POWDER FOR ORAL SUSPENSION. PHONE NUMBER: VN-22179-19. BATCH: NA1438. PRODUCTION: MAY 2023. HSD: 05/2026
300420310000
BINOZYT 200MG/ 5ML (AZITHROMYCIN (EQUIVALENT TO AZITHROMYCIN MONOHYDRATE) 200MG/ 5ML). BOX OF 1 VIAL OF 15 ML; POWDER FOR ORAL SUSPENSION. BATCH: LB6789. NSX: 01/2021. HSD: 01/2024. SDK: VN-22179-19
ACTAVIS INTERNATIONAL LTD (MALTA)

CROATIA
HS-Code
Products
300420000200
1. SUMAMED® FORTE (AZITHROMYCIN DIHYDRATE) POWDER FOR THE PREPARATION OF SUSPENSION FOR ORAL TAKEN 35.6 G OF POWDER IN BOTTLES 1 BOTTLE IN A CARDBOARD PACK 200 MG/5 ML SERIES NO. 1626093: MANUFACTURER: PLIVA HRVATSKA D. O.O. TRADEMARK: NO BRANDS:
300420000200
1. SUMAMED® FORTE (AZITHROMYCIN DIHYDRATE) POWDER FOR THE PREPARATION OF SUSPENSION FOR ORAL TAKEN 29.3 G OF POWDER IN BOTTLES 1 BOTTLE IN A CARDBOARD PACK 200 MG/5 ML SERIES NO. 1539083: MANUFACTURER: PLIVA HRVATSKA D. O.O. TRADEMARK: NO BRANDS:
300420000200
1. SUMAMED® FORTE (AZITHROMYCIN DIHYDRATE) POWDER FOR THE PREPARATION OF SUSPENSION FOR ORAL TAKEN 29.3 G OF POWDER IN BOTTLES 1 BOTTLE IN A CARDBOARD PACK 200 MG/5 ML SERIES NO. 1539083: MANUFACTURER: PLIVA HRVATSKA D. O.O. TRADEMARK: NO BRANDS:
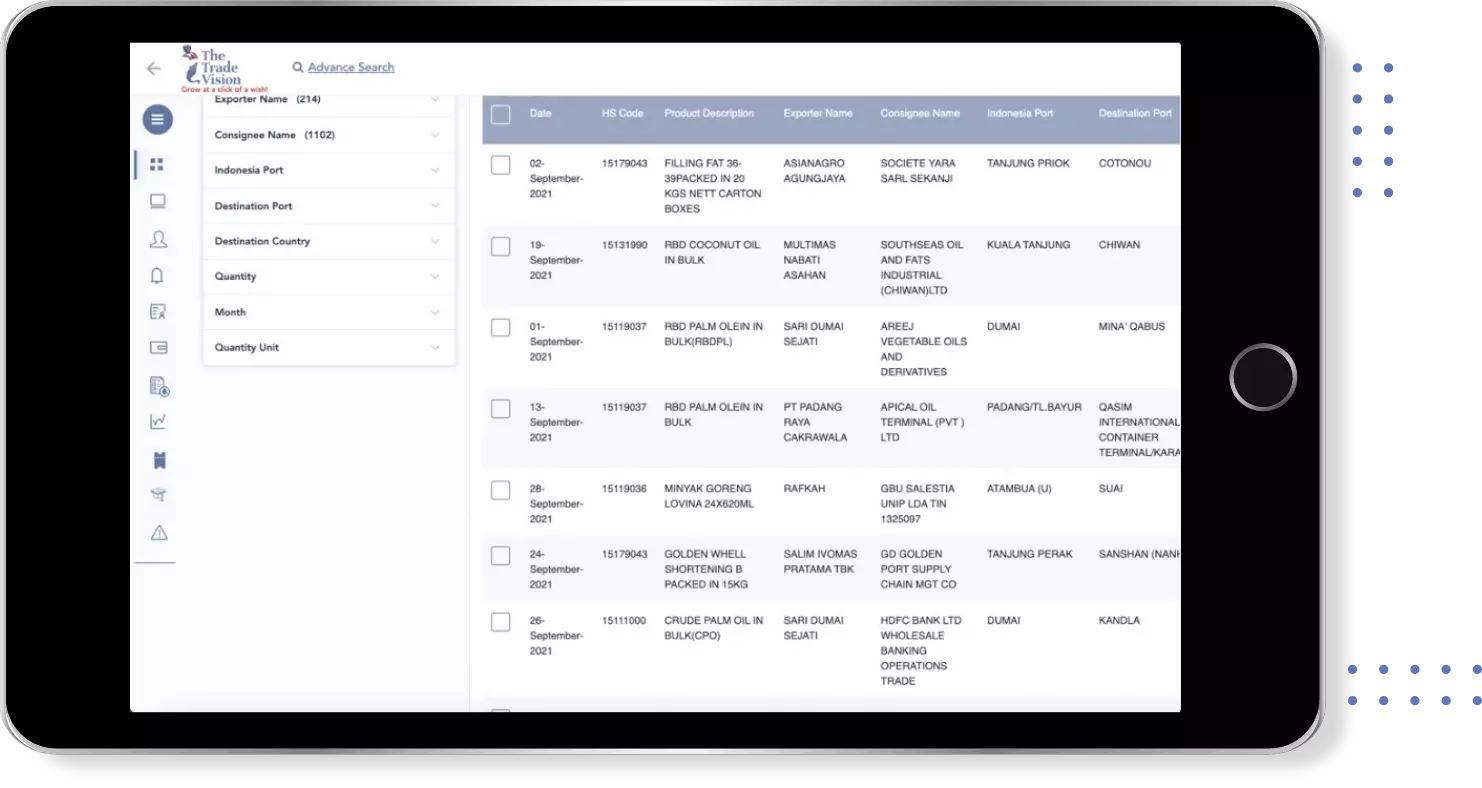
Global Export Import Trade Data for azithromycin susp
Country | Trade Type | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 INDIA INDIA | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 INDIA INDIA | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 VIETNAM VIETNAM | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 ITALY ITALY | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 BANGLADESH BANGLADESH | Import Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
 BANGLADESH BANGLADESH | Export Data | ||||||||||||||||||||||
| |||||||||||||||||||||||
Empower your business with Superior Trade Analysis used by the Top Industry Leaders at a click of a few buttons
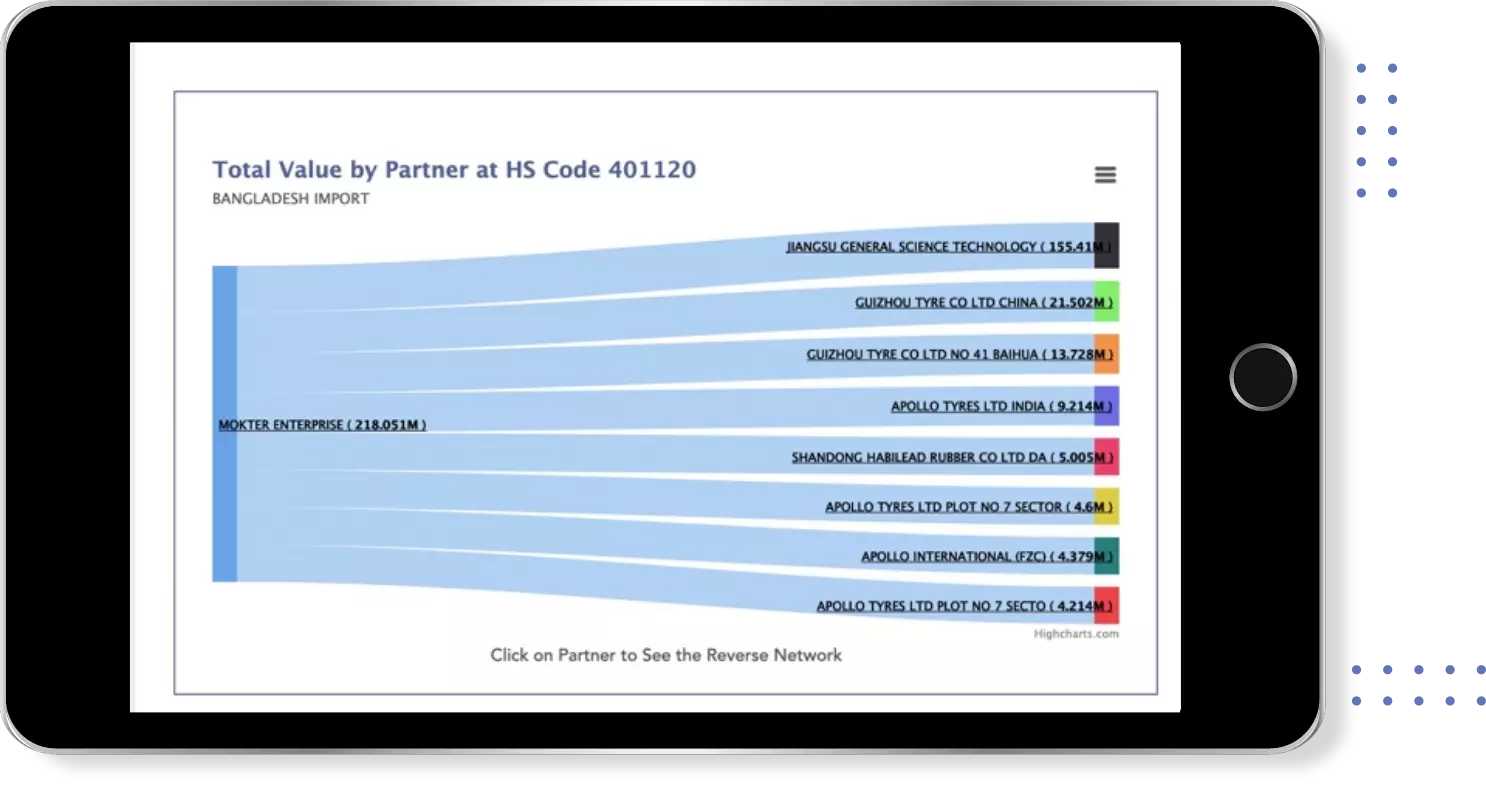
Get most profitable Buyers & Suppliers
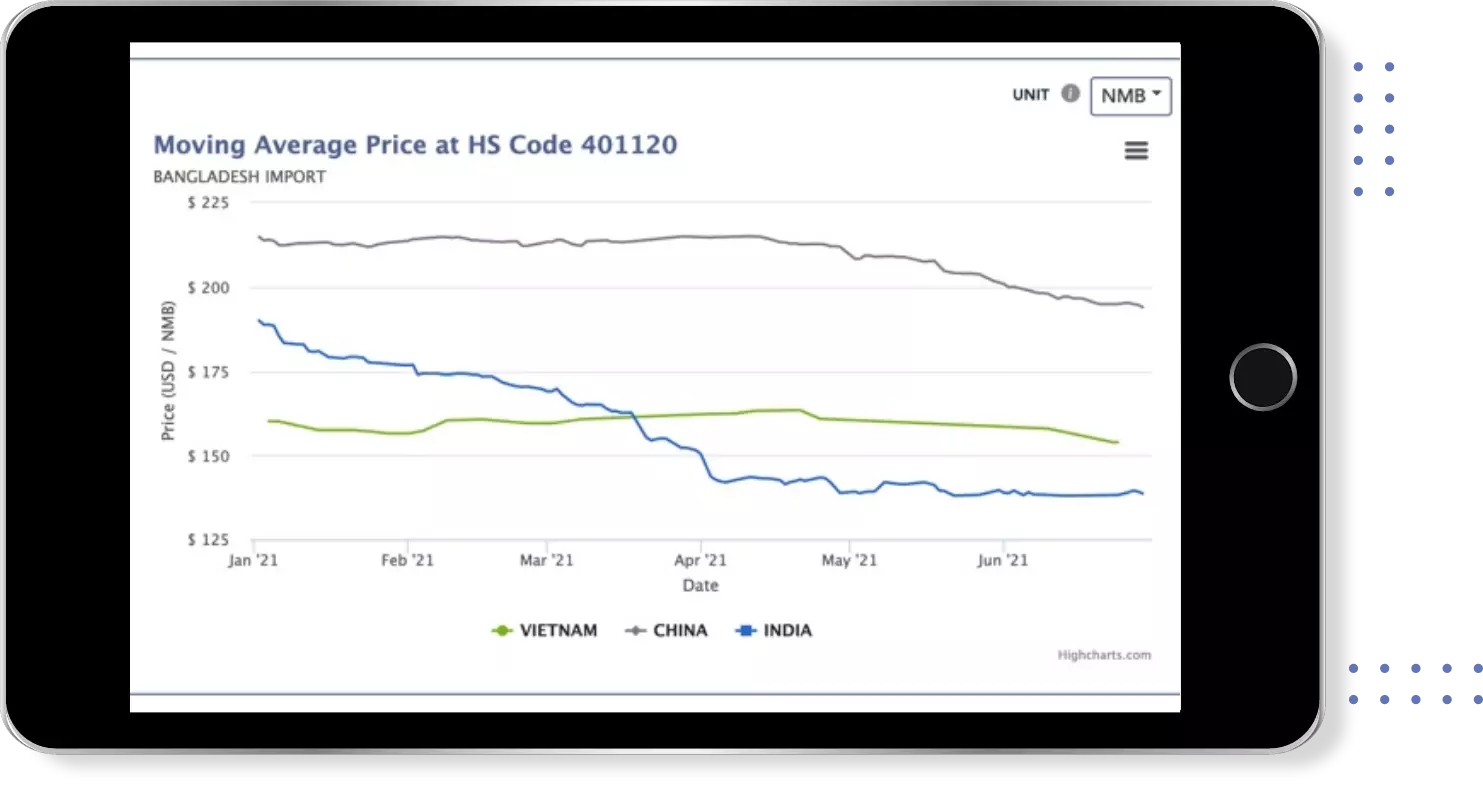
Instant insights on Price , Quantity & Value trends
Identify Top Countries, Top Exporters, Top Importers
Identify most Profitable New Countries to enter
Export Import shipment Data of 180+ Countries